Dextromethorphan
 | |
 | |
| Names | |
|---|---|
| Trade names | Robitussin, Delsym, DM, DexAlone, Duract, others |
| |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682492 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 11%[1] |
| Metabolism | Liver enzymes: major CYP2D6, minor CYP3A4, and minor CYP3A5 |
| Elimination half-life | 2–4 hours (extensive metabolizers); 24 hours (poor metabolizers)[2] |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C18H25NO |
| Molar mass | 271.404 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 111 °C (232 °F) |
| |
| |
Dextromethorphan is a medication often used in over-the-counter cough medicines.[3] In cough medicine it may be combined with paracetamol (acetaminophine), chlorpheniramine, pseudoephedrine, or guaifenesin.[3] There is no clear evidence of benefit in children under six to twelve years of age with some evidence of harm; thus such use is not recommended.[3][4] It is taken by mouth.[3]
Side effects may include nausea, sleepiness, and dizziness.[3] Serious side effects may include serotonin syndrome and abuse.[3] While safety in pregnancy and breastfeeding has not been studied, such use is believed to be okay.[5][6] It is in the morphinan class of medications and acts within the brain to suppress coughing.[3][4]
Dextromethorphan was patented in 1949 and approved for medical use in 1953.[7] Dextromethorphan is also used recreationally.[3] At high doses dextromethorphan acts as a dissociative hallucinogen and may result in seizures.[8] A number of other uses for dextromethorphan are being studied.[9]
Medical uses

Cough suppression
The primary use of dextromethorphan is as a cough suppressant, for the temporary relief of cough caused by minor throat and bronchial irritation (such as commonly accompanies the flu and common cold), as well as those resulting from inhaled particle irritants.[10] However, controlled studies have found the symptomatic effectiveness of dextromethorphan similar to placebo.[11]
Mental health
In 2010, the FDA approved the combination drug dextromethorphan/quinidine for the treatment of pseudobulbar affect (uncontrollable laughing/crying).[12] Dextromethorphan is active agent in the combination; quinidine merely serves to inhibit the degradation of dextromethorphan via inhibition of CYP2D6.[12]
Dosage
The maximum recommended dose in adults is 30 mg four times per day.[9]
Side effects
Side effects of dextromethorphan at normal therapeutic doses can include:[2][10][13]
A rare side effect is respiratory depression.[10]
Contraindications
Because dextromethorphan can trigger a histamine release (allergic reaction), atopic children, who are especially susceptible to allergic reactions, should be administered dextromethorphan only if absolutely necessary, and only under the strict supervision of a healthcare professional.[13]
Dependence and withdrawal
Dextromethorphan has produced psychological dependence in people who used it recreationally. However, it does not produce physical addiction, according to the WHO Committee on Drug Dependence.[14] It is considered less addictive than the other common weak opiate cough suppressant, codeine.[2] Since dextromethorphan also acts as a serotonin reuptake inhibitor, users report that regular recreational use over a long period of time can cause withdrawal symptoms similar to those of antidepressant discontinuation syndrome. Additionally, disturbances have been reported in sleep, senses, movement, mood, and thinking.
Overdose
Adverse effects of dextromethorphan in overdose at doses 3 to 10 times the recommended therapeutic dose:[15][failed verification]
- mild nausea
- restlessness
- insomnia
- talking fast
- dilated pupils
- glassy eyes
- dizziness
At doses 15 to 75 times the recommended therapeutic dose:[15][failed verification][16]
- hallucinations
- dissociation
- vomiting
- delusions of grandeur
- blurred vision
- double vision
- bloodshot eyes
- dilated pupils
- sweating
- fever
- bruxia (teeth grinding)
- hypotension
- hypertension
- tachycardia
- shallow respiration
- diarrhea
- urinary retention
- muscle spasms
- sedation
- paresthesia
- hypomania
- slurred speech
- false positives on drug screens
- blackouts
- inability to focus eyes
- skin rash
- severe itchiness
- spontaneous memory recall
- acute psychosis
Episodic acute psychosis can occur when high doses of dextromethorphan taken for recreational use, and an abundance of psychiatric symptoms can result, including dissociation and other PCP-like symptoms.[17]
Interactions
Dextromethorphan should not be taken with monoamine oxidase inhibitors[13] due to the potential for serotonin syndrome, which is a potentially life-threatening condition that can occur rapidly, due to a buildup of an excessive amount of serotonin in the body.
