Efavirenz
 | |
 | |
| Names | |
|---|---|
| Pronunciation | /ɪˈfævɪrɛnz/ i-FAV-i-renz |
| Trade names | Sustiva, others[1] |
| |
| Clinical data | |
| Main uses | HIV/AIDS[2] |
| Pregnancy category |
|
| Routes of use | By mouth (capsules, tablets) |
| Onset of action | 3–5 hours |
| Defined daily dose | 600 mg[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699004 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 40–45% (under fasting conditions) |
| Protein binding | 99.5–99.75% |
| Metabolism | Liver (CYP2A6 and CYP2B6-mediated) |
| Elimination half-life | 40–55 hours |
| Excretion | Urine (14–34%) and feces (16–61%) |
| Chemical and physical data | |
| Formula | C14H9ClF3NO2 |
| Molar mass | 315.68 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Efavirenz (EFV), sold under the brand names Sustiva among others, is an antiretroviral medication used to treat and prevent HIV/AIDS.[1] It is generally recommended for use with other antiretrovirals.[1] It may be used for prevention after a needlestick injury or other potential exposure.[1] It is sold both by itself and in combination as efavirenz/emtricitabine/tenofovir.[1] It is taken by mouth once a day.[1]
Common side effects include rash, nausea, headache, feeling tired, and trouble sleeping.[1] Some of the rashes may be serious such as Stevens–Johnson syndrome.[1] Other serious side effects include depression, thoughts of suicide, liver problems, and seizures.[1] It is not safe for use during pregnancy.[1] It is a non-nucleoside reverse transcriptase inhibitor (NNRTI) and works by blocking the function of reverse transcriptase.[1]
Efavirenz was approved for medical use in the United States in 1998.[1] It is on the World Health Organization's List of Essential Medicines.[4] As of 2016, it is available as a generic medication.[5][6] The wholesale cost in the developing world is about US$3.27 to 9.15 per month.[7] As of 2015 the cost for a typical month of medication in the United States is more than US$200.[8]
Medical uses
For HIV infection that has not previously been treated, the United States Department of Health and Human Services Panel on Antiretroviral Guidelines recommends the use of efavirenz in combination with tenofovir/emtricitabine (Truvada) as one of the preferred NNRTI-based regimens in adults and adolescents[9] and children.[10]
Efavirenz is also used in combination with other antiretroviral agents as part of an expanded post-exposure prophylaxis regimen to reduce the risk of HIV infection in people exposed to a significant risk (e.g. needlestick injuries, certain types of unprotected sex, etc.).[11][12]
Pregnancy and breastfeeding
Efavirenz is safe to use during the first trimester of pregnancy.[13] Efavirenz passes into breast milk and breast-fed infants may be exposed to efavirenz.[14]
Dosage
The defined daily dose is 600 mg (by mouth).[3] The dose in those who weight more than 40 kg is 600 mg once per day, in those 25 to 40 kg it is 400 mg once per day, in those who 14 to 25 kg it is 300 mg once per day and in those 10 to 14 kg it is 200 mg once per day.[2]
Side effects
Neuropsychiatric effects are the most common adverse effects, and include disturbed sleep (including nightmares, insomnia, disrupted sleep, and daytime fatigue), dizziness, headaches, vertigo, blurred vision, anxiety, and cognitive impairment (including fatigue, confusion, and memory and concentration problems), and depression, including suicidal thinking.[15][16] Some people experience euphoria.[15]Rash and nausea may occur.[17]Use of efavirenz can produce a false positive result in some urine tests for marijuana.[18]Efavirenz may lengthen the QT interval so should not be used in people with or at risk of torsades de pointes.[19]Efavirenz may cause convulsions in adult and pediatric populations who have a history of seizures.[17]
Contraindications
People who have taken this medication before and experienced an allergic reaction should avoid taking further efavirenz dosages. Hypersensitivity reactions include Steven-Johnson syndrome, toxic skin eruptions, and erythema multiforme.[17]
Interactions
Efavirenz is broken down in the liver by enzymes that belong to the cytochrome P450 system, which include both CYP2B6 and CYP3A4.[17] Efavirenz is a substrate of these enzymes and can decrease the metabolism of other drugs that require the same enzymes.[17] However, efavirenz also induces these enzymes, which means the enzyme activity is enhanced and the metabolism of other drugs broken down by CYP2B6 and CYP3A4 can be increased.[17]
One group of drugs that efavirenz affects is protease inhibitors, which are used for HIV/AIDS. Efavirenz will lower the blood levels of most protease inhibitors, including aprenavir, atazanavir, and indinavir.[17] At lowered levels, protease inhibitors may not be effective in people taking both drugs, which means the virus that causes HIV/AIDS won't be stopped from replicating.[20]
