Squamous cell skin cancer
| Squamous cell skin cancer | |
|---|---|
| Other names: Cutaneous squamous cell carcinoma (cSCC), epidermoid carcinoma, squamous cell epithelioma | |
 | |
| SCC of the skin tends to arise from pre-malignant lesions, actinic keratoses; surface is usually scaly and often ulcerates (as shown here). | |
| Specialty | Dermatology, plastic surgery, otorhinolaryngology |
| Symptoms | Hard lump with a scaly top, ulceration[1] |
| Complications | Distant spread[1] |
| Risk factors | Ultraviolet radiation, actinic keratosis, lighter skin, arsenic exposure, radiation therapy, poor immune system function, HPV infection[1] |
| Diagnostic method | Tissue biopsy[2][3] |
| Differential diagnosis | Keratoacanthoma, actinic keratosis, melanoma, warts, basal cell cancer[4] |
| Prevention | Decreased UV radiation exposure, sunscreen[5][6] |
| Treatment | Surgical removal, radiation therapy, chemotherapy, immunotherapy[2][7] |
| Prognosis | Usually good[5] |
| Frequency | 2.2 million (2015)[8] |
| Deaths | 51,900 (2015)[9] |
Squamous-cell skin cancer (SCC), is a type of skin cancer that typically presents as a hard, rough, scaly topped lesion, which may ulcerate.[1] Onset is often over months.[4] SSC is more likely to spread to distant areas than basal cell cancer.[10] When confined to the outermost layer of the skin, a precancerous or in situ form of SCC is known as Bowen's disease.[1]
The greatest risk factor is high total exposure to ultraviolet radiation from the Sun.[1] Other risks include prior scars, chronic wounds, actinic keratosis, lighter skin, Bowen's disease, arsenic exposure, radiation therapy, poor immune system function, previous basal cell carcinoma, and HPV infection.[2][11] Risk from UV radiation is related to total exposure, rather than early exposure.[12] Tanning beds are becoming another common source of ultraviolet radiation.[12] It begins from squamous cells found within the skin.[13] Diagnosis is often based on skin examination and confirmed by tissue biopsy.[2][3] Other common skin cancers include basal cell cancer, and melanoma.[14]
Decreasing exposure to ultraviolet radiation and the use of sunscreen appear to be effective methods of preventing squamous-cell skin cancer.[5][6] Treatment is typically by surgical removal.[2] This can be by simple excision if the cancer is small otherwise Mohs surgery is generally recommended.[2] Other options may include application of cold and radiation therapy.[7] In the cases in which distant spread has occurred chemotherapy or biologic therapy may be used.[7]
As of 2015, about 2.2 million people have SCC at any given time.[8] It makes up about 20% of all skin cancer cases.[15] About 12% of males and 7% of females in the United States developed SCC at some point in time.[2] While prognosis is usually good, if distant spread occurs five-year survival is ~34%.[4][5] In 2015 it resulted in about 51,900 deaths globally.[9] The usual age at diagnosis is around 66.[4] Following the successful treatment of one case of SCC people are at high risk of developing further cases.[2]
Signs and symptoms

SCC of the skin begins as a small nodule and as it enlarges the center becomes necrotic and sloughs and the nodule turns into an ulcer.
- The lesion caused by SCC is often asymptomatic
- Ulcer or reddish skin plaque that is slow growing
- Intermittent bleeding from the tumor, especially on the lip
- The clinical appearance is highly variable
- Usually the tumor presents as an ulcerated lesion with hard, raised edges
- The tumor may be in the form of a hard plaque or a papule, often with an opalescent quality, with tiny blood vessels
- The tumor can lie below the level of the surrounding skin, and eventually ulcerates and invades the underlying tissue
- The tumor commonly presents on sun-exposed areas (e.g. back of the hand, scalp, lip, and superior surface of pinna)
- On the lip, the tumor forms a small ulcer, which fails to heal and bleeds intermittently
- Evidence of chronic skin photodamage, such as multiple actinic keratoses (solar keratoses)
- The tumor grows relatively slowly
Spread
- Unlike basal-cell carcinoma (BCC), squamous cell carcinoma (SCC) has a higher risk of metastasis.
- Risk of metastasis is higher in SCC arising in scars, on the lower lips or mucosa, and occurring in immunosuppressed patients.
