Amisulpride
 | |
 | |
| Names | |
|---|---|
| Trade names | Solian, Barhemsys, Socian, Deniban, others |
| Other names | APD421 |
| |
| Clinical data | |
| Drug class | Atypical antipsychotics[1] |
| Main uses | Postoperative nausea and vomiting, schizophrenia[1][2] |
| Side effects | Nausea, breast pain, sexual dysfunction, low potassium[1][2] |
| Pregnancy category | |
| Routes of use | By mouth, intravenous |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 48%[6][7] |
| Protein binding | 16%[7] |
| Metabolism | Liver (minimal; most excreted unchanged)[7] |
| Elimination half-life | 12 hours[6] |
| Excretion | Kidney[6] (23–46%),[8][9] Faecal[7] |
| Chemical and physical data | |
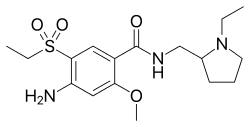
| Formula | C17H27N3O4S |
| Molar mass | 369.48 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Amisulpride, sold under the brand name Solian among others, is a medication used to treat and prevent postoperative nausea and vomiting (PONV) and to treat schizophrenia.[1][2] It is taken by mouth or by injection into a vein.[1][2]
Common side effects include nausea, breast pain, sexual dysfunction, and low potassium.[1][2] Other side effects may include QT prolongation, high blood sugar, and abnormal lipids.[1][2] While its safety in pregnancy is unclear, it has caused harm with use in other animals.[10] It is an atypical antipsychotics and is believed to work by blocking the dopamine D2 and D3 receptors.[1]
Amisulpride has been in medical use since 1992.[11] It is available as a generic medication.[1] In the United Kingdom 60 pills of 400 mg costs about £42 as of 2021.[1] In the United States a vial of 5 mg costs about 45 USD.[12]
Medical uses
Schizophrenia
Although according to other studies it appears to have comparable efficacy to olanzapine in the treatment of schizophrenia.[13] Amisulpride augmentation, similarly to sulpiride augmentation, has been considered a viable treatment option (although this is based on low-quality evidence) in clozapine-resistant cases of schizophrenia.[14][15] Another recent study concluded that amisulpride is an appropriate first-line treatment for the management of acute psychosis.[16]
Postoperative nausea
Amisulpride is indicated for use in the United States in adults for the prevention of postoperative nausea and vomiting (PONV), either alone or in combination with an antiemetic of a different class; and to treat PONV in those who have received antiemetic prophylaxis with an agent of a different class or have not received prophylaxis.[17]
Contraindications
Amisulpride's use is contraindicated in the following disease states[7][18][19]
- Pheochromocytoma
- Concomitant prolactin-dependent tumours e.g. prolactinoma, breast cancer
- Movement disorders (e.g. Parkinson's disease and dementia with Lewy bodies)
- Lactation
- Children before the onset of puberty
Neither is it recommended to use amisulpride in patients with hypersensitivities to amisulpride or the excipients found in its dosage form.[7]
Side effects
- Very Common (≥10% incidence)[5]
- Extrapyramidal side effects (EPS; including dystonia, tremor, akathisia, parkinsonism).
- Insomnia
- Hypersalivation
- Nausea
- Headache
- Hyperactivity
- Vomiting
- Hyperprolactinaemia (which can lead to galactorrhoea, breast enlargement and tenderness, sexual dysfunction, etc.)
