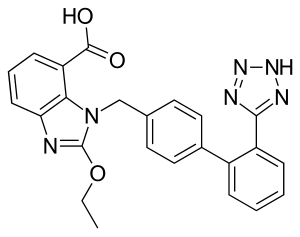
Candesartan
 | |
| Names | |
|---|---|
| Pronunciation | /ˌkændɪˈsɑːrtən/ |
| Trade names | Atacand, others |
| Other names | Candesartan cilexetil |
| |
| Clinical data | |
| Drug class | Angiotensin II receptor blocker (ARB)[1] |
| Main uses | High blood pressure, heart failure, diabetic kidney disease[1][2] |
| Side effects | Headache, dizziness, tiredness, cough, gastrointestinal upset[3] |
| Pregnancy category |
|
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601033 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 15% (candesartan cilexetil) |
| Metabolism | Candesartan cilexetil: intestinal wall; candesartan: liver (CYP2C9) |
| Elimination half-life | 9 hours |
| Excretion | Kidney 33%, faecal 67% |
| Chemical and physical data | |
| Formula | C24H20N6O3 |
| Molar mass | 440.463 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Candesartan is a medication used to treat high blood pressure, heart failure, and diabetic kidney disease.[1][2] In high blood pressure, it is one of a number of possible first line agents.[2] It is taken by mouth.[2]
Common side effects include headache, dizziness, tiredness, cough, and gastrointestinal upset.[3] Other side effects may include high potassium, low blood pressure, and kidney problems.[1] Use in pregnancy may harm the baby.[3] It is a angiotensin II receptor blocker (ARB).[1]
Candesartan was patented in 1990 and approved for medical use in 1997.[4] It is available as a generic medication.[5] In the United States a month of 32 mg costs about 25 USD as of 2021.[6] In the United Kingdom this amount costs the NHS about £2.[5]
Medical uses
High blood pressure
As with other angiotensin II receptor antagonists, candesartan is indicated for the treatment of hypertension.[7] Candesartan has an additive hypertensive effect when combined with a diuretic, such as chlorthalidone. It is available in a fixed-combination formulation with a low dose of the thiazide diuretic hydrochlorothiazide. Candesartan/hydrochlorothiazide combination preparations are marketed under various trade names including Atacand Plus, Hytacand, Blopress Plus, Advantec and Ratacand Plus.
Heart failure
Angiotensin receptor blockers such as candesartan and valsartan have been demonstrated in randomised controlled trials to reduce heart failure hospitalisations and cardiovascular deaths for chronic heart failure patients with reduced left ventricular ejection fraction (LVEF ≤40%) and are intolerant to angiotensin-converting enzyme inhibitors.[8][9][10]
Atrial fibrillation
A 2005 meta-analysis found a reduction in atrial fibrillation in people with systolic left ventricular dysfunction treated with candesartan, another angiotensin receptor blocker or an angiotensin converting enzyme inhibitor.[11] Evidence for the use of candesartan specifically is also supported by an analysis of the CHARM study which demonstrated a reduction in atrial fibrillation in patients with systolic left ventricular dysfunction.[12] While these studies have demonstrated a potential additional benefit for candesartan when used in patients with systolic left ventricular dysfunction, additional studies are required to further elucidate the role of candesartan in the prevention of atrial fibrillation in other population groups.
Diabetic retinopathy
Use of antihypertensive drugs has been demonstrated to slow the progression of diabetic retinopathy; the role of candesartan specifically in reducing progression in type 1 and type 2 diabetes is still up for debate.[13][14][15] Results from a 2008 study on patients with type 1 diabetes showed there was no benefit in using candesartan to reduce progression of diabetic retinopathy when compared to placebo.[14] Candesartan has been demonstrated to reverse the severity (cause regression) of mild to moderate diabetic retinopathy in patients with type 2 diabetes.[15] The patient populations investigated in these studies were limited to mostly Caucasians and those younger than 75 years of age, so generalization of these findings to other population groups should be done with caution.[14][15]
Migraines
In two small trials candesartan was useful to prevent migraines,[16][17] however, further studies would enhance confidence in its use for this indication.
