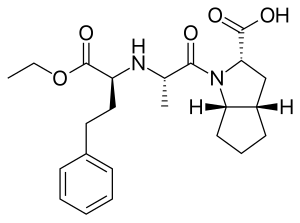
Ramipril
 | |
| Names | |
|---|---|
| Trade names | Altace, others |
| |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | 2.5 mg[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692027 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 28% |
| Protein binding | 73% (ramipril) 56% (ramiprilat) |
| Metabolism | Liver, to ramiprilat |
| Elimination half-life | 13 to 17 hours |
| Excretion | Kidney (60%) and fecal (40%) |
| Chemical and physical data | |
| Formula | C23H32N2O5 |
| Molar mass | 416.518 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 109 °C (228 °F) |
| |
| |
Ramipril, sold under the brand name Altace among others, is a medication used to treat high blood pressure, heart failure, and diabetic kidney disease.[2] Also used to prevent cardiovascular disease in those at high risk.[2] It is a reasonable initial treatment for high blood pressure.[2] It is taken by mouth.[2]
Common side effects include headaches, dizziness, feeling tired, and cough.[2] Serious side effects may include liver problems, angioedema, kidney problems, and high blood potassium.[2] Use in pregnancy and breastfeeding is not recommended.[3] It is an ACE inhibitor and works by decreasing renin-angiotensin-aldosterone system activity.[2]
Ramipril was patented in 1981 and approved for medical use in 1989.[4] It is available as a generic medication.[5] A month supply in the United Kingdom costs the NHS less than £1 as of 2020.[5] In the United States, the wholesale cost of this amount is about US$2.[6] In 2017, it was the 134th most commonly prescribed medication in the United States, with more than five million prescriptions.[7][8]
Medical uses
Medical uses include:
- High blood pressure
- Congestive heart failure[9]
- Following heart attack in people with evidence of heart failure
- People over 55 years at high risk: prevention of heart attack, stroke, cardiovascular death, or need of revascularization procedures
- Kidney damage due to diabetes with protein in the urine (In low doses it is used as a prophylaxis for developing nephropathy and related secondary cardiovascular events.)[10]
Dosage
The defined daily dose is 2.5 mg.[1]
Side effects
- Shakiness
- Dry cough
- Dizziness and light-headedness due to low blood pressure
- Fatigue, especially in the early stages
- Mouth dryness in the early stages
- Nausea
- Fainting
- Signs of infection (e.g., fever, chills, persistent sore throat)
- Chest pain
- Neutropenia (low white blood cells)
- Impotence (erectile dysfunction)[11]
- Hyperkalemia
Serious allergic reactions to this drug are unlikely, but immediate medical attention must be sought if they occur. Symptoms of a serious allergic reaction include, but are not limited to a rash or swelling of the face, mouth, tongue, or throat. In extreme cases, ramipril may lead to potentially fatal liver problems.
Contraindications to its use include renovascular disease (impaired blood flow in the kidneys), severe kidney impairment (especially in patients with one kidney or with bilateral renal artery stenosis), people who are volume-depleted, a history of angioedema while on an ACE inhibitors, pregnancy and hypotension.[citation needed]
People should not take ramipril (or any ACE inhibitors) if they have hyperkalemia. It is also recommended to avoid using salt-substitutes as this can further increase potassium levels in the blood.[2]
Mechanism of action
letter codes and icons may differ
ACE inhibitors inhibit the actions of angiotensin converting enzyme (ACE), thereby lowering the production of angiotensin II and decreasing the breakdown of bradykinin. The decrease in angiotensin II results in relaxation of arteriole smooth muscle leading to a decrease in total peripheral resistance, reducing blood pressure as the blood is pumped through widened vessels. Its effect on bradykinin is responsible for the dry cough side effect.
Ramipril, a prodrug or precursor drug, is converted to the active metabolite ramiprilat by carboxylesterase 1.[12][13] Ramiprilat is mostly excreted by the kidneys. Its half-life is variable (3–16 hours), and is prolonged by heart and liver failure, as well as kidney failure.
Society and culture
Cost
A month supply of 2.5mg/day in the United Kingdom costs the NHS less than £1 as of 2020.[5] In the United States, the wholesale cost of this amount is about US$2.[6] In 2017, it was the 134th most commonly prescribed medication in the United States, with more than five million prescriptions.[7][8]
-
Ramipril costs (US)
-
Ramipril prescriptions (US)
US patent
The compound was protected by a patent which was assigned to the German pharmaceutical company Hoechst AG (since merged into Aventis) on 29 October 1991.[14] The patent was scheduled to expire on 29 October 2008. On 11 September 2007, in an appeal by the Indian company Lupin Ltd., the United States Court of Appeals for the Federal Circuit reversed a district court trial verdict and found that Aventis's patent on ramipril was invalid for "obviousness", opening this drug to generic manufacturers.
