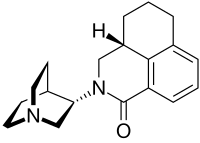
Palonosetron
 | |
| Names | |
|---|---|
| Pronunciation | /pæləˈnoʊsətrɒn/ pal-ə-NOH-sə-tron |
| Trade names | Aloxi |
| |
| Clinical data | |
| Drug class | 5-HT3 antagonist[1] |
| Main uses | Chemotherapy-induced nausea and vomiting (CINV), postoperative nausea and vomiting[2][3] |
| Side effects | QT prolongation, slow heart rate, headache, constipation, weakness, anaphylaxis, serotonin syndrome[3] |
| Pregnancy category |
|
| Routes of use | Intravenous, by mouth |
| Typical dose | 500 ucg PO, 250 ucg IV[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a610002 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 97% (oral) |
| Protein binding | 62% |
| Metabolism | Liver, 50% (mostly CYP2D6-mediated, CYP3A4 and CYP1A2 also involved) |
| Elimination half-life | Approximately 40–50 hours |
| Excretion | Kidney, 80% (of which 49% unchanged); fecal (5 to 8%) |
| Chemical and physical data | |
| Formula | C19H24N2O |
| Molar mass | 296.414 g·mol−1 |
| 3D model (JSmol) | |
| Specific rotation | [α]D −136° [α]D –94.1° (HCl) |
| Melting point | 87 to 88 °C (189 to 190 °F) |
| |
| |
Palonosetron, sold under the brand name Aloxi, is a medication used to prevent chemotherapy-induced nausea and vomiting (CINV) and postoperative nausea and vomiting.[2][3] In delayed CINV there is tentative data suggesting it may be more effective than granisetron.[4] It can be given by mouth or by injection into a vein.[1]
Common side effects include QT prolongation, slow heart rate, headache, constipation, and weakness.[3] Other side effects may include anaphylaxis and serotonin syndrome.[3] Use in pregnancy appears to be safe, but such use has not been well studied.[5] It is a 5-HT3 antagonist.[1]
Palonosetron was approved for medical use in the United States in 2003 and Europe in 2005.[3][2] It is on the World Health Organization's List of Essential Medicines as an alternative to ondansetron.[6] It is available as a generic medication.[1] In the United Kingdom it costs the NHS about £55 per dose as of 2021.[1] This amount in the United States costs about 50 USD.[7]
Medical use
A large clinical trial did not show oral administration to be as effective as intravenous use against delayed CINV.[8]
The combination medication netupitant/palonosetron is approved for both acute and delayed CINV.[9]
Dosage
By mouth it is taken at a dose of 500 ucg an hour before chemotherapy.[1] By injection it is take at a dose of 250 ucg 30 min before chemotherapy.[1]
Side effects
The most common adverse effects are headache, which occurs in 4–11% of patients, and constipation in up to 6% of patients. In less than 1% of patients, other gastrointestinal disorders occur, as well as sleeplessness, first- and second-degree atrioventricular block, muscle pain and shortness of breath. Palonosetron is similarly well tolerated as other 5-HT3 antagonists, and slightly less than placebo.[10][11]
Interactions
Palonosetron does not relevantly inhibit or induce cytochrome P450 liver enzymes. There are case reports about serotonin syndrome when the drug is combined with serotonergic substances such as selective serotonin reuptake inhibitors (SSRIs) and serotonin–norepinephrine reuptake inhibitors (SNRIs), two common types of antidepressants.[10][11]
Pharmacology
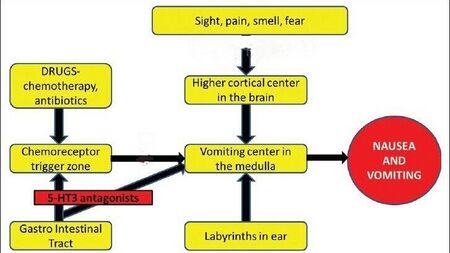
Mechanism of action
Palonosetron is a 5-HT3 antagonist, commonly known as a setron. These drugs act by blocking serotonin from binding to the 5-HT3 receptor.[4] It has a longer duration of action than other 5-HT3 antagonists.