Patients who are taking dextromethorphan should exercise caution when drinking grapefruit juice or eating grapefruit, as compounds in grapefruit affect a number of drugs, including dextromethorphan, through the inhibition of the cytochrome P450 system in the liver, and can lead to excessive accumulation of the drug which both increases and prolongs effects. Grapefruit and grapefruit juices (especially white grapefruit juice, but also including other citrus fruits such as bergamot and lime, as well as a number of noncitrus fruits[18]) generally are recommended to be avoided while using dextromethorphan and numerous other medications.
Pharmacology
Dextromethorphan possesses only a minimal degree of the mu-opioid receptor activity typical of morphinan compounds and exerts its therapeutic effects through several other receptors. In its pure form, dextromethorphan occurs as a white powder.[19]
Pharmacodynamics
| Site | DXM | DXO | Species | Ref |
|---|---|---|---|---|
| NMDAR (MK-801) |
2,120–8,945 | 486–906 | Rat | [21] |
| σ1 | 142–652 | 118–481 | Rat | [21] |
| σ2 | 11,060–22,864 | 11,325–15,582 | Rat | [21] |
| MOR | 1,280 ND |
420 >1,000 |
Rat Human |
[21] [24] |
| DOR | 11,500 | 34,700 | Rat | [21] |
| KOR | 7,000 | 5,950 | Rat | [21] |
| SERT | 23–40 | 401–484 | Rat | [21] |
| NET | ≥240 | ≥340 | Rat | [21] |
| DAT | >1,000 | >1,000 | Rat | [21] |
| 5-HT1A | >1,000 | >1,000 | Rat | [21] |
| 5-HT1B/1D | 61% at 1 μM | 54% at 1 μM | Rat | [21] |
| 5-HT2A | >1,000 | >1,000 | Rat | [21] |
| α1 | >1,000 | >1,000 | Rat | [21] |
| α2 | 60% at 1 μM | >1,000 | Rat | [21] |
| β | >1,000 | 35% at 1 μM | Rat | [21] |
| D2 | >1,000 | >1,000 | Rat | [21] |
| H1 | >1,000 | 95% at 1 μM | Rat | [21] |
| mAChRs | >1,000 | 100% at 1 μM | Rat | [21] |
| nAChRs | 700–8,900 (IC50) |
1,300–29,600 (IC50) |
Rat | [21] |
| VDSCs | >50,000 (IC50) | ND | Rat | [25][26] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. | ||||
Dextromethorphan has been found to possess the following actions (<1 μM) using rat tissues:[21][27]
- Uncompetitive antagonist of the NMDA receptor via the MK-801/PCP site[27]
- SERT and NET blocker (cf. serotonin–norepinephrine reuptake inhibitor)
- Sigma σ1 receptor agonist
- Negative allosteric modulator of nicotinic acetylcholine receptors
- Ligand of the serotonin 5-HT1B/1D, histamine H1, α2-adrenergic, and muscarinic acetylcholine receptors
Dextromethorphan is a prodrug of dextrorphan, which is the actual mediator of most of its dissociative effects through acting as a more potent NMDA receptor antagonist than dextromethorphan itself.[28] What role, if any, (+)-3-methoxymorphinan, dextromethorphan's other major metabolite, plays in its effects is not entirely clear.[29]
Pharmacokinetics
Following oral administration, dextromethorphan is rapidly absorbed from the gastrointestinal tract, where it enters the bloodstream and crosses the blood–brain barrier.[citation needed]
At therapeutic doses, dextromethorphan acts centrally (meaning that it acts on the brain) as opposed to locally (on the respiratory tract). It elevates the threshold for coughing, without inhibiting ciliary activity. Dextromethorphan is rapidly absorbed from the gastrointestinal tract and converted into the active metabolite dextrorphan in the liver by the cytochrome P450 enzyme CYP2D6. The average dose necessary for effective antitussive therapy is between 10 and 45 mg, depending on the individual. The International Society for the Study of Cough recommends "an adequate first dose of medication is 60 mg in the adult and repeat dosing should be infrequent rather than qds recommended."[30]
Dextromethorphan has an elimination half-life of approximately 4 hours in individuals with an extensive metabolizer phenotype; this is increased to approximately 13 hours when dextromethorphan is given in combination with quinidine.[23] The duration of action after oral administration is about three to eight hours for dextromethorphan hydrobromide, and 10 to 12 hours for dextromethorphan polistirex. Around one in 10 of the Caucasian population has little or no CYP2D6 enzyme activity, leading to long-lived high drug levels.[30]
Metabolism

The first pass through the hepatic portal vein results in some of the drug being metabolized by O-demethylation into an active metabolite of dextromethorphan called dextrorphan, the 3-hydroxy derivative of dextromethorphan. The therapeutic activity of dextromethorphan is believed to be caused by both the drug and this metabolite. Dextromethorphan also undergoes N-demethylation (to 3-methoxymorphinan or MEM),[32] and partial conjugation with glucuronic acid and sulfate ions. Hours after dextromethorphan therapy, (in humans) the metabolites (+)-3-hydroxy-N-methylmorphinan, (+)-3-morphinan, and traces of the unchanged drug are detectable in the urine.[13]