-
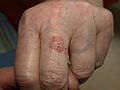
Biopsy-proven squamous cell carcinoma
-
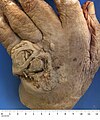
Squamous carcinoma of dorsum of hand
-
SCC in situ (Bowen's disease).
Causes
Squamous cell carcinoma is the second-most common cancer of the skin (after basal-cell carcinoma but more common than melanoma). It usually occurs in areas exposed to the sun. Sunlight exposure and immunosuppression are risk factors for SCC of the skin, with chronic sun exposure being the strongest environmental risk factor.[16] There is a risk of metastasis starting more than 10 years[citation needed] after diagnosable appearance of squamous cell carcinoma, but the risk is low,[specify] though much[specify] higher than with basal-cell carcinoma. Squamous cell cancers of the lip and ears have high rates of local recurrence and distant metastasis.[17] In a recent study, it has also been shown that the deletion or severe down-regulation of a gene titled Tpl2 (tumor progression locus 2) may be involved in the progression of normal keratinocytes into becoming squamous cell carcinoma.[18]
SCCs represent about 20% of the non-melanoma skin cancers, but due to their more obvious nature and growth rates, they represent 90% of all head and neck cancers that are initially presented.[19]
The vast majority of SCCs are those of the skin, and are often the result of ultraviolet exposure. SCCs usually occur on portions of the body commonly exposed to the Sun; the face, ears, neck, hands, or arm. The main symptom is a growing bump that may have a rough, scaly surface and flat reddish patches. Unlike basal-cell carcinomas, SCCs carry a higher risk of metastasis, and may spread to the regional lymph nodes,[20]
Erythroplasia of Queyrat (SCC in situ of the glans or prepuce in males,[21]M[22]: 733 [23]: 656 [24] or the vulvae in females.[25]) may be induced by human papilloma virus.[26] It is reported to occur in the corneoscleral limbus.[27] Erythroplasia of Queyrat may also occur on the anal mucosa or the oral mucosa.[28] Some sources state that this condition is synonymous with Bowen's disease,[25] however generally speaking Bowen's disease refers to carcinoma in situ of any location on the skin such as the lower leg.
Immunosuppression
People who have received solid organ transplants are at a significantly increased risk of developing squamous cell carcinoma due to the use of chronic immunosuppressive medication. While the risk of developing all skin cancers increases with these medications, this effect is particularly severe for SCC, with hazard ratios as high as 250 being reported, versus 40 for basal cell carcinoma.[29] The incidence of SCC development increases with time posttransplant.[30] Heart and lung transplant recipients are at the highest risk of developing SCC due to more intensive immunosuppressive medications used.
Squamous cell cancers of the skin in individuals on immunotherapy or suffering from lymphoproliferative disorders (i.e. leukemia) tend to be much more aggressive, regardless of their location.[31] The risk of SCC, and non-melanoma skin cancers generally, varies with the immunosuppressive drug regimen chosen. The risk is greatest with calcineurin inhibitors like cyclosporine and tacrolimus, and least with mTOR inhibitors, such as sirolimus and everolimus. The antimetabolites azathioprine and mycophenolic acid have an intermediate risk profile.[32]
Diagnosis
Diagnosis is confirmed via biopsy of the tissue(s) suspected to be affected by SCC. For the skin, look under skin biopsy.
The pathological appearance of a squamous cell cancer varies with the depth of the biopsy. For that reason, a biopsy including the subcutaneous tissue and basalar epithelium, to the surface is necessary for correct diagnosis. The performance of a shave biopsy (see skin biopsy) might not acquire enough information for a diagnosis. An inadequate biopsy might be read as actinic keratosis with follicular involvement. A deeper biopsy down to the dermis or subcutaneous tissue might reveal the true cancer. An excision biopsy is ideal, but not practical in most cases. An incisional or punch biopsy is preferred. A shave biopsy is least ideal, especially if only the superficial portion is acquired.[citation needed]
Characteristics
Histopathologically, the epidermis in SCC in situ (Bowen’s disease) will show hyperkeratosis and parakeratosis. There will also be marked acanthosis with elongation and thickening of the rete ridges. These changes will overly keratinocytic cells which are often highly atypical and may in fact have a more unusual appearance than invasive SCC. The atypia spans the full thickness of the epidermis, with the keratinocytes demonstrating intense mitotic activity, pleomorphism, and greatly enlarged nuclei. They will also show a loss of maturity and polarity, giving the epidermis a disordered or “windblown” appearance.
