Calcium channel blocker
| Calcium channel blocker | |
|---|---|
| Drug class | |
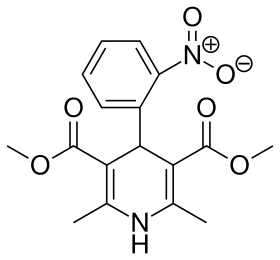 Skeletal formula of nifedipine, the first CCB | |
| Names | |
| Stem | -dipine, -fradil, -tiazem, -pamil[2][3] |
| Other names | Calcium channel antagonists, calcium antagonists, calcium entry blockers[1] |
| Clinical data | |
| Uses | High blood pressure, stable angina, atrial fibrillation[4] |
| Side effects | Headaches, swelling, slow heart rate, constipation, heart failure[4] |
| Interactions | Certain agents with beta blockers[4] |
| Common types | Dihydropyridines: amlodipine, nifedipine[4] Non-dihydropyridines: diltiazem, verapamil[4][5] |
| Mechanism of action | Block the inward movement of calcium[6] |
| External links | |
| Drugs.com | Drug Classes |
Calcium channel blockers (CCB) are a class of medication made up of two main groups dihydropyridines and non-dihydropyridines.[5] Common dihydropyridines include amlodipine and nifedipine and non-dihydropyridines include diltiazem and verapamil.[4] Both types are used for stable angina, while dihydropyridines are used for high blood pressure, and non-dihydropyridines are used for atrial fibrillation.[4] Other uses include cluster headache, pulmonary hypertension, and subarachnoid hemorrhage.[7][6] They can be take by mouth or given by injection into a vein.[5]
Common side effects of dihydropyridines include headaches and swelling while non-dihydropyridines can result in a slow heart rate, constipation, and heart failure.[4] They work by blocking movement of calcium (Ca2+
) into cell.[4] Dihydropyridines mainly affect blood vessels resulting in vasodilation while non-dihydropyridines mainly affect the heart resulting in decreased contraction.[5] Overdose is associated with a high risk of death.[6]
Calcium channel blockers were discovered in 1964, with the first useful agent being verapamil.[8][9] They are commonly used.[6] A number of agents are available as generic medication and not very expensive.[4]
Types
There are three clinically distinct classes; phenylalkylamines, dihydropyridines and benzothiazepines.[10][11] They can also be classed in two main groups dihydropyridines and non-dihydropyridines.[5]
Dihydropyridine
Dihydropyridine (DHP) calcium channel blockers are derived from the molecule dihydropyridine and often used to reduce systemic vascular resistance and arterial pressure. Sometimes when they are used to treat angina, the vasodilation and hypotension can lead to reflex tachycardia, which can be detrimental for patients with ischemic symptoms because of the resulting increase in myocardial oxygen demand. Dihydropyridine calcium channel blockers can worsen proteinuria in patients with nephropathy.[12]
This CCB class is easily identified by the suffix "-dipine".
- Amlodipine (Norvasc)[13]
- Aranidipine (Sapresta)[13]
- Azelnidipine (Calblock)[13]
- Barnidipine (HypoCa)[13]
- Benidipine (Coniel)[13]
- Cilnidipine (Atelec, Cinalong, Siscard)[13]
- Clevidipine (Cleviprex)[13]
- Efonidipine (Landel)[13]
- Felodipine (Plendil)[13]
- Isradipine (DynaCirc, Prescal)[13]
- Lacidipine (Motens, Lacipil)[13]
- Lercanidipine (Zanidip)[13]
- Manidipine (Calslot, Madipine)[13]
- Nicardipine (Cardene, Carden SR)[13]
- Nifedipine (Procardia, Adalat)[13]
- Nilvadipine (Nivadil)[13]
- Nimodipine (Nimotop)[13]
- Nisoldipine (Baymycard, Sular, Syscor)[13]
- Nitrendipine (Cardif, Nitrepin, Baylotensin)
- Pranidipine (Acalas)[13]
Non-dihydropyridine
Phenylalkylamine

Phenylalkylamine calcium channel blockers are relatively selective for myocardium, reduce myocardial oxygen demand and reverse coronary vasospasm, and are often used to treat angina. They have minimal vasodilatory effects compared with dihydropyridines and therefore cause less reflex tachycardia, making it appealing for treatment of angina, where tachycardia can be the most significant contributor to the heart's need for oxygen. Therefore, as vasodilation is minimal with the phenylalkylamines, the major mechanism of action is causing negative inotropy. Phenylalkylamines are thought to access calcium channels from the intracellular side, although the evidence is somewhat mixed.[14]
- Fendiline
- Gallopamil
- Verapamil (Calan, Isoptin)
Benzothiazepine
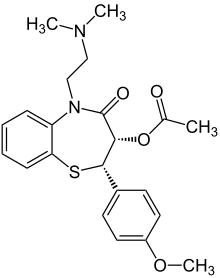
Benzothiazepine calcium channel blockers belong to the benzothiazepine class of compounds and are an intermediate class between phenylalkylamine and dihydropyridines in their selectivity for vascular calcium channels. By having both cardiac depressant and vasodilator actions, benzothiazepines are able to reduce arterial pressure without producing the same degree of reflex cardiac stimulation caused by dihydropyridines.