A major metabolic catalyst involved is the cytochrome P450 enzyme known as 2D6, or CYP2D6. A significant portion of the population has a functional deficiency in this enzyme and are known as poor CYP2D6 metabolizers. O-demethylation of dextromethorphan to dextrorphan contributes to at least 80% of the dextrorphan formed during dextromethorphan metabolism.[32] As CYP2D6 is a major metabolic pathway in the inactivation of dextromethorphan, the duration of action and effects of dextromethorphan can be increased by as much as three times in such poor metabolizers.[33] In one study on 252 Americans, 84.3% were found to be "fast" (extensive) metabolizers, 6.8% to be "intermediate" metabolizers, and 8.8% were "slow" metabolizers of dextromethorphan.[34] A number of alleles for CYP2D6 are known, including several completely inactive variants. The distribution of alleles is uneven amongst ethnic groups.
A large number of medications are potent inhibitors of CYP2D6. Some types of medications known to inhibit CYP2D6 include certain SSRIs and tricyclic antidepressants, some antipsychotics, and the commonly available antihistamine diphenhydramine. Therefore, the potential of interactions exists between dextromethorphan and medications that inhibit this enzyme, particularly in slow metabolizers.[citation needed]
Dextromethorphan is also metabolized by CYP3A4. N-demethylation is primarily accomplished by CYP3A4, contributing to at least 90% of the MEM formed as a primary metabolite of dextromethorphan.[32]
A number of other CYP enzymes are implicated as minor pathways of dextromethorphan metabolism. CYP2B6 is more effective than CYP3A4 at N-demethylation of dextromethorphan, but since the average individual has a much lower CYP2B6 content in the liver compared to CYP3A4, most N-demethylation of dextromethorphan is catalyzed by CYP3A4.[32]
Chemistry
Dextromethorphan is the dextrorotatory enantiomer of levomethorphan, which is the methyl ether of levorphanol, both opioid analgesics. It is named according to IUPAC rules as (+)-3-methoxy-17-methyl-9α,13α,14α-morphinan. As its pure form, dextromethorphan occurs as an odorless, opalescent white powder. It is freely soluble in chloroform and insoluble in water; the hydrobromide salt is water-soluble up to 1.5 g/100 mL at 25 °C.[35] Dextromethorphan is commonly available as the monohydrated hydrobromide salt, however some newer extended-release formulations contain dextromethorphan bound to an ion-exchange resin based on polystyrene sulfonic acid. Dextromethorphan's specific rotation in water is +27.6° (20 °C, Sodium D-line).[citation needed]
History
The racemic parent compound racemorphan was first described in a Swiss and US patent application from Hoffmann-La Roche in 1946 and 1947, respectively; a patent was granted in 1950. A resolution of the two isomers of racemorphan with tartaric acid was published in 1952,[36] and dextromethorphan was successfully tested in 1954 as part of US Navy and CIA-funded research on nonaddictive substitutes for codeine.[37] Dextromethorphan was approved by the FDA in 1958 as an over-the-counter antitussive.[36] As had been initially hoped, dextromethorphan was a solution for some of the problems associated with the use of codeine phosphate as a cough suppressant, such as sedation and opiate dependence, but like the dissociative anesthetics phencyclidine and ketamine, dextromethorphan later became associated with nonmedical use.[36][38]
During the 1960s and 1970s, dextromethorphan became available in an over-the-counter tablet form by the brand name Romilar. In 1973, Romilar was taken off the shelves after a burst in sales because of frequent misuse. A few years later, products with an unpleasant taste were introduced (such as Robitussin, Vicks-44, and Dextrotussion), but later the same manufacturers began producing products with a better taste.[38] The advent of widespread internet access in the 1990s allowed users to rapidly disseminate information about dextromethorphan, and online discussion groups formed around use and acquisition of the drug. As early as 1996, dextromethorphan hydrobromide powder could be purchased in bulk from online retailers, allowing users to avoid consuming dextromethorphan in syrup preparations.[36] As of January 1, 2012, dextromethorphan is prohibited for sale to minors in the State of California and in the State of Oregon as of January 1, 2018, except with a doctor's prescription.[39] Several other states have also began regulating sales of dextromethorphan to minors.
