Piperidione
Jump to navigation
Jump to search
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Ascron, Dihyprylon, Dihyprylone, Sedulon, Tusseval |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.909 |
| Chemical and physical data | |
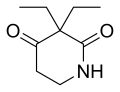
| Formula | C9H15NO2 |
| Molar mass | 169.224 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Piperidione (trade name Sedulon) is a sedative drug, structurally related to methyprylon and pyrithyldione. It used to be marketed by Roche as a cough medicine available in liquid form.[1][2][3] In the US, it was approved by the FDA on grounds of safety alone in 1947. After Roche failed to submit evidence of efficacy to the Drug Efficacy Study Implementation program in 1972, it was withdrawn from the US market.[4]
See also
References
- ^ Wolff PO (1949). "On pethidine and methadone derivatives". Bulletin of the World Health Organization. 2 (2): 193–204. PMC 2553950. PMID 15409516.
- ^ Jacobs S (October 1948). "The use of piperidione as a cough sedative". Medical Times. 76 (10): 445–7. PMID 18102053.
- ^ Rimoldi R, Fioretti M, Bandella M (February 1985). "[Use of an antitussive drug in pulmonary pathology]". Bollettino Chimico Farmaceutico. 124 (2): 1S–6S. PMID 3839404.
- ^ Certain Preparations Containing Dihyprylone or Pipazethate Hydrochloride; Notice of Withdrawal of Approval of New-Drug Applications (PDF). Federal Register (Report). Vol. 37. August 5, 1972. p. 15887. FDC–D–458.
Categories:
- Articles with short description
- Short description matches Wikidata
- Articles with changed CASNo identifier
- ECHA InfoCard ID from Wikidata
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Withdrawn drugs
- Antitussives
- 2-Piperidinones
- GABAA receptor positive allosteric modulators
- Sedatives
- 4-Piperidinones
- All stub articles
- Respiratory system drug stubs