Efavirenz also affects antifungal drugs, which are used for fungal infections such as urinary tract infections. Similar to the effect seen with protease inhibitors, efavirenz lowers the blood levels of antifungal drugs like voriconazole, itraconazole, ketoconazole, and posaconazole.[17] As a result of lowered levels, antifungal drugs may not be effective in people taking both drugs.[20]
Mechanism of action
Anti-HIV effects
Efavirenz falls in the NNRTI class of antiretrovirals.[20] Both nucleoside and non-nucleoside RTIs inhibit the same target, the reverse transcriptase enzyme, an essential viral enzyme which transcribes viral RNA into DNA. Unlike nucleoside RTIs, which bind at the enzyme's active site, NNRTIs act allosterically by binding to a distinct site away from the active site known as the NNRTI pocket.[21][22]
Efavirenz is not effective against HIV-2, as the pocket of the HIV-2 reverse transcriptase has a different structure, which confers intrinsic resistance to the NNRTI class.[23]As most NNRTIs bind within the same pocket, viral strains which are resistant to efavirenz are usually also resistant to the other NNRTIs, nevirapine and delavirdine. The most common mutation observed after efavirenz treatment is K103N, which is also observed with other NNRTIs.[17] Nucleoside reverse-transcriptase inhibitors (NRTIs) and efavirenz have different binding targets, so cross-resistance is unlikely; the same is true with regard to efavirenz and protease inhibitors.[1]
Neuropsychiatric effects
As of 2016 the mechanism of efavirenz' neuropsychiatric adverse effects was not clear.[15][16] Efavirenz appears to have neurotoxicity, possibly by interfering with mitochondrial function, which may in turn possibly be caused by inhibiting creatine kinase but also possibly by disrupting mitochondrial membranes or by interfering with nitric oxide signalling.[15] Some neuropsychiatric adverse effects may be mediated through cannabinoid receptors, or through activity at the 5-HT2A receptor, but efavirenz interacts with many CNS receptors, so this is not clear.[15] The neuropsychiatric adverse effects are dose-dependent.[15]
Chemistry
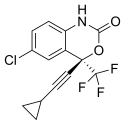
Efavirenz is chemically described as (S)-6-chloro-(cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-2H-3,1-benzoxazin-2-one. Its empirical formula is C14H9ClF3NO2. Efavirenz is a white to slightly pink crystalline powder with a molecular mass of 315.68 g/mol.[22]
History
Efavirenz was approved by the FDA on September 21, 1998[24]On February 17, 2016, the FDA approved the generic tablet formulation to be produced by Mylan.[5][6]
In late-2018, Thailand's Government Pharmaceutical Organization (GPO) announced that it will produce efavirenz after receiving WHO approval.[25] Efavirenz code name is DMP 266, discovered by Du pont Pharma. European countries are set to receive the license for manufacturing of Efavirenz in May 1999.[26]
Society and culture
Pricing information
A one-month supply of 600 mg tablets costs approximately US$1,010 in July 2016.[27] In 2007, Merck provided Efavirenz in certain developing countries and countries largely affected by HIV for about US$0.65 per day.[28] Some emerging countries have opted to purchase Indian generics.[29]
In Thailand, a one-month supply of efavirenz + Truvada, as of June 2012, cost 2,900 baht (US$90), and there is a social program for patients who cannot afford the medication. As of 2018[update] Thailand will produce efavirenz domestically. Its Government Pharmaceutical Organization product costs 180 baht per bottle of thirty 600 mg tablets. The imported version in Thailand retails for more than 1,000 baht per bottle. GPO will devote 2.5 percent of its manufacturing capacity to make 42 million efavirenz pills in 2018, allowing it to serve export markets as well as domestic. The Philippines alone will order about 300,000 bottles of efavirenz for 51 million baht.[25]
In South Africa, a license has been granted to generics giant Aspen Pharmacare to manufacture, and distribute to Sub-Saharan Africa, a cost-effective antiretroviral drug.[30]
Recreational use
Abuse of efavirenz by crushing and smoking the tablets for supposed hallucinogenic and dissociative effects has been reported in South Africa, where it is used in a mixture known as whoonga and nyaope.[31][32][33]
Brands
As of 2016, efavirenz was marketed in various jurisdictions under the brand names Adiva, Avifanz, Efamat, Efatec, Efavir, Efavirenz, Efcure, Eferven, Efrin, Erige, Estiva, Evirenz, Filginase, Stocrin, Sulfina V, Sustiva, Virorrever, and Zuletel.[34]As of 2016, the combination of efavirenz, tenofovir, and emtricitabine was marketed in various jurisdictions under the brand names Atripla, Atroiza, Citenvir, Oditec, Teevir, Trustiva, Viraday, and Vonavir.[34]As of 2016, the combination of efavirenz, tenofovir, and lamivudine was marketed under the brand name Eflaten.[34]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 "Efavirenz". The American Society of Health-System Pharmacists. Archived from the original on 17 November 2016. Retrieved 28 November 2016.