In Indonesia, the National Agency of Drug and Food Control (BPOM-RI) prohibited single-component dextromethorphan drug sales with or without prescription. Indonesia is the only country in the world that makes single-component dextromethorphan illegal even by prescription[40] and violators may be prosecuted by law. National Anti-Narcotics Agency (BNN RI) has even threatened to revoke pharmacies' and drug stores' licenses if they still stock dextromethorphan, and will notify the police for criminal prosecution.[41] As a result of this regulation, 130 drugs have been withdrawn from the market, but drugs containing multicomponent dextromethorphan can be sold over the counter.[42] In its official press release, BPOM-RI also stated that dextromethorphan is often used as a substitute for marijuana, amphetamine, and heroin by drug abusers, and its use as an antitussive is less beneficial nowadays.[43]
Society and culture
Cost
The cost of this medication at 15 mg is about $11 for 20 capsules in US[44]
-
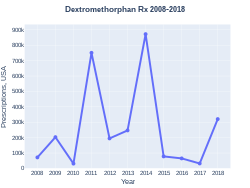
Dextromethorphan costs (US)
-
Dextromethorphan prescriptions (US)
Marketing
It may be used in generic labels and store brands, Benylin DM, Mucinex DM, Camydex-20 tablets, Robitussin, NyQuil, Dimetapp, Vicks, Coricidin, Delsym, TheraFlu, Charcoal D, Cinfatós and others. It has been used in counterfeit medications.[45]
Recreational use

Over-the-counter preparations containing dextromethorphan have been used in manners inconsistent with their labeling, often as a recreational drug.[38] At doses much higher than medically recommended, dextromethorphan and its major metabolite, dextrorphan, acts as an NMDA receptor antagonist, which produces dissociative hallucinogenic states somewhat similar to ketamine and phencyclidine.[46] It may produce distortions of the visual field – feelings of dissociation, distorted bodily perception, excitement, and a loss of sense of time. Some users report stimulant-like euphoria, particularly in response to music. Dextromethorphan usually provides its recreational effects in a non-linear fashion, so that they are experienced in significantly varied stages. These stages are commonly referred to as "plateaus". These plateaus are numbered from one to four, with the first having the mildest effects to fourth being the most intense. Each plateau is said to come with different related effects and experiences.[47] Teens tend to have a higher likelihood to use dextromethorphan-related drugs as they are easier to access.[48]
Research
Dextromethorphan/quinidine is also under investigation for the treatment of a variety of other neurological and neuropsychiatric conditions besides pseudobulbar affect, such as agitation associated with Alzheimer's disease and major depressive disorder.[12]
The combination medicine dextromethorphan/bupropion is in clinical trials for major depressive disorder, with fast track designation granted by the FDA in 2017.[49] The combination showed positive results in phase 2 and phase 3 clinical trials.[50][51]
See also
References
- ↑ Kukanich B, Papich MG (October 2004). "Plasma profile and pharmacokinetics of dextromethorphan after intravenous and oral administration in healthy dogs". Journal of Veterinary Pharmacology and Therapeutics. 27 (5): 337–41. doi:10.1111/j.1365-2885.2004.00608.x. PMID 15500572.
- ↑ 2.0 2.1 2.2 "Balminil DM, Benylin DM (dextromethorphan) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Archived from the original on 31 March 2019. Retrieved 15 April 2014.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 "Dextromethorphan Hydrobromide Monograph for Professionals". Drugs.com. Archived from the original on 6 November 2020. Retrieved 5 October 2020.