Brand names
It is marketed as Prilace by Arrow Pharmaceuticals in Australia, Ramipro by Westfield Pharma in the Philippines, Tritace by Sanofi-Aventis in Italy and United States and Altace by King Pharmaceuticals in the United States, Novapril by Pharmanova in Ghana, Ramitens by PharmaSwiss, Ampril by Krka in Slovenia, Corpril by Cemelog-BRS in Hungary, Piramil and Prilinda by Hemofarm in Serbia, by Lek in Poland and by Novartis and Opsonin Pharma Limited as Ramace in Bangladesh, and in Canada as Altace (Sonfi) and Ramipril (Pharmascience).
Ramipril is marketed in India under the brand names Cardace, Zigpril, Ramistar, Odipril and Zorem . Ramipril is marketed in Myanmar under brand name Endpril .
Research
The Heart Outcomes and Prevention Evaluation trial[15][16] seemed to show ramipril possessed cardioprotective qualities which extended beyond its qualities as an antihypertensive. However, the trial and the interpretation of its results have been criticised.[17]
The AIRE trial[12][18] showed a 27% reduction in mortality for patients receiving ramipril for chronic heart failure following a myocardial infarction.
Ramipril was found to have similar results as telmisartan, an angiotensin II receptor blocker.[19]
References
- ↑ 1.0 1.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 18 October 2020. Retrieved 7 September 2020.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 "Ramipril Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 28 June 2019. Retrieved 3 March 2019.
- ↑ "Ramipril Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 13 October 2017. Retrieved 3 March 2019.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 469. ISBN 9783527607495. Archived from the original on 1 March 2019. Retrieved 1 March 2019.
- ↑ 5.0 5.1 5.2 BNF (80 ed.). London: BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. pp. 184–185. ISBN 978-0-85711-369-6.
- ↑ 6.0 6.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 6 March 2019. Retrieved 3 March 2019.
- ↑ 7.0 7.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- ↑ 8.0 8.1 "Ramipril - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
- ↑ Pilote L; Abrahamowicz M; Eisenberg M; Humphries K; Behlouli H; Tu JV (May 2008). "Effect of different angiotensin-converting-enzyme inhibitors on mortality among elderly patients with congestive heart failure". CMAJ. 178 (10): 1303–11. doi:10.1503/cmaj.060068. PMC 2335176. PMID 18458262.
- ↑ Remuzzi, Giuseppe (April 2006). "Prevention and Treatment of Diabetic Renal Disease in Type 2 Diabetes: The BENEDICT Study". Journal of the American Society of Nephrology. 2. 14 (4): 590–597.
- ↑ "Ramipril Side Effects". eMedTV: Health Information Brought To Life. Archived from the original on 17 August 2011. Retrieved 3 May 2011.
- ↑ 12.0 12.1 Frampton JE, Peters DH (March 1995). "Ramipril. An updated review of its therapeutic use in essential hypertension and heart failure". Drugs. 49 (3): 440–66. doi:10.2165/00003495-199549030-00008. PMID 7774515.
- ↑ Thomsen R; Rasmussen HB; Linnet K; INDICES Consortium. (January 2014). "In vitro drug metabolism by human carboxylesterase 1: focus on angiotensin-converting enzyme inhibitors". Drug Metab Dispos. 42 (1): 126–33. doi:10.1124/dmd.113.053512. PMID 24141856. Archived from the original on 29 August 2021. Retrieved 10 December 2019.
- ↑ U.S. Patent 5,061,722
- ↑ Hypertension Online Slides - ramipril, mortality, kidney failure, HOPE Archived 2 August 2012 at archive.today
- ↑ Ltd, BMJ Publishing Group (1 March 2000). "Ramipril reduced mortality and cardiovascular morbidity in high risk adults". BMJ Evidence-Based Medicine. 5 (2): 47–47. doi:10.1136/ebm.5.2.47. Archived from the original on 3 March 2020. Retrieved 3 March 2020 – via ebm.bmj.com.
- ↑ http://www.medscape.com/viewarticle/430926 Archived 13 December 2003 at the Wayback Machine "Debate: Do ACE Inhibitors Have Unique Properties, Beyond Their Antihypertensive Effect?"
- ↑ "Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators". Lancet. 342 (8875): 821–8. October 1993. doi:10.1016/0140-6736(93)92693-N. PMID 8104270.
- ↑ Yusuf S, Teo KK, Pogue J, et al. (April 2008). "Telmisartan, ramipril, or both in patients at high risk for vascular events". N. Engl. J. Med. 358 (15): 1547–59. doi:10.1056/NEJMoa0801317. hdl:2437/81925. PMID 18378520.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Webarchive template archiveis links
- Webarchive template wayback links
- Use dmy dates from April 2020
- Articles with invalid date parameter in template
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from August 2017
- Articles with hatnote templates targeting a nonexistent page
- ACE inhibitors
- Carboxamides
- Carboxylic acids
- Enantiopure drugs
- Ethyl esters
- Prodrugs
- Sanofi
- Pfizer brands
- AstraZeneca brands
- RTT