- ↑ 2.0 2.1 "EFAVIRENZ = EFV = EFZ oral - Essential drugs". medicalguidelines.msf.org. Archived from the original on 2 March 2021. Retrieved 30 August 2020.
- ↑ 3.0 3.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 30 July 2020. Retrieved 21 September 2020.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 5.0 5.1 "Efavirenz Drug Profile". DrugPatentWatch. Archived from the original on 10 November 2016. Retrieved 9 November 2016.
- ↑ 6.0 6.1 "Efavirenz: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 31 July 2020. Retrieved 25 April 2020.
- ↑ "Efavirenz". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 28 November 2016.
- ↑ Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 62. ISBN 9781284057560.
- ↑ "Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents". 14 July 2016. Archived from the original on 23 May 2013.
- ↑ "Guidelines for the Use of Antiretroviral Agents in Pediatric HIV Infection". NIH AIDSinfo. 1 March 2016. Archived from the original on 15 November 2016.
- ↑ Kuhar DT, Henderson DK, Struble KA, Heneine W, Thomas V, Cheever LW, et al. (September 2013). "Updated US Public Health Service guidelines for the management of occupational exposures to human immunodeficiency virus and recommendations for postexposure prophylaxis". Infection Control and Hospital Epidemiology. 34 (9): 875–92. doi:10.1086/672271. PMID 23917901. Archived from the original on 23 June 2019. Retrieved 30 June 2019.
- ↑ "Antiretroviral Postexposure Prophylaxis After Sexual, Injection-Drug Use, or Other Nonoccupational Exposure to HIV in the United States =" (PDF). Centers for Disease Control and Prevention. Archived (PDF) from the original on 27 January 2017. Retrieved 7 November 2016.
- ↑ Ford N, Mofenson L, Shubber Z, Calmy A, Andrieux-Meyer I, Vitoria M, et al. (March 2014). "Safety of efavirenz in the first trimester of pregnancy: an updated systematic review and meta-analysis". AIDS. 28 Suppl 2: S123-31. doi:10.1097/qad.0000000000000231. PMID 24849471.
- ↑ Waitt CJ, Garner P, Bonnett LJ, Khoo SH, Else LJ (July 2015). "Is infant exposure to antiretroviral drugs during breastfeeding quantitatively important? A systematic review and meta-analysis of pharmacokinetic studies". The Journal of Antimicrobial Chemotherapy. 70 (7): 1928–41. doi:10.1093/jac/dkv080. PMC 4472329. PMID 25858354.
- ↑ 15.0 15.1 15.2 15.3 15.4 15.5 Treisman GJ, Soudry O (October 2016). "Neuropsychiatric Effects of HIV Antiviral Medications". Drug Safety. 39 (10): 945–57. doi:10.1007/s40264-016-0440-y. PMID 27534750.
- ↑ 16.0 16.1 Apostolova N, Funes HA, Blas-Garcia A, Galindo MJ, Alvarez A, Esplugues JV (October 2015). "Efavirenz and the CNS: what we already know and questions that need to be answered". The Journal of Antimicrobial Chemotherapy. 70 (10): 2693–708. doi:10.1093/jac/dkv183. PMID 26203180.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 17.6 17.7 17.8 "Efavirenz label" (PDF). FDA. October 2016. Archived (PDF) from the original on 10 November 2016.
- ↑ Dasgupta, Amitava (2019). Biotin and other interferences in immunoassays : a concise guide. St. Louis, Missouri. p. 125. ISBN 978-0-12-816741-0. Archived from the original on 28 August 2021. Retrieved 21 December 2020.
- ↑ El-Sherif, Nabil; Turitto, Gioia; Boutjdir, Mohamed (2019). "Acquired Long QT Syndrome and Electrophysiology of Torsade de Pointes". Arrhythmia & Electrophysiology Review. 8 (2): 122–130. doi:10.15420/aer.2019.8.3. ISSN 2050-3369. Archived from the original on 28 August 2021. Retrieved 18 December 2020.