- ↑ 4.0 4.1 BNF 79. London: Pharmaceutical Press. March 2020. p. 305. ISBN 978-0857113658.
- ↑ "Dextromethorphan Use During Pregnancy". Drugs.com. Archived from the original on 4 December 2020. Retrieved 5 October 2020.
- ↑ "Dextromethorphan". Drugs and Lactation Database (LactMed). 2006. PMID 30000516. Archived from the original on 2021-08-27. Retrieved 2020-10-05.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 527. ISBN 9783527607495. Archived from the original on 2021-08-29. Retrieved 2020-09-06.
- ↑ Abuse, National Institute on Drug (NaN). "What Are the Effects of Common Dissociative Drugs on the Brain and Body?". National Institute on Drug Abuse. Archived from the original on 10 October 2020. Retrieved 5 October 2020.
{{cite web}}: Check date values in:|date=(help) - ↑ 9.0 9.1 Oh, SR; Agrawal, S; Sabir, S; Taylor, A (January 2020). "Dextromethorphan". PMID 30855804.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 10.0 10.1 10.2 Rossi, S, ed. (2013). Australian Medicines Handbook. Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.[page needed]
- ↑ Van Amburgh JA. "Do Cough Remedies Work?". Medscape. Archived from the original on 31 March 2019. Retrieved 10 April 2016.
- ↑ 12.0 12.1 12.2 Nguyen L, Thomas KL, Lucke-Wold BP, Cavendish JZ, Crowe MS, Matsumoto RR (March 2016). "Dextromethorphan: An update on its utility for neurological and neuropsychiatric disorders". Pharmacology & Therapeutics. 159: 1–22. doi:10.1016/j.pharmthera.2016.01.016. PMID 26826604. Archived from the original on 2019-07-03. Retrieved 2019-07-03.
- ↑ 13.0 13.1 13.2 13.3 "Dextromethorphan". National Highway Traffic Safety Administration (NHTSA). Archived from the original on 2008-08-01.
- ↑ WHO Expert Committee on Drug Dependence, Seventeenth Report (PDF). World Health Organization. 1970. p. 24. Archived (PDF) from the original on 2009-10-18. Retrieved 2008-12-29.
- ↑ 15.0 15.1 "Teen Drug Abuse: Cough Medicine and DXM (Dextromethorphan)". webmd. Archived from the original on 2017-11-21.
- ↑ Martinak B, Bolis RA, Black JR, Fargason RE, Birur B (September 2017). "Dextromethorphan in Cough Syrup: The Poor Man's Psychosis". Psychopharmacology Bulletin. 47 (4): 59–63. PMC 5601090. PMID 28936010.
- ↑ Martinak B, Bolis RA, Black JR, Fargason RE, Birur B (September 2017). "Dextromethorphan in Cough Syrup: The Poor Man's Psychosis". Psychopharmacology Bulletin. 47 (4): 59–63. PMC 5601090. PMID 28936010.
- ↑ "Inhibitors of CYP3A4". ganfyd.org. Archived from the original on 2017-07-20. Retrieved 23 August 2013.
- ↑ "Reference Tables: Description and Solubility - D". Archived from the original on 2017-07-04. Retrieved 2011-05-06.
- ↑ Roth, BL; Driscol, J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Archived from the original on 28 August 2021. Retrieved 14 August 2017.
- ↑ 21.00 21.01 21.02 21.03 21.04 21.05 21.06 21.07 21.08 21.09 21.10 21.11 21.12 21.13 21.14 21.15 21.16 21.17 21.18 21.19 21.20 Nguyen L, Thomas KL, Lucke-Wold BP, Cavendish JZ, Crowe MS, Matsumoto RR (March 2016). "Dextromethorphan: An update on its utility for neurological and neuropsychiatric disorders" (PDF). Pharmacology & Therapeutics. 159: 1–22. doi:10.1016/j.pharmthera.2016.01.016. PMID 26826604. Archived (PDF) from the original on 2020-05-23. Retrieved 2019-09-24.
- ↑ Werling LL, Keller A, Frank JG, Nuwayhid SJ (October 2007). "A comparison of the binding profiles of dextromethorphan, memantine, fluoxetine and amitriptyline: treatment of involuntary emotional expression disorder". Experimental Neurology. 207 (2): 248–57. doi:10.1016/j.expneurol.2007.06.013. PMID 17689532. S2CID 38476281.
