Zolmitriptan
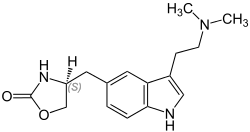 | |
| Names | |
|---|---|
| Trade names | Zomig, others |
| |
| Clinical data | |
| Drug class | Triptan[1] |
| Main uses | Migraines, cluster headaches[2] |
| Side effects | Tingling, nausea, dry mouth, dizziness, medication overuse headaches[1] |
| Interactions | MAO-A inhibitors[1] |
| Pregnancy category | |
| Routes of use | By mouth, nasal spray |
| Typical dose | 2.5 to 5 mg[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601129 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 40% (by mouth) |
| Protein binding | 25% |
| Metabolism | Liver (CYP1A2-mediated, to active metabolite) |
| Elimination half-life | 3 hours |
| Excretion | Kidney (65%) and fecal (35%) |
| Chemical and physical data | |
| Formula | C16H21N3O2 |
| Molar mass | 287.363 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Zolmitriptan, sold under the brand name Zomig among others, is a medication used to treat migraine attacks and cluster headaches.[2] It may also be used to prevent menstrual migraines.[2] It is used as a tablet by mouth or as a nose spray.[2]
Common side effects include tingling, nausea, dry mouth, and dizziness.[1] Other side effects may include medication overuse headaches.[1] Serious side effects may include coronary vasospasm, stroke, high blood pressure, anaphylaxis, and serotonin syndrome.[1] It is unclear if use during pregnancy or breastfeeding is safe.[3][2] It is a triptan that works as a selective serotonin receptor agonist of the 1B and 1D subtypes.[1]
Zolmitriptan was patented in 1990 and approved for medical use in 1997.[4] It is avaliable as a generic medication.[2] In the United Kingdom 6 doses costs the NHS about £12 as of 2020.[2] This amount in the United States costs about 30 USD as of 2021.[5]
Medical uses
Zolmitriptan is used for the acute treatment of migraines with or without aura.[1] It is not generally recommended to prevent migraines or for hemiplegic or basilar migraine.[1]
People who get migraines from aspartame should not use the disintegrating tablet (Zomig ZMT), which contains aspartame.[6]
A 2014 Cochrane review found that zolmitriptan 5 mg nasal spray was more effective than the 5 mg tablet by mouth.[7]
Other uses:
- Acute treatment of cluster headaches—Level A recommendation from the American Academy of Neurology[8]
- Acute treatment of menstrual migraine[8]
Dosage
It is generally used as 2.5 mg by mouth or 5 mg in the nose.[2] No more than 10 mg should be taken in a day.[2]
Zolmitriptan is available as a swallowable tablet, an oral disintegrating tablet, and a nasal spray, in doses of 2.5 and 5 mg.
Contraindications
Zolmitriptan is contraindicated in people with cerebrovascular or cardiovascular disease because 5-HT 1B receptors are present in coronary arteries. Such conditions include, but are not limited to, coronary artery disease, stroke, and peripheral vascular disease.[8]
It is also contraindicated in hemiplegic migraine.[8]
Side effects
As for cardiovascular side effects, zolmitriptan can increase systolic blood pressure in the elderly and increase diastolic blood pressure in both the elderly and young people. Additionally, there is the side effect of a dose-related increase in sedation. There is a risk of headaches caused by medication withdrawal or medication overuse.[8]
Zolmitriptan has a weak affinity for 5-HT 1A receptors; these receptors have implications in the development of serotonin syndrome.[8]
Following administration of cimetidine, the half-life and AUC of zolmitriptan and its active metabolites were approximately doubled.[8]
Mechanism of action
Zolmitriptan is a selective 5-hydroxytryptamine 1B/1D receptor agonist with a weak affinity for the 5-HT 1A receptor subtypes. Its action on 5-HT 1B/1D receptors causes vasoconstriction in intracranial blood vessels; as well it can inhibit the release of pro-inflammatory neuropeptides from trigeminal perivascular nerve endings. It crosses the blood-brain-barrier as evidenced by the presence of radioactive [3H]-zolmitriptan labels within the cells of the trigeminal nucleus caudalis and nucleus tractus solitaries.[8]
Pharmacokinetics
Zolmitriptan has a rapid onset of action and has been detected in the brain as early as within 5 minutes of intranasal administration. On average, zolmitriptan has an oral bioavailability of 40%, a mean volume of distribution of 8.3 L/kg after oral administration, and 2.4L/kg after intravenous administration.[8]
Zolmitriptan is metabolized into three major metabolites by the human hepatic cytochrome P450 enzymes—primarily CYP1A2. Two-thirds of the parent compound breaks down into the active metabolite N-desmethyl-zolmitriptan (183C91), while the remaining one-third separates into the other two inactive metabolites: zolmitriptan N-oxide and an indole acetic acid derivative. It has an elimination half-life of about three hours before it undergoes renal elimination; its clearance is greater than the glomerular filtration rate suggesting that there is some renal tubular secretion of the compound.[8]
Society and culture
Names
Zolmitriptan is marketed by AstraZeneca with the brand names Zomig, Zomigon (Argentina, Canada & Greece), AscoTop (Germany) and Zomigoro (France).
