Felodipine
 | |
 | |
| Names | |
|---|---|
| Trade names | Plendil |
| |
| Clinical data | |
| Drug class | Calcium channel blocker[1] |
| Main uses | High blood pressure, angina[1][2] |
| Side effects | Swelling, headache, flushing[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Onset of action | 2.5–5 hours |
| Typical dose | 2.5 to 10 mg OD[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692016 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 15% |
| Metabolism | Liver |
| Elimination half-life | 25 hours[3] |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C18H19Cl2NO4 |
| Molar mass | 384.25 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Felodipine, sold under the brand name Plendil among others, is a medication is used to treat high blood pressure and heart related chest pain (angina).[1][2] It is one of several first line options.[1] It is taken by mouth.[1]
Common side effects include swelling, headache, and flushing.[1] Other side effects may include syncope and low blood pressure.[1] There are concerns regarding negative effects if used during pregnancy.[5] It is a calcium channel blocker of the dihydropyridine type.[1]
Felodipine was patented in 1978 and approved for medical use in 1988.[6] It is available as a generic medication.[2] In the United Kingdom 4 weeks of 5 mg per day costs about £4 as of 2021.[2] This amount is about 11 USD in the United States.[7] It is also available in combination with ramipril.[2]
Medical uses
Felodipine is used to treat high blood pressure and stable angina.[3][8]
It should not be used for people who are pregnant, have acute heart failure, are having a heart attack, have an obstructed heart valve, or have obstructions that block bloodflow out of the heart.[3]
For people with liver failure the dose needs to be lowered, because felodipine is cleared by the liver.[3]
Dosage
It is generally started at 5 mg per day and may be increased to 10 mg per day if needed.[2] In the elderly 2.5 mg per day may be used.[2]
Side effects
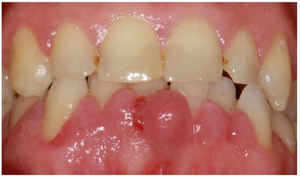
The only very common side effect, occurring in more than 1/10 people, is pain and swelling in the arms and legs.[3]
Common side effects, occurring in between 1% and 10% of people, include flushing, headache, heart palpitations, dizziness and fatigue.[3]
Felodipine can exacerbate gingivitis.[3]
Interactions
Felodipine is metabolized by cytochrome P450 3A4, so substances that inhibit or activate CYP3A4 can strongly effect how much felodipine is present.[3]
CYP3A4 inhibitors, which increase the amount of felodipine available per dose, include cimetidine, erythromycin, itraconazole, ketoconazole, HIV protease inhibitors, and grapefruit juice.[3][9]
CYP3A4 activators, which decrease the amount of felodipine available per dose, include phenytoin, carbamazepine, rifampicin, barbiturates, efavirenz, nevirapine, and Saint John's wort.[3]
Mechanism of action
Felodipine is a calcium channel blocker.[3] Felodipine has additionally been found to act as an antagonist of the mineralocorticoid receptor, or as an antimineralocorticoid.[10]
Different calcium channels are present in vascular tissue and cardiac tissue; an in vitro study on human vascular and cardiac tissues comparing how selective various calcium channel blockers are for vascular compared to cardiac tissue found the following vascular/cardiac tissue ratios: mibefradil 41, felodipine 12; nifedipine 7, amlodipine 5, and verapamil 0.2.[11]: 172
Chemistry
Felodipine is a member of the 1,4-dihydropyridine class of calcium channel blockers.[11]: 20–21 It is a racemic mixture, and is insoluble in water but is soluble in dichloromethane and ethanol.[11]: 25
History
The Swedish company Hässle, a division of Astra AB, discovered felodipine;[12] it filed a patent application in 1979 claiming felodipine as an antihypertensive drug.[13][14] Astra partnered this drug and others with Merck & Co. in the US under a 1982 agreement between the companies.[12] The drug was approved by the FDA in 1991 after a three and a half year review; the drug entered a very crowded market the included the other calcium channel blockers nifedipine, verapamil, nicardipine, and isradipine.[12] The FDA gave the drug a 1C rating, meaning that it found little difference between felodipine and the drugs already approved for the same use.[12]
In 1994 Astra AB and Merck changed their partnership to a joint venture called Astra Merck,[15] and in 1998 Astra (by that time, AstraZeneca) bought out Merck's rights in the joint venture.[16]
The first generics became available in Sweden in 2003[17] and in the US in 2004.[18]: 155
In April 2016, AstraZeneca announced that they were selling the right to market felodipine in China to China Medical System Holdings for $310 million; AZ would continue to manufacture the drug.[19]
Society and culture
As of 2016, felodipine was marketed under many brand names worldwide: Auronal, Cardioplen, Catrazil, Dewei, Dilahex, Enfelo, Erding, Fedil, Fedisyn, Feldil, Felicipin, Felo, Felocard, Felocor, Feloday, Felodil, Felodin, Felodip, Felodipin, Felodipina, Felodipine, Felodipino, Felodistad, Felogard, Felohexal, Felop, Felopine, Felostad, Feloten, Felotens, Felpin, Flodicar, Flodil, Keliping, Keydipin, Lodistad, Modip, Munobal, Nirmadil, Parmid, Penedil, Perfudal, Phelop, Phenodical, Plendil, Plentopine, Polo, Presid, Preslow, Prevex, Renedil, Sistar, Splendil, Stapin, Topidil, Vascalpha, Versant, and XiaoDing.[20]
The combination of felodipine and candesartan was marketed as Atacand.[20]
The combination of felodipine and ramipril was marketed as Delmuno, Tazko, Triacor, Triapin, Triasyn, Tri-Plen, Unimax, and Unitens.[20]
The combination of felodipine and enalapril was marketed as Lexxel.[20]
The combination of felodipine and metoprolol was marketed as Logimat, Logimax, and Mobloc.[20]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 "Felodipine Monograph for Professionals". Drugs.com. Archived from the original on 15 August 2020. Retrieved 10 December 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 174. ISBN 978-0857114105.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 "Felopidine UK label". UK Electronic Medicines Compendium. 15 September 2015. Archived from the original on 16 August 2019. Retrieved 9 March 2021.