Pharmacokinetics
Orally taken palonosetron is absorbed well from the gut and has a bioavailability of 97%. Highest blood plasma levels are reached after 5.1±1.7 hours, independently of food intake, and plasma protein binding is 62%. 40% of the substance are eliminated in the unchanged form, and a further 45–50% are metabolized by the liver enzyme CYP2D6 and to a lesser extent by CYP3A4 and CYP1A2. The two main metabolites, the N-oxide and a hydroxy derivative, have less than 1% of palonosetron's antagonistic effect and are thus practically inactive.[10][11]

Palonosetron and its metabolites are mainly (to 80–93%) eliminated via the kidney. Biological half-life in healthy persons was 37±12 hours in a study, and 48±19 hours in cancer patients. In 10% of patients, half-life is over 100 hours.[10][11]
Most other marketed setrons have half-lives in the range of about two to 15 hours.
Chemistry
The substance is solid at room temperature and melts at 87 to 88 °C (189 to 190 °F).[13] The infusions and capsules contain palonosetron hydrochloride,[10] which is also a solid. The hydrochloride is easily soluble in water, soluble in propylene glycol, and slightly soluble in ethanol and isopropyl alcohol.[11][14]
The molecule has two asymmetric carbon atoms. It is used in form of the pure (S,S)-stereoisomer.[14]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 456. ISBN 978-0857114105.
- ↑ 2.0 2.1 2.2 "Aloxi". Archived from the original on 14 April 2021. Retrieved 25 October 2021.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "Palonosetron Monograph for Professionals". Drugs.com. Archived from the original on 21 January 2021. Retrieved 25 October 2021.
- ↑ 4.0 4.1 Billio A, Morello E, Clarke MJ (January 2010). Billio A (ed.). "Serotonin receptor antagonists for highly emetogenic chemotherapy in adults". The Cochrane Database of Systematic Reviews (1): CD006272. doi:10.1002/14651858.CD006272.pub2. PMID 20091591. (Retracted, see doi:10.1002/14651858.cd006272.pub3)
- ↑ "Palonosetron (Aloxi) Use During Pregnancy". Drugs.com. Archived from the original on 25 November 2020. Retrieved 25 October 2021.
- ↑ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ↑ "Palonosetron Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 19 January 2021. Retrieved 25 October 2021.
- ↑ Waknine Y (September 4, 2008). "FDA Approvals: Nplate, Aloxi, Vidaza". Medscape. Archived from the original on 2008-12-02. Retrieved 2008-09-04. Freely available with registration.
- ↑ 9.0 9.1 "Akynzeo: Summary of Product Characteristics" (PDF). European Medicines Agency. Archived (PDF) from the original on 26 June 2016. Retrieved 12 July 2016.
- ↑ 10.0 10.1 10.2 10.3 10.4 Haberfeld H, ed. (2015). Austria-Codex (in German). Vienna: Österreichischer Apothekerverlag.
{{cite book}}: CS1 maint: unrecognized language (link) - ↑ 11.0 11.1 11.2 11.3 11.4 Dinnendahl V, Fricke U, eds. (2010). Arzneistoff-Profile (in German). Vol. 7 (23 ed.). Eschborn, Germany: Govi Pharmazeutischer Verlag. ISBN 978-3-7741-9846-3.
{{cite book}}: CS1 maint: unrecognized language (link) - ↑ Shetti, AN; Singh, DR; Nag, K; Shetti, RA; Kumar, VR (January 2014). "Improved prophylaxis of postoperative nausea vomiting: Palonosetron a novel antiemetic". Anesthesia, essays and researches. 8 (1): 9–12. doi:10.4103/0259-1162.128894. PMID 25886096.
- ↑ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (14 ed.). Merck & Co. 2006. p. 1206. ISBN 978-0-911910-00-1.
- ↑ 14.0 14.1 "Chemistry Revire – Aloxi (Palonosetron HCl) Capsules, 0.5 mg" (PDF). Center for Drug Evaluation and Research. 13 August 2008. Archived (PDF) from the original on 5 August 2019. Retrieved 4 October 2021.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Articles citing retracted publications
- CS1 maint: unrecognized language
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with changed CASNo identifier
- Articles with changed KEGG identifier
- Articles with changed EBI identifier
- 5-HT3 antagonists
- Quinuclidines
- Isoquinolines
- Hoffmann-La Roche brands
- Lactams
- World Health Organization essential medicines (alternatives)
- RTT