- Weight gain (produces less weight gain than chlorpromazine, clozapine, iloperidone, olanzapine, paliperidone, quetiapine, risperidone, sertindole, zotepine and more (although not statistically significantly) weight gain than haloperidol, lurasidone, ziprasidone and approximately as much weight gain as aripiprazole and asenapine)[21]
- Anticholinergic side effects (although it does not bind to the muscarinic acetylcholine receptors and hence these side effects are usually quite mild) such as
- - constipation
- - dry mouth
- - disorder of accommodation
- - Blurred vision
- Rare (<1% incidence)[5][7][20][18][19]
- Blood dyscrasias such as leucopenia, neutropenia and agranulocytosis
- QT interval prolongation (in a recent meta-analysis of the safety and efficacy of 15 antipsychotic drugs amisulpride was found to have the 2nd highest effect size for causing QT interval prolongation[21])
Hyperprolactinaemia results from antagonism of the D2 receptors located on the lactotrophic cells found in the anterior pituitary gland. Amisulpride has a high propensity for elevating plasma prolactin levels as a result of its poor blood-brain barrier penetrability and hence the resulting greater ratio of peripheral D2 occupancy to central D2 occupancy. This means that to achieve the sufficient occupancy (~60–80%[22]) of the central D2 receptors in order to elicit its therapeutic effects a dose must be given that is enough to saturate peripheral D2 receptors including those in the anterior pituitary.[23][24]
Discontinuation
The British National Formulary recommends a gradual withdrawal when discontinuing antipsychotics to avoid acute withdrawal syndrome or rapid relapse.[25] Symptoms of withdrawal commonly include nausea, vomiting, and loss of appetite.[26] Other symptoms may include restlessness, increased sweating, and trouble sleeping.[26] Less commonly there may be a feeling of the world spinning, numbness, or muscle pains.[26] Symptoms generally resolve after a short period of time.[26]
There is tentative evidence that discontinuation of antipsychotics can result in psychosis.[27] It may also result in reoccurrence of the condition that is being treated.[28] Rarely tardive dyskinesia can occur when the medication is stopped.[26]
Overdose
Torsades de pointes is common in overdose.[29][30] Amisulpride is moderately dangerous in overdose (with the TCAs being very dangerous and the SSRIs being modestly dangerous).[31][32]
Interactions
Amisulpride should not be used in conjunction with drugs that prolong the QT interval (such as citalopram, bupropion, clozapine, tricyclic antidepressants, sertindole, ziprasidone, etc.),[31] reduce heart rate and those that can induce hypokalaemia. Likewise it is imprudent to combine antipsychotics due to the additive risk for tardive dyskinesia and neuroleptic malignant syndrome.[31]
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | >10,000 | Human | [34] |
| NET | >10,000 | Human | [34] |
| DAT | >10,000 | Human | [34] |
| 5-HT1A | >10,000 | Human | [34] |
| 5-HT1B | 1,744 | Human | [34] |
| 5-HT1D | 1,341 | Human | [34] |
| 5-HT1E | >10,000 | Human | [34] |
| 5-HT2A | 8,304 | Human | [34] |
| 5-HT2B | 13 | Human | [34] |
| 5-HT2C | >10,000 | Human | [34] |
| 5-HT3 | >10,000 | Human | [34] |
| 5-HT5A | >10,000 | Human | [34] |
| 5-HT6 | 4,154 | Human | [34] |
| 5-HT7 | 11.5 | Human | [34] |
| α1A | >10,000 | Human | [34] |
| α1B | >10,000 | Human | [34] |
| α1D | >10,000 | Human | [34] |
| α2A | 1,114 | Human | [34] |
| α2C | 1,540 | Human | [34] |
| β1 | >10,000 | Human | [34] |
| β2 | >10,000 | Human | [34] |
| β3 | >10,000 | Human | [34] |
| D1 | >10,000 | Human | [34] |
| D2 | 3.0 | Human | [34] |
| D3 | 3.5 | Rat | [34] |
| D4 | 2,369 | Human | [34] |
| D5 | >10,000 | Human | [34] |
| H1 | >10,000 | Human | [34] |
| H2 | >10,000 | Human | [34] |
| H4 | >10,000 | Human | [34] |
| M1 | >10,000 | Human | [34] |
| M2 | >10,000 | Human | [34] |
| M3 | >10,000 | Human | [34] |
| M4 | >10,000 | Human | [34] |
| M5 | >10,000 | Human | [34] |
| σ1 | >10,000 | Rat | [34] |
| σ2 | >10,000 | Rat | [34] |
| MOR | >10,000 | Human | [34] |
| DOR | >10,000 | Human | [34] |
| KOR | >10,000 | Human | [34] |
| GHBHigh | 50 (IC50) | Rat | [35] |
| NMDA (PCP) |
>10,000 | Rat | [36] |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | |||
Amisulpride functions primarily as a dopamine D2 and D3 receptor antagonist. It has high affinity for these receptors with dissociation constants of 3.0 and 3.5 nM, respectively.[34] Although standard doses used to treat psychosis inhibit dopaminergic neurotransmission, low doses preferentially block inhibitory presynaptic autoreceptors. This results in a facilitation of dopamine activity, and for this reason, low-dose amisulpride has also been used to treat dysthymia.[7]