Two types of multinucleated cells may be seen: the first will present as a multinucleated giant cell, and the second will appear as a dyskeratotic cell engulfed in the cytoplasm of a keratinocyte. Occasionally, cells of the upper epidermis will undergo vacuolization, demonstrating an abundant and strongly eosinophilic cytoplasm. There may be a mild to moderate lymphohistiocytic infiltrate detected in the upper dermis.[33]
-
Histopathology of squamous cell carcinoma in situ (black arrow), compared to normal skin, showing marked atypia.
-
Squamous cell carcinoma in situ, showing prominent dyskeratosis and aberrant mitoses at all levels of the epidermis, along with marked parakeratosis.[33]
In situ disease
Bowen's disease is essentially equivalent to and used interchangeably with SCC in situ, when not having invaded through the basement membrane.[33] Depending on source, it is classified as precancerous[34] or SCC in situ (technically cancerous but non-invasive).[35][36] In SCC in situ (Bowen's disease), atypical squamous cells proliferate through the whole thickness of the epidermis.[33] The entire tumor is confined to the epidermis and does not invade into the dermis.[33] The cells are often highly atypical under the microscope, and may in fact look more unusual than the cells of some invasive squamous cell carcinomas.[33]
-
SCC in situ, high magnification, demonstrating an intact basement membrane.[33]
-
SCC in situ
-
SCC in situ
-
SCC in situ
-
SCC in situ
Erythroplasia of Queyrat is a particular type of Bowen's disease that can arise on the glans or prepuce in males,[21][22]: 733 [23]: 656 [24] and the vulvae in females.[25] It mainly occurs in uncircumcised males,[25][37] over the age of 40.[28] It is named for French dermatologist Louis Queyrat (1856–1933),[38][39][40] who was head of the dermatology service of l'Hôpital Ricord, a venereal hospital in Paris, now Hôpital Cochin.[41]
Invasive disease
In invasive SCC, tumor cells infiltrate through the basement membrane. The infiltrate can be somewhat difficult to detect in the early stages of invasion: however, additional indicators such as full thickness epidermal atypia and the involvement of hair follicles can be used to facilitate the diagnosis. Later stages of invasion are characterized by the formation of nests of atypical tumor cells in the dermis, often with a corresponding inflammatory infiltrate.[33]
-
Superficially invasive squamous cell carcinoma (SCCSI). These lesions often do not show the marked pleomorphism and atypical nuclei of SCC in situ, but demonstrate early keratinocyte invasion of the dermis.[33]
-
High magnification demonstrates the pleomorphism of the invading keratinocytes.[33]
-
Invasive nests with characteristic large celled centers. Ulceration (at left) is common in invasive SCC.
Degree of differentiation
-
Well-differentiated (and yet invasive) SCC, showing prominent keratinization and may form “pearllike” structures where dermal nests of keratinocytes attempt to mature in a layered fashion. Well-differentiated SCC has slightly enlarged, hyperchromatic nuclei with abundant amounts of cytoplasm. Intercellular bridges will frequently be visible.[33]
-
Moderately differentiated lesions of invasive SCC show much less organization and maturation with significantly less keratin formation.[33]
-
Poorly differentiated, where attempts at keratinization are often no longer evident. This is a clear-cell squamous cell carcinoma. The dysplastic cells here infiltrate in cords through the dermis. Poorly differentiated SCC has greatly enlarged, pleomorphic nuclei demonstrating a high degree of atypia and frequent mitoses.[33]
-
Poorly differentiated clear-cell squamous cell carcinoma. For this type of SCC, immunostains will likely be required to classify it unless other areas of the tumor show obvious squamous cell features such as seen here (arrow).
Prevention
Appropriate sun-protective clothing, use of broad-spectrum (UVA/UVB) sunscreen with at least SPF 50, and avoidance of intense sun exposure may prevent skin cancer.[42] A 2016 review of sunscreen for preventing squamous cell skin cancer found insufficient evidence to demonstrate whether it was effective.[43]
Management
Most squamous cell carcinomas are removed with surgery. A few selected cases are treated with topical medication. Surgical excision with a free margin of healthy tissue is a frequent treatment modality. Radiotherapy, given as external beam radiotherapy or as brachytherapy (internal radiotherapy), can also be used to treat squamous cell carcinomas. There is little evidence comparing the effectiveness of different treatments for non-metastatic SCC of the skin.[44]
Mohs surgery is frequently utilized; considered the treatment of choice for squamous cell carcinoma of the skin, physicians have also utilized the method for the treatment of squamous cell carcinoma of the mouth, throat, and neck.[45] An equivalent method of the CCPDMA standards can be utilized by a pathologist in the absence of a Mohs-trained physician. Radiation therapy is often used afterward in high risk cancer or patient types.[citation needed]
Electrodessication and curettage or EDC can be done on selected squamous cell carcinoma of the skin. In areas where SCC's are known to be non-aggressive, and where the patient is not immunosuppressed, EDC[clarification needed] can be performed with good to adequate cure rate.