- Diltiazem (Cardizem) (also used to prevent migraine)
Nonselective
While most of the agents listed above are relatively selective, there are additional agents that are considered nonselective. These include mibefradil, bepridil, flunarizine (BBB crossing), fluspirilene (BBB crossing),[15] and fendiline.[16]
Others
Gabapentinoids, such as gabapentin and pregabalin, are selective blockers of α2δ subunit-containing voltage-gated calcium channels. They are used primarily to treat epilepsy and neuropathic pain.
Ziconotide, a peptide compound derived from the omega-conotoxin, is a selective N-type calcium channel blocker that has potent analgesic properties that are equivalent to approximate 1,000 times that of morphine. It must be delivered via the intrathecal (directly into the cerebrospinal fluid) route via an intrathecal infusion pump.
Medical uses
Depending on the type of calcium channel blocker, they are used for high blood pressure, irregular heart rhythms, or angina.[10]
CCBs have been shown to be slightly more effective than beta blockers at lowering cardiovascular mortality, but they are associated with more side effects.[17][18] Risks; however, were mainly found to be associated with short-acting CCBs.[19]
Side effects
Side effects of calcium channel blockers are mostly an exaggeration of their therapeutic actions.[10] The commonest side effects include abdominal pain and constipation resulting from effects on calcium channels in smooth muscle of gastrointestinal tract, and flushing and headache resulting from vasodilation. Dizziness, palpitations, rash and swelling of ankles can also occur.[20][10] Angioedema, erectile dysfunction, gum swelling, depression and muscle pains can occur but are uncommon.[20][10]
Swelling
Swelling of the feet, ankles and legs (peripheral edema), is a common side effect with CCB use, particularly dihydropyridines, and more likely in women, older people, if standing for long, in warm environments and if there is heart failure.[10][21] It is more likely if used for long periods of time and results from dilated arterioles and leakiness of very small veins.[10][22] Since lymphatic drainage relies on contraction of the smooth muscle inside the lymphatic vessel supported by voltage-gated calcium channels, inhibition of voltage-gated calcium channel poses a threat towards lymphatic removal of interstitial fluid essential for normal lymphatic system functioning.[21][23]
Toxicity

Mild CCB toxicity is treated with supportive care. Nondihydropyridine CCBs may produce profound toxicity and early decontamination, especially for slow-release agents, is essential. For severe overdoses, treatment usually includes close monitoring of vital signs and the addition of vasopressive agents and intravenous fluids for blood pressure support. Intravenous calcium gluconate (or calcium chloride if a central line is available) and atropine are first-line therapies. If the time of the overdose is known and presentation is within two hours of ingestion, activated charcoal, gastric lavage, and polyethylene glycol may be used to decontaminate the gut. Efforts for gut decontamination may be extended to within 8 hours of ingestion with extended-release preparations.
Hyperinsulinemia-euglycemia therapy has emerged as a viable form of treatment.[24] Although the mechanism is unclear, increased insulin may mobilize glucose from peripheral tissues to serve as an alternative fuel source for the heart (the heart mainly relies on oxidation of fatty acids). Theoretical treatment with lipid emulsion therapy has been considered in severe cases, but is not yet standard of care.
