Tegaserod
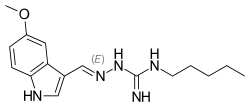 | |
 | |
| Names | |
|---|---|
| Trade names | Zelnorm, Zelmac |
| |
| Clinical data | |
| Main uses | Irritable bowel syndrome with constipation[1] |
| Side effects | Headache, abdominal pain, diarrhea, heart attacks, stroke, suicide[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Typical dose | 6 mg BID[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 10% |
| Protein binding | 98% |
| Metabolism | Stomach and liver |
| Elimination half-life | 11 ± 5 hours |
| Excretion | Fecal and Kidney |
| Chemical and physical data | |
| Formula | C16H23N5O |
| Molar mass | 301.394 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Tegaserod, sold under the brand name Zelnorm among others, is a medication used to treat irritable bowel syndrome with constipation.[1] It is taken by mouth.[1]
Common side effects include headache, abdominal pain, diarrhea, and dizziness.[1] Other side effects may include heart attacks, stroke, and suicide.[1] It is a 5-HT4 activator which stimulates both contraction and increased fluid release by the intestines.[1]
Tegaserod was approved for medical use in the United States in 2002.[1] It was removed from the market in 2007 due to concerns related to the heart but than re-allowed in 2019.[1] In Europe it was refused approval in both 2005 and 2006.[2] In the United States it costs about 360 USD per month as of 2021.[3] It was withdrawn from the market due to commercial reasons in 2022.[4]
Medical uses
Dosage
It is generally taken as 6 mg twice per day.[1]
Mechanism of action
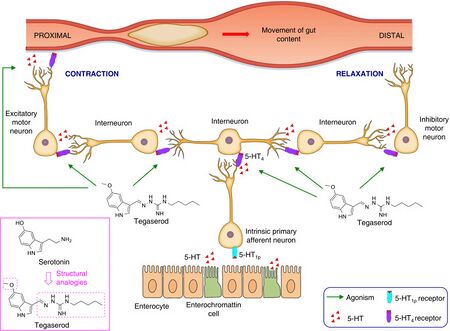
The drug functions as a motility stimulant, achieving its desired therapeutic effects through activation of the 5-HT4 receptors of the enteric nervous system in the gastrointestinal tract. It also stimulates gastrointestinal motility and the peristaltic reflex, and allegedly reduces abdominal pain.[6] Additionally, tegaserod is a 5-HT2B receptor antagonist.[7]
History
On 30 March 2007, the United States Food and Drug Administration requested that Novartis withdraw tegaserod from shelves.[8] The FDA alleged a relationship between prescriptions of the drug and increased risks of heart attack or stroke. An analysis of data collected on over 18,000 patients demonstrated adverse cardiovascular events in 13 of 11,614 patients treated with tegaserod (a rate of 0.11%) as compared with 1 of 7,031 patients treated with placebo (a rate of 0.01%). Novartis alleged all of the affected patients had preexisting cardiovascular disease or risk factors for such, and further alleged that no causal relationship between tegaserod use and cardiovascular events has been demonstrated.[9] On the same day as the FDA announcement, Novartis Pharmaceuticals Canada announced that it was suspending marketing and sales of the drug in Canada in response to a request from Health Canada.[10] In a large cohort study based on a US health insurance database, no increase in the risk of cardiovascular events were found under tegaserod treatment.[11] In 2019, tegaserod was reintroduced as for use in irritable bowel syndrome with constipation (IBS-C) in women under 65.[12]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Tegaserod Monograph for Professionals". Drugs.com. Archived from the original on 28 January 2021. Retrieved 24 September 2021.
- ↑ "Zelnorm". Archived from the original on 6 October 2018. Retrieved 24 September 2021.
- ↑ "Zelnorm Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 24 September 2021.
- ↑ "Pharmacist's Letter". pharmacist.therapeuticresearch.com. Archived from the original on 8 August 2022. Retrieved 12 January 2023.
- ↑ Madia, Valentina Noemi; Messore, Antonella; Saccoliti, Francesco; Tudino, Valeria; De Leo, Alessandro; De Vita, Daniela; Bortolami, Martina; Scipione, Luigi; Pindinello, Ivano; Costi, Roberta; Di Santo, Roberto (15 October 2020). "Tegaserod for the Treatment of Irritable Bowel Syndrome". Anti-Inflammatory & Anti-Allergy Agents in Medicinal Chemistry. 19 (4): 342–369. doi:10.2174/1871523018666190911121306.
- ↑ Rossi, S. (2004). Australian Medicines Handbook. Adelaide: Health Communication Network. ISBN 0-9578521-4-2.
- ↑ Beattie DT, Smith JA, Marquess D, et al. (November 2004). "The 5-HT4 receptor agonist, tegaserod, is a potent 5-HT2B receptor antagonist in vitro and in vivo". Br. J. Pharmacol. 143 (5): 549–60. doi:10.1038/sj.bjp.0705929. PMC 1575425. PMID 15466450.
- ↑ "FDA Announces Discontinued Marketing of GI Drug, Zelnorm, for Safety Reasons". FDA Press Release. 30 March 2007. Archived from the original on 18 January 2017. Retrieved 5 January 2021.
- ↑ "Zelnorm" (PDF). Novartis. Archived from the original (PDF) on 2007-04-10. Retrieved 2007-03-30.
- ↑ "Novartis suspends Canadian marketing and sales of Zelnorm in response to request from Health Canada". Archived from the original on 2007-06-05. Retrieved 2007-03-30.
- ↑ Loughlin J, Quinn S, Rivero E, Wong J, Huang J, Kralstein J, Earnest DL, Seeger JD (2010). "Tegaserod and the Risk of Cardiovascular Ischemic Events: An Observational Cohort Study". J Cardiovasc Pharmacol Ther. 15 (2): 151–7. doi:10.1177/1074248409360357. PMID 20200325. S2CID 37964792.
- ↑ WorldMeds, U. S. "FDA approves the reintroduction of Zelnorm™ (tegaserod) for Irritable Bowel Syndrome with Constipation (IBS-C) in women under 65". www.prnewswire.com. Archived from the original on 2019-05-09. Retrieved 2019-09-17.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with changed CASNo identifier
- Motility stimulants
- Serotonin receptor agonists
- Withdrawn drugs
- Indoles
- Phenol ethers
- Guanidines
- RTT