Treatment options for SCC in situ (Bowen's disease) include photodynamic therapy with 5-aminolevulinic acid, cryotherapy, topical 5-fluorouracil or imiquimod, and excision. A meta-analysis showed evidence that PDT is more effective than cryotherapy and has better cosmetic outcomes. There is generally a lack of evidence comparing the effectiveness of all treatment options.[46]
High-risk squamous cell carcinoma, as defined by those occurring around the eye, ear, or nose, is of large size, is poorly differentiated, and grows rapidly, requires more aggressive, multidisciplinary management.
Nodal spread:
- Surgical block dissection if palpable nodes or in cases of Marjolin's ulcers but the benefit of prophylactic block lymph node dissection with Marjolin's ulcers is not proven.
- Radiotherapy
- Adjuvant therapy may be considered in those with high-risk SCC even in the absence of evidence for local mestastasis. Imiquimod (Aldara) has been used with success for squamous cell carcinoma in situ of the skin and the penis, but the morbidity and discomfort of the treatment is severe. An advantage is the cosmetic result: after treatment, the skin resembles normal skin without the usual scarring and morbidity associated with standard excision. Imiquimod is not FDA-approved for any squamous cell carcinoma.
In general, squamous cell carcinomas have a high risk of local recurrence, and up to 50% do recur.[47] Frequent skin exams with a dermatologist is recommended after treatment.
Prognosis
The long-term outcome of squamous cell carcinomas is dependent upon several factors: the sub-type of the carcinoma, available treatments, location(s) and severity, and various patient health-related variables (accompanying diseases, age, etc.). Generally, the long-term outcome is positive, as less than 4% of Squamous cell carcinoma cases are at risk of metastasis.[48][49] Some particular forms of squamous cell carcinomas have a higher mortality rate.
One study found squamous cell carcinoma of the penis had a much greater rate of mortality than some other forms of squamous cell carcinoma, that is, about 23%,[50] although this relatively high mortality rate may be associated with possibly latent diagnosis of the disease due to patients avoiding genital exams until the symptoms are debilitating, or refusal to submit to a possibly scarring operation upon the genitalia. Squamous cell carcinoma occurring in the organ transplant population is also associated with a higher risk of mortality.[51]
Epidemiology

The incidence of squamous cell carcinoma continues to rise around the world. A recent study estimated that there are between 180,000 and 400,000 cases of SCC in the United States in 2013.[53] Risk factors for squamous cell carcinoma varies with age, gender, race, geography, and genetics. The incidence of SCC increases with age and the peak incidence is usually around 60 years old. Males are affected with SCC at a ratio of 2:1 in comparison to females. Caucasians are more likely to be affected, especially those with fair skin or those chronically exposed to UV radiation.
Squamous cell carcinoma of the skin can be found on all areas of the body but is most common on frequently sun-exposed areas, such as the face, legs and arms.[54] Solid organ transplant recipients (heart, lung, liver, pancreas, among others) are also at a heightened risk of developing aggressive, high-risk SCC. There are also a few rare congenital diseases predisposed to cutaneous malignancy. In certain geographic locations, exposure to arsenic in well water or from industrial sources may significantly increase the risk of SCC.[16]
See also
- List of skin tumors
- List of cutaneous conditions associated with increased risk of nonmelanoma skin cancer
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 DE, Elder; D, Massi; RA, Scolyer; R, Willemze (2018). "Squamous cell carcinoma". WHO Classification of Skin Tumours. Vol. 11 (4th ed.). Lyon (France): World Health Organization. pp. 35–45. ISBN 978-92-832-2440-2. Archived from the original on 2022-07-11. Retrieved 2022-08-08.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Gandhi, SA; Kampp, J (November 2015). "Skin Cancer Epidemiology, Detection, and Management". The Medical Clinics of North America. 99 (6): 1323–35. doi:10.1016/j.mcna.2015.06.002. PMID 26476255.