Economics
In 2008, Zomig generated nearly $154 million in sales.[9]
AstraZeneca's U.S. patent on the tablets expired on November 14, 2012, and its pediatric exclusivity extension expired on May 14, 2013.[10] The patent in certain European countries has already expired too, and generic drug maker Actavis released a generic version in those countries, starting in March 2012.[11]
A generic version of the nose spray was first approved in 2021 in the USA.[12]
Legal status
In Russia versions of zolmitriptan, which are not registered in the National registry of medications, may be regarded as narcotic drugs (derivatives of dimethyltriptamine).[13]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 "Zolmitriptan Monograph for Professionals". Drugs.com. Archived from the original on 12 August 2020. Retrieved 3 August 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 2.9 BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 504. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ 3.0 3.1 3.2 "Zolmitriptan Use During Pregnancy". Drugs.com. Archived from the original on 26 November 2020. Retrieved 4 August 2021.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 531. ISBN 9783527607495. Archived from the original on 2020-10-23. Retrieved 2021-01-20.
- ↑ "Zolmitriptan Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 3 August 2021.
- ↑ Newman LC, Lipton RB (2001). "Migraine MLT-Down: An Unusual Presentation of Migraine in Patients With Aspartame-Triggered Headaches". Headache: The Journal of Head and Face Pain (abstract). 41 (9): 899–901. doi:10.1046/j.1526-4610.2001.01164.x. PMID 11703479.
- ↑ Bird S, Derry S, Moore RA (May 2014). "Zolmitriptan for acute migraine attacks in adults". Cochrane Database Syst Rev (5): CD008616. doi:10.1002/14651858.CD008616.pub2. PMC 6485805. PMID 24848613.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 8.9 Abram JA, Patel P (2020). "Zolmitriptan". Statpearls. PMID 32491581.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine.
- ↑ "2008 Top 200 generic drugs by retail dollars" (PDF). Archived from the original (PDF) on 2009-05-21. (332 KB). Drug Topics (May 26, 2009). Retrieved on August 25, 2009.
- ↑ "Archive copy". Archived from the original on 2017-03-17. Retrieved 2021-01-20.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ "Archive copy". Archived from the original on 2012-04-15. Retrieved 2021-01-20.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Research, Center for Drug Evaluation and (10 February 2022). "2021 First Generic Drug Approvals". FDA. Archived from the original on 21 June 2022. Retrieved 22 October 2022.
- ↑ "Постановление Правительства РФ от 30 июня 1998 г. N 681 "Об утверждении перечня наркотических средств, психотропных веществ и их прекурсоров, подлежащих контролю в Российской Федерации" (с изменениями и дополнениями)" (in русский). Гарант. Archived from the original on 2013-04-19. Retrieved 2019-04-29.
ДМТ (диметилтриптамин) и его производные, за исключением производных, включенных в качестве самостоятельных позиций в перечень
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Zolmitriptan Nasal Spray". MedlinePlus. Archived from the original on 2021-01-24. Retrieved 2021-01-20.
- Pages using duplicate arguments in template calls
- CS1 maint: date format
- Webarchive template wayback links
- CS1 maint: archived copy as title
- CS1 русский-language sources (ru)
- Drugs with non-standard pregnancy category
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- Triptans
- 2-Oxazolidinones
- AstraZeneca brands
- 5-HT1D agonists
- RTT