Caution should be taken when using verapamil with a beta blocker due to the risk of severe bradycardia. If unsuccessful, ventricular pacing should be used.[25]
Mechanism of action

The term calcium channel blocker is a more accurate term than calcium antagonist, as they define drugs that block cellular entry of calcium through calcium channels rather than prevent its actions within cells.[10][20]
In the body's tissues, the concentration of calcium ions (Ca2+
) outside cells is normally about 10000-fold higher than the concentration inside cells. Embedded in the membrane of some cells are calcium channels. When these cells receive a certain signal, the channels open, letting calcium rush into the cell. The resulting increase in intracellular calcium has different effects in different types of cells. Calcium channel blockers prevent or reduce the opening of these channels and thereby reduce these effects.
Several types of calcium channels occur, with a number of classes of blockers, but almost all of them preferentially or exclusively block the L-type voltage-gated calcium channel.[26]
Voltage-dependent calcium channels are responsible for excitation-contraction coupling of skeletal, smooth, and cardiac muscle and for regulating aldosterone and cortisol secretion in endocrine cells of the adrenal cortex.[27] In the heart, they are also involved in the conduction of the pacemaker signals. CCBs used as medications primarily have four effects:
- By acting on vascular smooth muscle, they reduce contraction of the arteries and cause an increase in arterial diameter, a phenomenon called vasodilation (CCBs do not work on venous smooth muscle).
- By acting on cardiac muscles (myocardium), they reduce the force of contraction of the heart.
- By slowing down the conduction of electrical activity within the heart, they slow down the heart beat.
- By blocking the calcium signal on adrenal cortex cells, they directly reduce aldosterone production, which correlates to lower blood pressure.
Since blood pressure is in intimate feedback with cardiac output and peripheral resistance, with relatively low blood pressure, the afterload on the heart decreases; this decreases how hard the heart must work to eject blood into the aorta, so the amount of oxygen required by the heart decreases accordingly. This can help ameliorate symptoms of ischaemic heart disease such as angina pectoris.

Reducing the force of contraction of the myocardium is known as the negative inotropic effect of calcium channel blockers.
Slowing down the conduction of electrical activity within the heart, by blocking the calcium channel during the plateau phase of the action potential of the heart (see: cardiac action potential), results in a negative chronotropic effect, or a lowering of heart rate. This can increase the potential for heart block. The negative chronotropic effects of CCBs make them a commonly used class of agents in individuals with atrial fibrillation or flutter in whom control of the heart rate is generally a goal. Negative chronotropy can be beneficial when treating a variety of disease processes because lower heart rates represent lower cardiac oxygen requirements. Elevated heart rate can result in significantly higher "cardiac work", which can result in symptoms of angina.
The class of CCBs known as dihydropyridines mainly affect arterial vascular smooth muscle and lower blood pressure by causing vasodilation. The phenylalkylamine class of CCBs mainly affect the cells of the heart and have negative inotropic and negative chronotropic effects. The benzothiazepine class of CCBs combine effects of the other two classes.
Because of the negative inotropic effects, the nondihydropyridine calcium channel blockers should be avoided (or used with caution) in individuals with cardiomyopathy.[28]
Unlike beta blockers, calcium channel blockers do not decrease the responsiveness of the heart to input from the sympathetic nervous system. Since moment-to-moment blood pressure regulation is carried out by the sympathetic nervous system (via the baroreceptor reflex), calcium channel blockers allow blood pressure to be maintained more effectively than do beta blockers. However, because dihydropyridine CCBs result in a decrease in blood pressure, the baroreceptor reflex often initiates a reflexive increase in sympathetic activity leading to increased heart rate and contractility.
Ionic calcium is antagonized by magnesium ions in the nervous system. Because of this, bioavailable supplements of magnesium, possibly including magnesium chloride, magnesium lactate, and magnesium aspartate, may increase or enhance the effects of calcium channel blockade.[29]
N-type calcium channels are found in neurons and are involved in the release of neurotransmitter at synapses. Ziconotide is a selective blocker of these calcium channels and acts as an analgesic.