- ↑ 3.0 3.1 "Skin Cancer Treatment". National Cancer Institute. 21 June 2017. Archived from the original on 4 July 2017. Retrieved 2 July 2017.
- ↑ 4.0 4.1 4.2 4.3 Ferri, Fred F. (2016). Ferri's Clinical Advisor 2017 E-Book: 5 Books in 1. Elsevier Health Sciences. p. 1199. ISBN 9780323448383. Archived from the original on 29 August 2017. Retrieved 2 July 2017.
- ↑ 5.0 5.1 5.2 5.3 World Cancer Report 2014. World Health Organization. 2014. pp. Chapter 5.14. ISBN 978-9283204299.
- ↑ 6.0 6.1 Jou, PC; Feldman, RJ; Tomecki, KJ (June 2012). "UV protection and sunscreens: what to tell patients". Cleveland Clinic Journal of Medicine. 79 (6): 427–36. doi:10.3949/ccjm.79a.11110. PMID 22660875.
- ↑ 7.0 7.1 7.2 "Skin Cancer Treatment". National Cancer Institute. 21 June 2017. Archived from the original on 4 July 2017. Retrieved 2 July 2017.
- ↑ 8.0 8.1 GBD 2015 Disease and Injury Incidence and Prevalence, Collaborators. (8 October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- ↑ 9.0 9.1 GBD 2015 Mortality and Causes of Death, Collaborators. (8 October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/s0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
- ↑ Cakir, BÖ; Adamson, P; Cingi, C (November 2012). "Epidemiology and economic burden of nonmelanoma skin cancer". Facial Plastic Surgery Clinics of North America. 20 (4): 419–22. doi:10.1016/j.fsc.2012.07.004. PMID 23084294.
- ↑ Opel, Sophia (2016). Skin Cancer for the Plastic Surgeon. Textbook of Plastic and Reconstructive Surgery (1 ed.). UCL Press. pp. 61–76. ISBN 9781910634394. JSTOR j.ctt1g69xq0.9.
- ↑ 12.0 12.1 Gallagher, RP; Lee, TK; Bajdik, CD; Borugian, M (2010). "Ultraviolet radiation". Chronic Diseases in Canada. 29 Suppl 1: 51–68. PMID 21199599.
- ↑ "NCI Dictionary of Cancer Terms". National Cancer Institute. 2011-02-02. Archived from the original on 9 November 2016. Retrieved 9 November 2016.
- ↑ "Skin Cancer Treatment (PDQ®)". NCI. 2013-10-25. Archived from the original on 5 July 2014. Retrieved 30 June 2014.
- ↑ Stratigos, A; Garbe, C; Lebbe, C; Malvehy, J; del Marmol, V; Pehamberger, H; Peris, K; Becker, JC; Zalaudek, I; Saiag, P; Middleton, MR; Bastholt, L; Testori, A; Grob, JJ; European Dermatology Forum, (EDF).; European Association of Dermato-Oncology, (EADO).; European Organization for Research and Treatment of Cancer, (EORTC). (September 2015). "Diagnosis and treatment of invasive squamous cell carcinoma of the skin: European consensus-based interdisciplinary guideline". European Journal of Cancer. 51 (14): 1989–2007. doi:10.1016/j.ejca.2015.06.110. PMID 26219687.
- ↑ 16.0 16.1 "MD Consult - Important Notice". Archived from the original on 3 March 2016. Retrieved 15 March 2017.
- ↑ "Squamous Cell Carcinoma". American Academy of Dermatology. 25 December 2010. Archived from the original on 25 December 2010. Retrieved 15 March 2017.
- ↑ Decicco-Skinner, KL; Trovato, EL; Simmons, JK; Lepage, PK; Wiest, JS (2011). "Loss of tumor progression locus 2 (tpl2) enhances tumorigenesis and inflammation in two-stage skin carcinogenesis". Oncogene. 30 (4): 389–97. doi:10.1038/onc.2010.447. PMC 3460638. PMID 20935675.
- ↑ "Cutaneous Squamous Cell Carcinoma: Practice Essentials, Background, Pathophysiology". 28 June 2016. Archived from the original on 6 April 2017. Retrieved 15 March 2017.