Amisulpride and its relatives sulpiride, levosulpiride, and sultopride have been shown to bind to the high-affinity GHB receptor at concentrations that are therapeutically relevant (IC50 = 50 nM for amisulpride).[35]
Amisulpride, sultopride and sulpiride respectively present decreasing in vitro affinities for the D2 receptor (IC50 = 27, 120 and 181 nM) and the D3 receptor (IC50 = 3.6, 4.8 and 17.5 nM).[37]
Though it was long widely assumed that dopaminergic modulation is solely responsible for the respective antidepressant and antipsychotic properties of amisulpride, it was subsequently found that the drug also acts as a potent antagonist of the serotonin 5-HT7 receptor (Ki = 11.5 nM).[34] Several of the other atypical antipsychotics such as risperidone and ziprasidone are potent antagonists at the 5-HT7 receptor as well, and selective antagonists of the receptor show antidepressant properties themselves. To characterize the role of the 5-HT7 receptor in the antidepressant effects of amisulpride, a study prepared 5-HT7 receptor knockout mice.[34] The study found that in two widely used rodent models of depression, the tail suspension test, and the forced swim test, those mice did not exhibit an antidepressant response upon treatment with amisulpride.[34] These results suggest that 5-HT7 receptor antagonism mediates the antidepressant effects of amisulpride.[34]
Amisulpride also appears to bind with high affinity to the serotonin 5-HT2B receptor (Ki = 13 nM), where it acts as an antagonist.[34] The clinical implications of this, if any, are unclear.[34] In any case, there is no evidence that this action mediates any of the therapeutic effects of amisulpride.[34]
Amisulpride shows stereoselectivity in its actions.[38] Aramisulpride ((R)-amisulpride) has higher affinity for the 5-HT7 receptor (Ki = 47 nM vs. 1,900 nM) while esamisulpride ((S)-amisulpride) has higher affinity for the D2 receptor (4.0 nM vs. 140 nM).[38][39] An 85:15 ratio of aramisulpride to esamisulpride (SEP-4199) which provides more balanced 5-HT7 and D2 receptor antagonism than racemic amisulpride (50:50 ratio of enantiomers) is under development for the treatment of bipolar depression.[40]
Society and culture
Brand names
Brand names include: Amazeo, Amipride (AU), Amival, Solian (AU, IE, RU, UK, ZA), Soltus, Sulpitac (IN), Sulprix (AU), Midora (RO) and Socian (BR).[41][42]
Availability
Amisulpride was not approved by the Food and Drug Administration for use in the United States until February 2020, but it is used in Europe,[42] Israel, Mexico, India, New Zealand and Australia[7] to treat psychosis and schizophrenia.[43][44]
An IV formulation of Amisulpride was approved for the treatment of postoperative nausea and vomiting ("PONV") in the United States in February 2020.[45][17][46]
History
The U.S. Food and Drug Administration (FDA) approved amisulpride based on evidence from four clinical trials of 2323 subjects undergoing surgery or experiencing nausea and vomiting after the surgery.[46] The trials were conducted at 80 sites in the United States, Canada and Europe.[46]
Two trials (Trials 1 and 2) enrolled subjects scheduled to have surgery.[46] Subjects were randomly assigned to receive either amisulpride or a placebo drug at the beginning of general anesthesia.[46] In Trial 1, subjects received amisulpride or placebo alone, and in Trial 2, they received amisulpride or placebo in combination with one medication approved for prevention of nausea and vomiting.[46] Neither the subjects nor the health care providers knew which treatment was being given until after the trial was complete.[46]
The trials counted the number of subjects who had no vomiting and did not use additional medications for nausea or vomiting in the first day (24 hours) after the surgery.[46] The results then compared amisulpride to placebo.[46]
The other two trials (Trials 3 and 4) enrolled subjects who were experiencing nausea and vomiting after surgery.[46] In Trial 3, subjects did not receive any medication to prevent nausea and vomiting before surgery and in Trial 4 they received the medication, but the treatment did not work.[46] In both trials, subjects were randomly assigned to receive either amisulpride or placebo.[46] Neither the subjects nor the health care providers knew which treatment was being given until after the trial was complete.[46]
The trials counted the number of subjects who had no vomiting and did not use additional medications for nausea or vomiting in the first day (24 hours) after the treatment.[46] The trial compared amisulpride to placebo.[46]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 415. ISBN 978-0857114105.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 "Amisulpride Monograph for Professionals". Drugs.com. Retrieved 14 January 2022.
- ↑ "Australian Product Information – Solian (Amisulpride) Tablets And Solution". TGA eBS. Archived from the original on 31 December 2019. Retrieved 10 May 2020.