- ↑ 23.0 23.1 Taylor CP, Traynelis SF, Siffert J, Pope LE, Matsumoto RR (August 2016). "Pharmacology of dextromethorphan: Relevance to dextromethorphan/quinidine (Nuedexta®) clinical use". Pharmacology & Therapeutics. 164: 170–82. doi:10.1016/j.pharmthera.2016.04.010. PMID 27139517.
- ↑ Raynor K, Kong H, Mestek A, Bye LS, Tian M, Liu J, et al. (January 1995). "Characterization of the cloned human mu opioid receptor". The Journal of Pharmacology and Experimental Therapeutics. 272 (1): 423–8. PMID 7815359.
- ↑ Lee JH, Shin EJ, Jeong SM, Lee BH, Yoon IS, Lee JH, et al. (June 2007). "Effects of dextrorotatory morphinans on brain Na+ channels expressed in Xenopus oocytes". European Journal of Pharmacology. 564 (1–3): 7–17. doi:10.1016/j.ejphar.2007.01.088. PMID 17346698.
- ↑ Gao XF, Yao JJ, He YL, Hu C, Mei YA (2012). "Sigma-1 receptor agonists directly inhibit Nav1.2/1.4 channels". PLOS ONE. 7 (11): e49384. Bibcode:2012PLoSO...749384G. doi:10.1371/journal.pone.0049384. PMC 3489664. PMID 23139844.
- ↑ 27.0 27.1 Burns JM, Boyer EW (2013). "Antitussives and substance abuse". Substance Abuse and Rehabilitation. 4: 75–82. doi:10.2147/SAR.S36761. PMC 3931656. PMID 24648790.
- ↑ Chou YC, Liao JF, Chang WY, Lin MF, Chen CF (March 1999). "Binding of dimemorfan to sigma-1 receptor and its anticonvulsant and locomotor effects in mice, compared with dextromethorphan and dextrorphan". Brain Research. 821 (2): 516–9. doi:10.1016/S0006-8993(99)01125-7. PMID 10064839. S2CID 22762264.
- ↑ Schmider J, Greenblatt DJ, Fogelman SM, von Moltke LL, Shader RI (April 1997). "Metabolism of dextromethorphan in vitro: involvement of cytochromes P450 2D6 and 3A3/4, with a possible role of 2E1". Biopharmaceutics & Drug Disposition. 18 (3): 227–40. doi:10.1002/(SICI)1099-081X(199704)18:3<227::AID-BDD18>3.0.CO;2-L. PMID 9113345.
- ↑ 30.0 30.1 Morice AH. "Cough". International Society for the Study of Cough. Archived from the original on 2017-05-09.
- ↑ Strauch K, Lutz U, Bittner N, Lutz WK (August 2009). "Dose-response relationship for the pharmacokinetic interaction of grapefruit juice with dextromethorphan investigated by human urinary metabolite profiles". Food and Chemical Toxicology. 47 (8): 1928–35. doi:10.1016/j.fct.2009.05.004. PMID 19445995.
- ↑ 32.0 32.1 32.2 32.3 Yu A, Haining RL (November 2001). "Comparative contribution to dextromethorphan metabolism by cytochrome P450 isoforms in vitro: can dextromethorphan be used as a dual probe for both CTP2D6 and CYP3A activities?". Drug Metabolism and Disposition. 29 (11): 1514–20. PMID 11602530. Archived from the original on 2020-03-12. Retrieved 2015-04-26.
- ↑ Capon DA, Bochner F, Kerry N, Mikus G, Danz C, Somogyi AA (September 1996). "The influence of CYP2D6 polymorphism and quinidine on the disposition and antitussive effect of dextromethorphan in humans". Clinical Pharmacology and Therapeutics. 60 (3): 295–307. doi:10.1016/S0009-9236(96)90056-9. PMID 8841152. S2CID 10147669.
- ↑ Woodworth JR, Dennis SR, Moore L, Rotenberg KS (February 1987). "The polymorphic metabolism of dextromethorphan". Journal of Clinical Pharmacology. 27 (2): 139–43. doi:10.1002/j.1552-4604.1987.tb02174.x. PMID 3680565. S2CID 37950361.