- ↑ "Cutaneous Squamous Cell Carcinoma: Practice Essentials, Background, Pathophysiology". 28 June 2016. Archived from the original on 6 April 2017. Retrieved 15 March 2017.
- ↑ 21.0 21.1 Marks, James G; Miller, Jeffery (2006). Lookingbill and Marks' Principles of Dermatology (4th ed.). Elsevier Inc. Page 63. ISBN 1-4160-3185-5.
- ↑ 22.0 22.1 Freedberg, et al. (2003). Fitzpatrick's Dermatology in General Medicine. (6th ed.). McGraw-Hill. ISBN 0-07-138076-0.
- ↑ 23.0 23.1 James, William D.; Berger, Timothy G.; et al. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. ISBN 0-7216-2921-0.
- ↑ 24.0 24.1 Rapini, Ronald P.; Bolognia, Jean L.; Jorizzo, Joseph L. (2007). Dermatology: 2-Volume Set. St. Louis: Mosby. p. 1050. ISBN 978-1-4160-2999-1.
- ↑ 25.0 25.1 25.2 25.3 Zenilman JM, Shahmanesh M, eds. (2012). Sexually transmitted infections diagnosis, management, and treatment. Sudbury, Mass.: Jones & Bartlett Learning. p. 109. ISBN 9781449619848.
- ↑ J Invest Dermatol 2000 Sep;115(3):396–401
- ↑ Syed Imtiaz Ali Shah, Saeed Ahmed Sangi, Seraj Ahmed Abbasi: Bowen’s Disease: Pak J Ophthalmol 1998 Vol. 14, No. 1, pp. 37–38.
- ↑ 28.0 28.1 Katsambas AD, Lotti TM, eds. (2003). European handbook of dermatological treatments (2nd ed.). Berlin [u.a.]: Springer. p. 169. ISBN 9783540008781.
- ↑ Tessari, G.; Girolomoni, G. (2012). "Nonmelanoma skin cancer in solid organ transplant recipients: update on epidemiology, risk factors, and management". Dermatol. Surg. 38 (10): 1622–30. doi:10.1111/j.1524-4725.2012.02520.x. PMID 22805312.
- ↑ Zwald, FO (2011). "Skin cancer in solid organ transplant recipients: advances in therapy and management: part I. Epidemiology of skin cancer in solid organ transplant recipients". Journal of the American Academy of Dermatology. 65 (2): 253–61, quiz 262. doi:10.1016/j.jaad.2010.11.062. PMID 21763561.
- ↑ "Squamous Cell Carcinoma: What it Looks Like". Archived from the original on 2008-12-21.
- ↑ Kuschal, C.; Thoms, K. M.; Schubert, S.; Schäfer, A.; Boeckmann, L.; Schön, M. P.; Emmert, Steffen (2012). "Skin cancer in organ transplant recipients: effects of immunosuppressive medications on DNA repair". Exp. Dermatol. 21 (1): 2–6. doi:10.1111/j.1600-0625.2011.01413.x. PMID 22151386.
{{cite journal}}: Unknown parameter|displayauthors=ignored (help) - ↑ 33.00 33.01 33.02 33.03 33.04 33.05 33.06 33.07 33.08 33.09 33.10 33.11 33.12 Yanofsky, Valerie R.; Mercer, Stephen E.; Phelps, Robert G. (2011). "Histopathological Variants of Cutaneous Squamous Cell Carcinoma: A Review". Journal of Skin Cancer. 2011: 1–13. doi:10.1155/2011/210813. ISSN 2090-2905. PMC 3018652. PMID 21234325.
 This article incorporates text available under the CC BY-SA 3.0 license.
This article incorporates text available under the CC BY-SA 3.0 license.
- ↑ Bath-Hextall FJ, Matin RN, Wilkinson D, Leonardi-Bee J (June 2013). "Interventions for cutaneous Bowen's disease". Cochrane Database Syst Rev (6): CD007281. doi:10.1002/14651858.CD007281.pub2. PMC 6464151. PMID 23794286.
- ↑ Janine Khalyl-Mawad. "Pathology of Cutaneous Squamous Cell Carcinoma and Bowen Disease". Medscape. Archived from the original on 2019-10-28. Retrieved 2019-10-28. Updated: Jun 11, 2019
- ↑ "Bowen's disease". National Health Service. Archived from the original on 2019-10-28. Retrieved 2019-10-28. Page last reviewed: 21 May 2019
- ↑ Berkow, Robert (1997). The Merck manual of medical information. Whitehouse Station, N.J: Merck Research Laboratories. p. 1057. ISBN 0911910875.