- ↑ 4.0 4.1 "Amisulpride (Barhemsys) Use During Pregnancy". Drugs.com. 2 September 2020. Archived from the original on 28 January 2021. Retrieved 24 September 2020.
- ↑ 5.0 5.1 5.2 5.3 "Amisulpride 100 mg Tablets - Summary of Product Characteristics (SmPC)". (emc). 5 July 2019. Archived from the original on 26 February 2020. Retrieved 26 February 2020.
- ↑ 6.0 6.1 6.2 Rosenzweig, P.; Canal, M.; Patat, A.; Bergougnan, L.; Zieleniuk, I.; Bianchetti, G. (2002). "A review of the pharmacokinetics, tolerability and pharmacodynamics of amisulpride in healthy volunteers". Human Psychopharmacology. 17 (1): 1–13. doi:10.1002/hup.320. PMID 12404702. S2CID 23877366.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 7.9 "Solian tablets and solution product information" (PDF). TGA eBusiness Services. Sanofi-Aventis Australia Pty Ltd. 27 September 2019. Archived from the original on 31 December 2019. Retrieved 26 February 2020.
- ↑ Caccia, S (May 2000). "Biotransformation of Post-Clozapine Antipsychotics Pharmacological Implications". Clinical Pharmacokinetics. 38 (5): 393–414. doi:10.2165/00003088-200038050-00002. PMID 10843459. S2CID 68853079.
- ↑ Noble, S; Benfield, P (December 1999). "Amisulpride: A Review of its Clinical Potential in Dysthymia". CNS Drugs. 12 (6): 471–483. doi:10.2165/00023210-199912060-00005.
- ↑ "Amisulpride (Barhemsys) Use During Pregnancy". Drugs.com. Archived from the original on 28 January 2021. Retrieved 14 January 2022.
- ↑ Advances in the Neurochemistry and Neuropharmacology of Tourette Syndrome. Academic Press. 27 November 2013. p. 318. ISBN 978-0-12-411561-3. Archived from the original on 14 January 2022. Retrieved 14 January 2022.
- ↑ "Barhemsys Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 14 January 2022.
- ↑ Komossa, K; Rummel-Kluge, C; Hunger, H; Schmid, F; Schwarz, S; Silveira da Mota Neto, JI; Kissling, W; Leucht, S (January 2010). "Amisulpride versus other atypical antipsychotics for schizophrenia". The Cochrane Database of Systematic Reviews (1): CD006624. doi:10.1002/14651858.CD006624.pub2. PMC 4164462. PMID 20091599.
- ↑ Solanki, RK; Sing, P; Munshi, D (October–December 2009). "Current perspectives in the treatment of resistant schizophrenia". Indian Journal of Psychiatry. 51 (4): 254–60. doi:10.4103/0019-5545.58289. PMC 2802371. PMID 20048449.
- ↑ Mouaffak, F; Tranulis, C; Gourevitch, R; Poirier, MF; Douki, S; Olié, JP; Lôo, H; Gourion, D (2006). "Augmentation Strategies of Clozapine With Antipsychotics in the Treatment of Ultraresistant Schizophrenia". Clinical Neuropharmacology. 29 (1): 28–33. doi:10.1097/00002826-200601000-00009. PMID 16518132. S2CID 29682562.
- ↑ Nuss, P.; Hummer, M.; Tessier, C. (2007). "The use of amisulpride in the treatment of acute psychosis". Therapeutics and Clinical Risk Management. 3 (1): 3–11. doi:10.2147/tcrm.2007.3.1.3. PMC 1936283. PMID 18360610.
- ↑ 17.0 17.1 "Barhemsys (amisulpride) injection, for intravenous use" (PDF). U.S. Food and Drug Administration (FDA). February 2020. Archived (PDF) from the original on 26 February 2020. Retrieved 26 February 2020.
- ↑ 18.0 18.1 18.2 Joint Formulary Committee (2013). British National Formulary (BNF) (65 ed.). London, UK: Pharmaceutical Press. ISBN 978-0-85711-084-8.
- ↑ 19.0 19.1 19.2 Rossi, S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- ↑ 20.0 20.1 Truven Health Analytics, Inc. DRUGDEX System (Internet) [cited 2013 Sep 19]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- ↑ 21.0 21.1 Leucht, S; Cipriani, A; Spineli, L; Mavridis, D; Orey, D; Richter, F; Samara, M; Barbui, C; Engel, RR; Geddes, JR; Kissling, W; Stapf, MP; Lässig, B; Salanti, G; Davis, JM (September 2013). "Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis". Lancet. 382 (9896): 951–962. doi:10.1016/S0140-6736(13)60733-3. PMID 23810019. S2CID 32085212.