- ↑ "Dextromethorphan (PIM 179)". www.inchem.org. Archived from the original on 2017-03-10. Retrieved 2018-03-24.
- ↑ 36.0 36.1 36.2 36.3 Morris H, Wallach J (2014). "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis. 6 (7–8): 614–32. doi:10.1002/dta.1620. PMID 24678061.
- ↑ "Memorandum for the Secretary of Defense" (PDF). Archived (PDF) from the original on 2017-09-16. Retrieved 2013-07-28.
- ↑ 38.0 38.1 38.2 "Dextromethorphan (DXM)". Cesar.umd.edu. Archived from the original on 2018-01-06. Retrieved 2013-07-28.
- ↑ "Senate Bill No. 514" (PDF). An act to add Sections 11110 and 11111 to the Health and Safety Code, relating to nonprescription drugs. State of California, Legislative Counsel. Archived (PDF) from the original on 2018-03-08.
- ↑ http://nasional.news.viva.co.id/news/read/506418-bpom-tetap-batalkan-izin-edar-obat-dekstrometorfan Archived 2015-05-28 at the Wayback Machine[full citation needed]
- ↑ "SINDOnews | Berita Daerah Dan Provinsi Di Indonesia". daerah.sindonews.com (in Bahasa Indonesia). Retrieved 2017-12-10.[dead link]
- ↑ "Pimpinan dan Apoteker Penanggung Jawab" (PDF). Archived from the original (PDF) on 2017-08-10.
- ↑ "Badan Pengawas Obat dan Makanan - Republik Indonesia". www.pom.go.id. Archived from the original on 2017-02-03. Retrieved 2017-12-10.
- ↑ "Dextromethorphan Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 3 February 2020. Retrieved 18 February 2021.
- ↑ "Fake drugs: the global industry putting your life at risk". Mosaic. 30 October 2018. Archived from the original on 11 April 2020. Retrieved 13 December 2018.
- ↑ "Dextromethorphan" (PDF). Drugs and Chemicals of Concern. Drug Enforcement Administration. August 2010. Archived from the original (PDF) on 2012-10-16.
- ↑ Giannini AJ (1997). Drugs of abuse (2nd ed.). Los Angeles: Practice Management Information Corp. ISBN 1570660530.[page needed]
- ↑ Ackerman, Sarah C.; Hammel, John L.; Brunette, Mary F. (2010-12-20). "Dextromethorphan Abuse and Dependence in Adolescents". Journal of Dual Diagnosis. 6 (3–4): 266–278. doi:10.1080/15504263.2010.537515. S2CID 70666093.
- ↑ Inc, Axsome Therapeutics (2017-02-14). "Axsome Therapeutics Receives FDA Fast Track Designation for AXS-05 for Treatment Resistant Depression". GlobeNewswire News Room. Archived from the original on 2019-12-22. Retrieved 2019-12-22.
- ↑ "Dextromethorphan/bupropion combo is remarkably fast-acting antidepressant". www.mdedge.com. Archived from the original on 2019-12-22. Retrieved 2019-12-22.
- ↑ "Axsome Therapeutics Announces AXS-05 Achieves Primary Endpoint in GEMINI Phase 3 Trial in Major Depressive Disorder". Axsome Therapeutics, Inc. Archived from the original on 2019-12-22. Retrieved 2019-12-22.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- CS1 errors: dates
- CS1 errors: missing periodical
- Wikipedia articles needing page number citations from April 2015
- Articles with invalid date parameter in template
- Webarchive template wayback links
- All articles with incomplete citations
- Articles with incomplete citations from April 2015
- CS1 Bahasa Indonesia-language sources (id)
- All articles with dead external links
- Articles with dead external links from March 2018
- Wikipedia articles needing page number citations from March 2011
- Articles with hatnote templates targeting a nonexistent page
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- All articles with failed verification
- Articles with failed verification from July 2018
- All articles with unsourced statements
- Articles with unsourced statements from March 2011
- Antitussives
- Benzomorphans
- Dissociative drugs
- Enantiopure drugs
- Morphinans
- Mu-opioid agonists
- Nicotinic antagonists
- NMDA receptor antagonists
- Oneirogens
- Phenol ethers
- Prodrugs
- Serotonin-norepinephrine reuptake inhibitors
- Sigma agonists
- RTT