- ↑ synd/3373 at Who Named It?
- ↑ A. Queyrat. Érythroplasie du gland. Bulletin de la Société française de dermatologie et de syphiligraphie, Paris, 1911, 22: 378–382.
- ↑ Steffen C (2007). "Squamous cell carcinoma in situ: a historical note". Skinmed. 6 (1): 7–10. doi:10.1111/j.1540-9740.2007.06033.x. PMID 17215613.
- ↑ Fonds d'archives, Lieu de conservation Archived 2013-10-23 at the Wayback Machine : Institut Pasteur, Service des Archives.
- ↑ Gallagher, RP; Lee, TK; Bajdik, CD; Borugian, M (2010). "Ultraviolet radiation". Chronic Diseases in Canada. 29 (Suppl 1): 51–68. PMID 21199599.
- ↑ Sánchez, Guillermo; Nova, John; Rodriguez-Hernandez, Andrea Esperanza; Medina, Roger David; Solorzano-Restrepo, Carolina; Gonzalez, Jenny; Olmos, Miguel; Godfrey, Kathie; Arevalo-Rodriguez, Ingrid (25 July 2016). "Sun protection for preventing basal cell and squamous cell skin cancers". Cochrane Database of Systematic Reviews. 7: CD011161. doi:10.1002/14651858.CD011161.pub2. PMC 6457780. PMID 27455163.
- ↑ Lansbury, Louise; Leonardi-Bee, Jo; Perkins, William; Goodacre, Timothy; Tweed, John A; Bath-Hextall, Fiona J (2010-04-14). "Interventions for non-metastatic squamous cell carcinoma of the skin". Cochrane Database of Systematic Reviews (4): CD007869. doi:10.1002/14651858.CD007869.pub2. ISSN 1465-1858. PMID 20393962. Archived from the original on 2018-06-21. Retrieved 2018-06-21.
- ↑ Gross, K.G.; et al. (1999). Mohs Surgery, Fundamentals and Techniques. Mosby.
- ↑ Bath-Hextall, Fiona J; Matin, Rubeta N; Wilkinson, David; Leonardi-Bee, Jo (2013-06-24). "Interventions for cutaneous Bowen's disease". Cochrane Database of Systematic Reviews (6): CD007281. doi:10.1002/14651858.CD007281.pub2. ISSN 1465-1858. PMC 6464151. PMID 23794286. Archived from the original on 2019-10-25. Retrieved 2019-10-25.
- ↑ Jennings, C (2010). "Management of High-Risk Cutaneous Squamous Cell Carcinoma". Journal of Clinical and Aesthetic Dermatology. 61 (3): 282–5. doi:10.1016/0022-3913(89)90128-5. PMID 2921745.
- ↑ Chollet A, Hohl D, Perrier P (April 2012). "[Risk of cutaneous squamous cell carcinomas: the role of clinical and pathological reports]". Rev Med Suisse. 8 (335): 743–6. PMID 22545495.
- ↑ Brantsch Kay D; Meisner Christoph; Schönfisch Birgitt; Trilling Birgit; Wehner-Caroli Jörg; Röcken Martin; Breuninger Helmut (2008). "Analysis of risk factors determining prognosis of cutaneous squamous-cell carcinoma: A prospective study". The Lancet Oncology. 9 (8): 713–720. doi:10.1016/S1470-2045(08)70178-5. PMID 18617440.
- ↑ Bethune, G; Campbell, J; Rocker, A; Bell, D; Rendon, R; Merrimen, J (2012). "Clinical and pathologic factors of prognostic significance in penile squamous cell carcinoma in a North American population". Urology. 79 (5): 1092–7. doi:10.1016/j.urology.2011.12.048. PMID 22386252.
- ↑ Chapman, JR (2013). "Cancer in the Transplant Recipient". PerspectivesMedicine.
- ↑ "WHO Disease and injury country estimates". World Health Organization. 2009. Archived from the original on 2009-11-11. Retrieved Nov 11, 2009.
- ↑ Kari, PS (2012). "Cutaneous squamous cell carcinoma: estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012". Journal of the American Academy of Dermatology. 68 (6): 957–66. doi:10.1016/j.jaad.2012.11.037. PMID 23375456.