- ↑ Brunton, L; Chabner, B; Knollman, B (2010). Goodman and Gilman's The Pharmacological Basis of Therapeutics (12th ed.). New York: McGraw-Hill Professional. ISBN 978-0-07-162442-8.
- ↑ McKeage, K; Plosker, GL (2004). "Amisulpride: a review of its use in the management of schizophrenia". CNS Drugs. 18 (13): 933–956. doi:10.2165/00023210-200418130-00007. ISSN 1172-7047. PMID 15521794.
- ↑ Natesan, S; Reckless, GE; Barlow, KB; Nobrega, JN; Kapur, S (October 2008). "Amisulpride the 'atypical' atypical antipsychotic — Comparison to haloperidol, risperidone and clozapine". Schizophrenia Research. 105 (1–3): 224–235. doi:10.1016/j.schres.2008.07.005. PMID 18710798. S2CID 11315672.
- ↑ Joint Formulary Committee, BMJ, ed. (March 2009). "4.2.1". British National Formulary (57 ed.). United Kingdom: Royal Pharmaceutical Society of Great Britain. p. 192. ISBN 978-0-85369-845-6.
Withdrawal of antipsychotic drugs after long-term therapy should always be gradual and closely monitored to avoid the risk of acute withdrawal syndromes or rapid relapse.
- ↑ 26.0 26.1 26.2 26.3 26.4 Haddad, Peter; Haddad, Peter M.; Dursun, Serdar; Deakin, Bill (2004). Adverse Syndromes and Psychiatric Drugs: A Clinical Guide. OUP Oxford. pp. 207–216. ISBN 9780198527480. Archived from the original on 14 April 2021. Retrieved 21 October 2021.
- ↑ Moncrieff J (July 2006). "Does antipsychotic withdrawal provoke psychosis? Review of the literature on rapid onset psychosis (supersensitivity psychosis) and withdrawal-related relapse". Acta Psychiatrica Scandinavica. 114 (1): 3–13. doi:10.1111/j.1600-0447.2006.00787.x. PMID 16774655. S2CID 6267180.
- ↑ Sacchetti, Emilio; Vita, Antonio; Siracusano, Alberto; Fleischhacker, Wolfgang (2013). Adherence to Antipsychotics in Schizophrenia. Springer Science & Business Media. p. 85. ISBN 9788847026797. Archived from the original on 14 April 2021. Retrieved 21 October 2021.
- ↑ Isbister, GK; Balit, CR; Macleod, D; Duffull, SB (August 2010). "Amisulpride overdose is frequently associated with QT prolongation and torsades de pointes". Journal of Clinical Psychopharmacology. 30 (4): 391–395. doi:10.1097/JCP.0b013e3181e5c14c. PMID 20531221. S2CID 205710487.
- ↑ Joy, JP; Coulter, CV; Duffull, SB; Isbister, GK (August 2011). "Prediction of Torsade de Pointes From the QT Interval: Analysis of a Case Series of Amisulpride Overdoses". Clinical Pharmacology & Therapeutics. 90 (2): 243–245. doi:10.1038/clpt.2011.107. PMID 21716272. S2CID 26412012.
- ↑ 31.0 31.1 31.2 Taylor, D; Paton, C; Shitij, K (2012). Maudsley Prescribing Guidelines in Psychiatry (11th ed.). West Sussex: Wiley-Blackwell. ISBN 978-0-47-097948-8.
- ↑ Levine, M; Ruha, AM (July 2012). "Overdose of atypical antipsychotics: clinical presentation, mechanisms of toxicity and management". CNS Drugs. 26 (7): 601–611. doi:10.2165/11631640-000000000-00000. PMID 22668123. S2CID 24628641.
- ↑ Roth, BL; Driscol, J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Archived from the original on 5 November 2021. Retrieved 14 August 2017.
- ↑ 34.00 34.01 34.02 34.03 34.04 34.05 34.06 34.07 34.08 34.09 34.10 34.11 34.12 34.13 34.14 34.15 34.16 34.17 34.18 34.19 34.20 34.21 34.22 34.23 34.24 34.25 34.26 34.27 34.28 34.29 34.30 34.31 34.32 34.33 34.34 34.35 34.36 34.37 34.38 34.39 34.40 34.41 34.42 34.43 34.44 34.45 34.46 34.47 34.48 Abbas AI, Hedlund PB, Huang XP, Tran TB, Meltzer HY, Roth BL (2009). "Amisulpride is a potent 5-HT7 antagonist: relevance for antidepressant actions in vivo". Psychopharmacology. 205 (1): 119–28. doi:10.1007/s00213-009-1521-8. PMC 2821721. PMID 19337725.