- ↑ 20.0 20.1 20.2 "Efavirenz Monograph for Professionals". Drugs.com. Archived from the original on 17 November 2016. Retrieved 26 December 2020.
- ↑ Rakhmanina, Natella Y.; van den Anker, John N. (2010). "Efavirenz in the Therapy of HIV Infection". Expert opinion on drug metabolism & toxicology. 6 (1): 95–103. doi:10.1517/17425250903483207. ISSN 1742-5255. Archived from the original on 28 August 2021. Retrieved 23 December 2020.
- ↑ 22.0 22.1 "Efavirenz". pubchem.ncbi.nlm.nih.gov. Archived from the original on 11 November 2020. Retrieved 21 December 2020.
- ↑ Ren J, Bird LE, Chamberlain PP, Stewart-Jones GB, Stuart DI, Stammers DK (October 2002). "Structure of HIV-2 reverse transcriptase at 2.35-A resolution and the mechanism of resistance to non-nucleoside inhibitors". Proceedings of the National Academy of Sciences of the United States of America. 99 (22): 14410–5. doi:10.1073/pnas.222366699. PMC 137897. PMID 12386343.
- ↑ "Drug Approval Package: Sustiva (efavirenz) NDA# 20-972". U.S. Food and Drug Administration (FDA). 15 December 2011. Archived from the original on 30 July 2020. Retrieved 25 April 2020.
- ↑ 25.0 25.1 Wipatayotin, Apinya (3 November 2018). "Thailand gets nod to make HIV/Aids drug". Bangkok Post. Archived from the original on 28 August 2021. Retrieved 3 November 2018.
- ↑ "Efavirenz (Sustiva)". aidsmap. June 2017. Archived from the original on 27 July 2019. Retrieved 27 July 2019.
- ↑ "Cost Considerations and Antiretroviral Therapy | Adult and Adolescent ARV Guidelines | AIDSinfo". AIDSinfo. Archived from the original on 17 November 2016. Retrieved 16 November 2016.
- ↑ "Merck & Co., Inc., Again Reduces Price of Stocrin (Efavirenz) for Patients in Least Developed Countries and Countries Hardest Hit by Epidemic". Drugs.com MedNews. Archived from the original on 14 July 2014.
- ↑ IndiaDaily - A new trend in emerging nations - Brazil opts for Indian generic drug ignoring US pharmaceutical giant Merck's patent on AIDS drug Efavirenz Archived 2008-02-19 at the Wayback Machine
- ↑ Osewe, Patrick Lumumba; Korkoi Nkrumah, Yvonne; Sackey, Emmanuel K. (15 June 2008). Improving Access to HIV/AIDS Medicines in Africa: Trade-Related Aspects of Intellectual Property Rights (TRIPS) Flexibilities Utilization. World Bank Publications. pp. 35–39. ISBN 978-0-8213-7544-0. Archived from the original on 16 June 2013. Retrieved 30 June 2012.
- ↑ IOL: Thugs get high on stolen Aids drugs Archived 2013-08-06 at the Wayback Machine IOL News May 12, 2007
- ↑ Getting high on HIV drugs in S Africa. BBC News, 8 December 2008. Archived 9 December 2008 at the Wayback Machine
- ↑ 'No Turning Back': Teens Abuse HIV Drugs. ABC News, April 6, 2009. Archived April 8, 2009, at the Wayback Machine
- ↑ 34.0 34.1 34.2 "International brands for Efavirenz". Drugs.com. Archived from the original on 10 November 2016. Retrieved 10 November 2016.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Pages with non-numeric formatnum arguments
- CS1: long volume value
- Webarchive template wayback links
- Use dmy dates from April 2020
- Articles with invalid date parameter in template
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to watched fields
- Articles containing potentially dated statements from 2018
- All articles containing potentially dated statements
- 5-HT2A agonists
- Alkyne derivatives
- Benzoxazines
- Bristol-Myers Squibb
- Carbamates
- Cyclopropanes
- Estrogens
- GABAA receptor positive allosteric modulators
- Hepatotoxins
- Serotonin-dopamine reuptake inhibitors
- Non-nucleoside reverse transcriptase inhibitors
- Chloroarenes
- Trifluoromethyl compounds
- Psychedelic drugs
- VMAT inhibitors
- World Health Organization essential medicines
- RTT