- ↑ "Squamous Cell Carcinoma". Skin Cancer.Org. 2018. Archived from the original on December 11, 2018. Retrieved Dec 12, 2018.
External links
| Classification | |
|---|---|
| External resources |
- Information on Squamous Cell Carcinoma from The Skin Cancer Foundation Archived 2011-11-26 at the Wayback Machine
- DermNet NZ: Squamous cell carcinoma Archived 2010-12-07 at the Wayback Machine
- Pages with script errors
- CS1: long volume value
- CS1 errors: unsupported parameter
- Articles with imported Creative Commons Attribution-ShareAlike 3.0 text
- Webarchive template wayback links
- All articles with unsourced statements
- Articles with unsourced statements from July 2012
- Articles with invalid date parameter in template
- Articles needing more detailed references
- Articles with unsourced statements from July 2017
- Articles with unsourced statements from September 2016
- Wikipedia articles needing clarification from September 2016
- Anatomical pathology
- Gastrointestinal cancer
- Gynaecological cancer
- Lung cancer
- Integumentary neoplasia
- Epidermal nevi, neoplasms, and cysts
- Carcinoma
- Infectious causes of cancer
- RTT
- Papillomavirus-associated diseases


![Squamous cell carcinoma in situ, showing prominent dyskeratosis and aberrant mitoses at all levels of the epidermis, along with marked parakeratosis.[33]](https://upload.wikimedia.org/wikipedia/commons/thumb/b/ba/Micrograph_of_squamous_cell_carcinoma_in_situ_-_100x.jpg/367px-Micrograph_of_squamous_cell_carcinoma_in_situ_-_100x.jpg)
![SCC in situ, high magnification, demonstrating an intact basement membrane.[33]](https://upload.wikimedia.org/wikipedia/commons/thumb/0/01/Micrograph_of_squamous_cell_carcinoma_in_situ_-_400x.jpg/299px-Micrograph_of_squamous_cell_carcinoma_in_situ_-_400x.jpg)




![Superficially invasive squamous cell carcinoma (SCCSI). These lesions often do not show the marked pleomorphism and atypical nuclei of SCC in situ, but demonstrate early keratinocyte invasion of the dermis.[33]](https://upload.wikimedia.org/wikipedia/commons/thumb/f/f8/Micrograph_of_invasive_squamous_cell_carcinoma_-_150x.jpg/305px-Micrograph_of_invasive_squamous_cell_carcinoma_-_150x.jpg)
![High magnification demonstrates the pleomorphism of the invading keratinocytes.[33]](https://upload.wikimedia.org/wikipedia/commons/thumb/2/26/Micrograph_of_invasive_squamous_cell_carcinoma_-_200x.jpg/340px-Micrograph_of_invasive_squamous_cell_carcinoma_-_200x.jpg)

![Well-differentiated (and yet invasive) SCC, showing prominent keratinization and may form “pearllike” structures where dermal nests of keratinocytes attempt to mature in a layered fashion. Well-differentiated SCC has slightly enlarged, hyperchromatic nuclei with abundant amounts of cytoplasm. Intercellular bridges will frequently be visible.[33]](https://upload.wikimedia.org/wikipedia/commons/thumb/3/3c/Micrograph_of_well-differentiated_and_invasive_squamous-cell_carcinoma.jpg/306px-Micrograph_of_well-differentiated_and_invasive_squamous-cell_carcinoma.jpg)
![Moderately differentiated lesions of invasive SCC show much less organization and maturation with significantly less keratin formation.[33]](https://upload.wikimedia.org/wikipedia/commons/thumb/f/f2/Micrograph_of_moderately_differentiated_and_invasive_squamous-cell_carcinoma.jpg/310px-Micrograph_of_moderately_differentiated_and_invasive_squamous-cell_carcinoma.jpg)
![Poorly differentiated, where attempts at keratinization are often no longer evident. This is a clear-cell squamous cell carcinoma. The dysplastic cells here infiltrate in cords through the dermis. Poorly differentiated SCC has greatly enlarged, pleomorphic nuclei demonstrating a high degree of atypia and frequent mitoses.[33]](https://upload.wikimedia.org/wikipedia/commons/thumb/6/61/Micrograph_of_clear-cell_squamous-cell_carcinoma.jpg/296px-Micrograph_of_clear-cell_squamous-cell_carcinoma.jpg)