- ↑ Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, Volume 115, 2013, pp. 887-890
- ↑ "Felodipine (Plendil) Use During Pregnancy". Drugs.com. Archived from the original on 29 November 2020. Retrieved 10 December 2021.
- ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 465. ISBN 9783527607495. Archived from the original on 27 August 2021. Retrieved 9 March 2021.
- ↑ "Felodipine ER Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 10 December 2021.
- ↑ "Felodipine US label" (PDF). FDA. October 2012. Archived (PDF) from the original on 31 March 2021. Retrieved 9 March 2021.
- ↑ Kiani J, Imam SZ (October 2007). "Medicinal importance of grapefruit juice and its interaction with various drugs". Nutrition Journal. 6 (33): 33. doi:10.1186/1475-2891-6-33. PMC 2147024. PMID 17971226..
- ↑ Luther JM (September 2014). "Is there a new dawn for selective mineralocorticoid receptor antagonism?". Current Opinion in Nephrology and Hypertension. 23 (5): 456–61. doi:10.1097/MNH.0000000000000051. PMC 4248353. PMID 24992570.
- ↑ 11.0 11.1 11.2 Joshi GS, Burnett JC, Abraham DJ (2003). "Cardiac Drugs: Antianginal, Vasodilators, Antiarrhythmic". In Abraham DJ (ed.). Burger's medicinal chemistry and drug discovery. Volume 3: Cardiovascular Agents and Endocrines (6th ed.). Hoboken, N.J.: Wiley. ISBN 9780471270904.
- ↑ 12.0 12.1 12.2 12.3 "Merck's Plendil (Felodipine) Approved with "1C" Rating". Pink Sheet. 5 August 1991. Archived from the original on 15 November 2016. Retrieved 9 March 2021.
- ↑ Astrazeneca AB v. Mutual Pharmaceutical Co. (United States Court of Appeals for the Federal Circuit 2004).Text
- ↑ US patent 4,264,611, Berntsson, Peder; Carlsson, Stig & Gaarder, Jan et al., "2,6-Dimethyl-4-2,3-Disubstituted Phenyl-1,4-Dihydro-Pyridine-3,5 Dicarboxylic Acid-3,5-Asymmetric Diesters having Hypotensive Properties, as well as Method for Treating Hypertensive Conditions and Pharmaceutical Preparations Containing Same", issued 28 April 1981, assigned to Aktiebolaget Hassle
- ↑ George J (28 July 1997). "Secret of Astra Merck". Philadelphia Business Journal. Philadelphia. Archived from the original on 10 November 2016. Retrieved 7 November 2016.
- ↑ "Astra, Merck restructure". CNNMoney. 19 June 1998. Archived from the original on 16 August 2019. Retrieved 7 November 2016.
- ↑ Jönsson B (2008). "Sweden". In Rapoport J, Jacobs P, Jonsson E (eds.). Cost Containment and Efficiency in National Health Systems a Global Comparison. Weinheim: Wiley-VCH. p. 218. ISBN 9783527622955. Archived from the original on 21 March 2017. Retrieved 9 March 2021.
- ↑ Approved Drug Products with Therapeutic Equivalence Evaluations (PDF) (36th ed.). FDA. 2014. Archived (PDF) from the original on 15 November 2016. Retrieved 9 March 2021.
- ↑ "AstraZeneca enters licensing agreement with China Medical System Holdings for hypertension medicine" (Press release). AstraZeneca. 29 February 2016. Archived from the original on 3 March 2021. Retrieved 7 November 2016.
- ↑ 20.0 20.1 20.2 20.3 20.4 "International brand names: Felodipine". Drugs.com. Archived from the original on 15 November 2016. Retrieved 15 November 2016.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Use dmy dates from February 2015
- Articles with invalid date parameter in template
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- Antimineralocorticoids
- Calcium channel blockers
- Dihydropyridines
- AstraZeneca brands
- Carboxylate esters
- Chloroarenes
- RTT