- ↑ 35.0 35.1 Maitre, M.; Ratomponirina, C.; Gobaille, S.; Hodé, Y.; Hechler, V. (April 1994). "Displacement of [3H] gamma-hydroxybutyrate binding by benzamide neuroleptics and prochlorperazine but not by other antipsychotics". European Journal of Pharmacology. 256 (2): 211–214. doi:10.1016/0014-2999(94)90248-8. PMID 7914168.
- ↑ Schoemaker H, Claustre Y, Fage D, Rouquier L, Chergui K, Curet O, Oblin A, Gonon F, Carter C, Benavides J, Scatton B (1997). "Neurochemical characteristics of amisulpride, an atypical dopamine D2/D3 receptor antagonist with both presynaptic and limbic selectivity". J. Pharmacol. Exp. Ther. 280 (1): 83–97. PMID 8996185.
- ↑ Blomme, Audrey; Conraux, Laurence; Poirier, Philippe; Olivier, Anne; Koenig, Jean-Jacques; Sevrin, Mireille; Durant, François; George, Pascal (2000), "Amisulpride, Sultopride and Sulpiride: Comparison of Conformational and Physico-Chemical Properties", Molecular Modeling and Prediction of Bioactivity, Springer US, pp. 404–405, doi:10.1007/978-1-4615-4141-7_97, ISBN 9781461368571
- ↑ 38.0 38.1 Hopkins SC, Wilkinson S, Corriveau TJ, Nishikawa H, Nakamichi K, Loebel A, Koblan KS (May 2021). "Discovery of Non-racemic Amisulpride to Maximize Benefit/Risk of 5-HT7 and D2 Receptor Antagonism for the Treatment of Mood Disorders". Clin Pharmacol Ther. 110 (3): 808–815. doi:10.1002/cpt.2282. PMC 8453756. PMID 33961287.
- ↑ Grattan V, Vaino AR, Prensky Z, Hixon MS (August 2019). "Antipsychotic Benzamides Amisulpride and LB-102 Display Polypharmacy as Racemates, S Enantiomers Engage Receptors D2 and D3, while R Enantiomers Engage 5-HT7". ACS Omega. 4 (9): 14151–14154. doi:10.1021/acsomega.9b02144. PMC 6714530. PMID 31497735.
- ↑ "SEP 4199 - AdisInsight". Archived from the original on 2 June 2021. Retrieved 21 October 2021.
- ↑ "Amisulpride international". Drugs.com. 3 February 2020. Archived from the original on 21 February 2020. Retrieved 26 February 2020.
- ↑ 42.0 42.1 "Active substance: amisulpride" (PDF). 28 September 2017. EMA/658194/2017; Procedure no.: PSUSA/00000167/201701. Archived (PDF) from the original on 15 June 2018. Retrieved 26 February 2020.
- ↑ Lecrubier, Y.; et al. (2001). "Consensus on the Practical Use of Amisulpride, an Atypical Antipsychotic, in the Treatment of Schizophrenia". Neuropsychobiology. 44 (1): 41–46. doi:10.1159/000054913. PMID 11408792. S2CID 21103201.
- ↑ Kaplan, A. (2004). "Psychotropic Medications Around the World". Psychiatric Times. 21 (5).
- ↑ "Barhemsys: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 1 April 2020. Retrieved 26 February 2020.
- ↑ 46.00 46.01 46.02 46.03 46.04 46.05 46.06 46.07 46.08 46.09 46.10 46.11 46.12 46.13 46.14 "Drug Trials Snapshots: Barhemsys". U.S. Food and Drug Administration (FDA). 26 February 2020. Archived from the original on 23 November 2020. Retrieved 27 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Wikipedia articles incorporating the PD-notice template
- Use dmy dates from February 2020
- Articles with invalid date parameter in template
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with changed CASNo identifier
- 5-HT2B antagonists
- 5-HT7 antagonists
- Anilines
- Antidepressants
- Antiemetics
- Atypical antipsychotics
- Benzosulfones
- D2 antagonists
- D3 antagonists
- GHB receptor ligands
- Pyrrolidines
- Salicylamide ethers
- RTT