History
In 1883, Sydney Ringer realised the importance of calcium in cellular function.[9] Calcium channel blockers were first identified in the laboratory of German pharmacologist Albrecht Fleckenstein beginning in 1964.[9][30]
In 1025, Avicenna introduced the medicinal use of Taxus baccata for phytotherapy in The Canon of Medicine. He named this herbal drug "Zarnab" and used it as a cardiac remedy. This was the first known use of a calcium channel blocker drug, which were not in wide use in the Western world until the 1960s.[31]
Research
Ethanol

Research indicates ethanol is involved in the inhibition of L-type calcium channels. One study showed the nature of ethanol binding to L-type calcium channels is according to first-order kinetics with a Hill coefficient around 1. This indicates ethanol binds independently to the channel, expressing noncooperative binding.[32] Early studies showed a link between calcium and the release of vasopressin by the secondary messenger system.[33] Vasopressin levels are reduced after the ingestion of alcohol.[34] The lower levels of vasopressin from the consumption of alcohol have been linked to ethanol acting as an antagonist to voltage-gated calcium channels (VGCCs). Studies conducted by Treistman et al. in the aplysia confirm inhibition of VGCC by ethanol. Voltage clamp recordings have been done on the aplysia neuron. VGCCs were isolated and calcium current was recorded using patch clamp technique having ethanol as a treatment. Recordings were replicated at varying concentrations (0, 10, 25, 50, and 100 mM) at a voltage clamp of +30 mV. Results showed calcium current decreased as concentration of ethanol increased.[35] Similar results have shown to be true in single-channel recordings from isolated nerve terminal of rats that ethanol does in fact block VGCCs.[36]
Agatoxin in spider venom
Research on the desert grass spider, Agelenopsis aperta, has shown that agatoxins IVA and IVB found in their venom selectively block calcium channels. These agatoxins are found in other spider species as well. Desert grass spider bites to insects result in rapid paralysis, but bites to humans are not considered medically significant.[37]
References
- ↑ Godfraind, Théophile (2012). "Preface". Calcium Channel Blockers. Springer Basel Ag. p. VII. ISBN 978-3-0348-9599-6. Archived from the original on 2021-08-28. Retrieved 2021-04-18.
- ↑ "Generic Name Stems - Drug Information Portal - U.S. National Library of Medicine". druginfo.nlm.nih.gov. Archived from the original on 13 April 2021. Retrieved 13 March 2021.
- ↑ The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances (PDF). WHO. 2011. p. 37. Archived (PDF) from the original on 2018-07-12. Retrieved 2021-04-12.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 4.9 Hitchings, Andrew; Lonsdale, Dagan; Burrage, Daniel; Baker, Emma (2019). The Top 100 Drugs: Clinical Pharmacology and Practical Prescribing (2nd ed.). Elsevier. p. 100. ISBN 978-0-7020-7442-4. Archived from the original on 2021-05-22. Retrieved 2021-11-09.
- ↑ 5.0 5.1 5.2 5.3 5.4 Hariri, L; Patel, J (January 2021). "Vasodilators". PMID 32119310.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 6.0 6.1 6.2 6.3 McKeever, RG; Hamilton, RJ (January 2021). "Calcium Channel Blockers". PMID 29494080.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Tfelt-Hansen, P; Tfelt-Hansen, J (2009). "Verapamil for cluster headache. Clinical pharmacology and possible mode of action". Headache. Review. 49 (1): 117–25. doi:10.1111/j.1526-4610.2008.01298.x. PMID 19125880.
- ↑ Katzung, Bertram G. (2020). "12. Vasodilators & the treatment of angina pectoris". In Katzung, Bertram G.; Trevor, Anthony J. (eds.). Basic and Clinical Pharmacology 15e. New York: McGraw-Hill. pp. 199–217. ISBN 978-1-260-45231-0. Archived from the original on 2021-10-10. Retrieved 2021-10-20.
- ↑ 9.0 9.1 9.2 Godfraind, Théophile (29 May 2017). "Discovery and Development of Calcium Channel Blockers". Frontiers in Pharmacology. 8: 286. doi:10.3389/fphar.2017.00286. ISSN 1663-9812. PMID 28611661. Archived from the original on 29 August 2021. Retrieved 13 April 2021.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 10.7 Ritter, James M.; Flower, Rod; Henderson, Graeme; Loke, Yoon Kong; Rang, Humphrey P. (2020). "22. The heart". Rang & Dale's Pharmacology. Elsevier. p. 287-288. ISBN 978-0-7020-7448-6. Archived from the original on 2021-08-28. Retrieved 2021-04-16.
- ↑ "CV Pharmacology | Calcium-Channel Blockers (CCBs)". www.cvpharmacology.com. Archived from the original on 16 April 2021. Retrieved 16 April 2021.
- ↑ Remuzzi G, Scheppati A, Ruggenenti P (2002). "Clinical Practice. Nephropathy in Patients with Type 2 Diabetes". New England Journal of Medicine. 346 (15): 1145–51. doi:10.1056/NEJMcp011773. PMID 11948275.
- ↑ 13.00 13.01 13.02 13.03 13.04 13.05 13.06 13.07 13.08 13.09 13.10 13.11 13.12 13.13 13.14 13.15 13.16 13.17 13.18 Shackett, Pete (2020). Nuclear Medicine Technology: Procedures and Quick Reference (3rd ed.). Philadelphia: Wolters Kluwer. p. 115. ISBN 978-1-9751-1983-6. Archived from the original on 2021-08-28. Retrieved 2021-04-20.
- ↑ Hockerman, G.H.; Peterson, B.Z.; Johnson, B.D.; Catterall, W.A. (1997). "Molecular Determinants of Drug Binding and Action on L-Type Calcium Channels". Annual Review of Pharmacology and Toxicology. 37: 361–96. doi:10.1146/annurev.pharmtox.37.1.361. PMID 9131258. S2CID 16275155.
- ↑ Bezprozvanny I, Tsien RW (1995). "Voltage-Dependent Blockade of Diverse Types of Voltage-Gated Ca2+
Channels Expressed in Xenopus Oocytes by the Ca2+
Channel Antagonist Mibefradil (Ro 40-5967)". Molecular Pharmacology. 48 (3): 540–49. PMID 7565636. Archived from the original on 2015-12-21. Retrieved 2012-07-09. - ↑ Scultéty S, Tamáskovits E (1991). "Effect of Ca2+
Antagonists on Isolated Rabbit Detrusor Muscle". Acta Physiologica Hungarica. 77 (3–4): 269–78. PMID 1755331. - ↑ Chen N, Zhou M, Yang M, Guo J, Zhu C, Yang J, Wang Y, Yang X, He L (2010). "Calcium channel blockers versus other classes of drugs for hypertension". Cochrane Database of Systematic Reviews. 8 (8): CD003654. doi:10.1002/14651858.CD003654.pub4. PMID 20687074.
- ↑ "Calcium Channel Blockers". MedicineNet. p. 2. Archived from the original on 2012-04-21. Retrieved 2012-05-06.
- ↑ Norman M Kaplan, MD; Burton D Rose, MD (Apr 3, 2000). "Major side effects and safety of calcium channel blockers". Chinese Medical & Biological Information. Archived from the original on December 30, 2011. Retrieved July 23, 2012.
- ↑ 20.0 20.1 20.2 BNF (80 ed.). London: BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 168-176. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ 21.0 21.1 Raymond, Rebekah; Flanagan, Madeleine (7 June 2017). "Chronic oedema: treatment and the impact of prescribed medicines". The Pharmaceutical Journal. Archived from the original on 2 March 2021. Retrieved 20 April 2021.
- ↑ Weir, Matthew R. (2003). "Incidence of Pedal Edema Formation With Dihydropyridine Calcium". Medscape. Archived from the original on 20 April 2021. Retrieved 20 April 2021.
- ↑ Scallan, Joshua P.; Zawieja, Scott D.; Castorena‐Gonzalez, Jorge A.; Davis, Michael J. (15 October 2016). "Lymphatic pumping: mechanics, mechanisms and malfunction". The Journal of Physiology. 594 (20): 5749–5768. doi:10.1113/JP272088. ISSN 0022-3751. PMID 27219461. Archived from the original on 28 August 2021. Retrieved 20 April 2021.
- ↑ Engebretsen, Kristin M.; Kaczmarek, Kathleen M.; Morgan, Jenifer; Holger, Joel S. (2011). "High-dose insulin therapy in beta-blocker and calcium channel-blocker poisoning". Clinical Toxicology. 49 (4): 277–283. doi:10.3109/15563650.2011.582471. ISSN 1556-9519. PMID 21563902.
- ↑ Buckley N, Dawson A, Whyte I (2007). "Calcium Channel Blockers". Medicine. 35 (11): 599–602. doi:10.1016/j.mpmed.2007.08.025.
- ↑ Yousef; et al. (2005). "The mechanism of action of calcium channel blockers in the treatment of diabetic nephropathy" (PDF). Int J Diabetes & Metabolism. 13 (2): 76–82. doi:10.1159/000497574. Archived from the original (PDF) on 2015-10-10. Retrieved 2013-06-29.
- ↑ 27.0 27.1 Felizola SJ, Maekawa T, Nakamura Y, Satoh F, Ono Y, Kikuchi K, Aritomi S, Ikeda K, Yoshimura M, Tojo K, Sasano H (2014). "Voltage-gated calcium channels in the human adrenal and primary aldosteronism". J Steroid Biochem Mol Biol. 144 (part B): 410–16. doi:10.1016/j.jsbmb.2014.08.012. PMID 25151951.
- ↑ Lehne R (2010). Pharmacology for Nursing Care (7th ed.). St. Louis, Missouri: Saunders Elsevier. p. 505. ISBN 978-1-4160-6249-3.
- ↑ Iseri LT, French JH (1984). "Magnesium: Nature's Physiologic Calcium Blocker". American Heart Journal. 108 (1): 188–93. doi:10.1016/0002-8703(84)90572-6. PMID 6375330.
- ↑ Fleckenstein, A. (1983). "History of calcium antagonists". Circulation Research. 52 (2 Pt 2): 13–16. PMID 6339106.
- ↑ Tekol, Y. (2007). "The medieval physician Avicenna used an herbal calcium channel blocker, Taxus baccata L". Phytotherapy Research. 21 (7): 701–02. doi:10.1002/ptr.2173. PMID 17533639.
- ↑ Wang X, Wang G, Lemos JR, Treistman SN (September 1994). "Ethanol directly modulates gating of a dihydropyridine-sensitive Ca2+
channel in neurohypophysial terminals". J. Neurosci. 14 (9): 5453–60. doi:10.1523/JNEUROSCI.14-09-05453.1994. PMC 6577079. PMID 7521910. - ↑ Tobin V, Leng G, Ludwig M (2012). "The involvement of actin, calcium channels and exocytosis proteins in somato-dendritic oxytocin and vasopressin release". Front Physiol. 3: 261. doi:10.3389/fphys.2012.00261. PMC 3429037. PMID 22934017.
- ↑ Chiodera P, Coiro V (May 1990). "Inhibitory effect of ethanol on the arginine vasopressin response to insulin-induced hypoglycemia and the role of endogenous opioids". Neuroendocrinology. 51 (5): 501–04. doi:10.1159/000125383. PMID 2112727.
- ↑ Treistman SN, Bayley H, Lemos JR, Wang XM, Nordmann JJ, Grant AJ (1991). "Effects of ethanol on calcium channels, potassium channels, and vasopressin release". Ann. N. Y. Acad. Sci. 625: 249–63. doi:10.1111/j.1749-6632.1991.tb33844.x. PMID 1647726.
- ↑ Walter HJ, Messing RO (August 1999). "Regulation of neuronal voltage-gated calcium channels by ethanol". Neurochem. Int. 35 (2): 95–101. doi:10.1016/s0197-0186(99)00050-9. PMID 10405992.
- ↑ Adams, Michael E. (April 2004). "Agatoxins: ion channel specific toxins from the american funnel web spider, Agelenopsis aperta". Toxicon. 43 (5): 509–525. doi:10.1016/j.toxicon.2004.02.004. ISSN 0041-0101. Archived from the original on 2021-08-29. Retrieved 2020-12-08.
External links
- Calcium+Channel+Blockers at the US National Library of Medicine Medical Subject Headings (MeSH)
| Classification | |
|---|---|
| Identifiers |