Dosage
The typical dose is 16 to 32 mg per day.[3]
Side effects
As with other drugs that inhibit the renin–angiotensin system, if candesartan is taken by pregnant women during the second or third trimester, it can cause injury and in some cases, death of the developing fetus. Symptomatic hypotension may occur in people who take candesartan and are volume-depleted or salt-depleted, as can also occur when diuretics are coadministered. Reduction in renal glomerular filtration rate may occur; people with renal artery stenosis may be at higher risk. Hyperkalemia may occur; people who are also taking spironolactone or eplerenone may be at higher risk.[7]
Anemia may occur, due to inhibition of the renin–angiotensin system.[18]
As with other angiotensin receptor blockers, candesartan can rarely cause severe liver injury.[19]
Chemistry
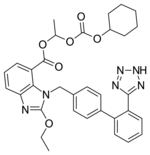
Candesartan is marketed as the cyclohexyl 1-hydroxyethyl carbonate (cilexetil) ester, known as "candesartan cilexetil". Candesartan cilexetil is metabolised completely by esterases in the intestinal wall during absorption to the active candesartan moiety.
The use of a prodrug form increases the bioavailability of candesartan. Despite this, absolute bioavailability is relatively poor at 15% (candesartan cilexetil tablets) to 40% (candesartan cilexetil solution). Its IC50 is 15 μg/kg.
History
The compound known as TCV-116 (candesartan) was studied by Japanese scientists using standard laboratory rats. Animal studies were published showing the effectiveness of the compound in 1992–1993, with a pilot study on humans published in the summer of 1993.[20][21]
Society and culture
Names
The prodrug candesartan cilexetil is marketed by AstraZeneca and Takeda Pharmaceuticals, commonly under the trade names Blopress, Atacand, Amias, and Ratacand. It is available in generic form.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "DailyMed - CANDESARTAN tablet". dailymed.nlm.nih.gov. Archived from the original on 7 February 2019. Retrieved 29 December 2021.
- ↑ 2.0 2.1 2.2 2.3 "Candesartan Monograph for Professionals". Drugs.com. Archived from the original on 23 January 2021. Retrieved 29 December 2021.
- ↑ 3.0 3.1 3.2 3.3 "Candesartan". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 27 August 2021. Retrieved 29 December 2021.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 471. ISBN 9783527607495. Archived from the original on 2021-10-31. Retrieved 2021-10-24.
- ↑ 5.0 5.1 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 189. ISBN 978-0857114105.
- ↑ "Candesartan Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 25 October 2021. Retrieved 29 December 2021.
- ↑ 7.0 7.1 "Candesartan label" (PDF). FDA. February 2016. Archived (PDF) from the original on 2021-07-01. Retrieved 2021-10-24. For label updates see FDA index page for IND 020838 Archived 2021-08-08 at the Wayback Machine
- ↑ Pfeffer, Marc A; Swedberg, Karl; Granger, Christopher B; Held, Peter; McMurray, John JV; Michelson, Eric L; Olofsson, Bertil; Östergren, Jan; Yusuf, Salim (September 2003). "Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM-Overall programme". The Lancet. 362 (9386): 759–766. doi:10.1016/s0140-6736(03)14282-1. ISSN 0140-6736. Archived from the original on 2021-10-31. Retrieved 2021-10-24.
- ↑ Yusuf, Salim; Pfeffer, Marc A; Swedberg, Karl; Granger, Christopher B; Held, Peter; McMurray, John JV; Michelson, Eric L; Olofsson, Bertil; Östergren, Jan (September 2003). "Effects of candesartan in patients with chronic heart failure and preserved left-ventricular ejection fraction: the CHARM-Preserved Trial". The Lancet. 362 (9386): 777–781. doi:10.1016/s0140-6736(03)14285-7. ISSN 0140-6736. Archived from the original on 2021-10-31. Retrieved 2021-10-24.
- ↑ Cohn, Jay N.; Tognoni, Gianni (2001-12-06). "A Randomized Trial of the Angiotensin-Receptor Blocker Valsartan in Chronic Heart Failure". New England Journal of Medicine. 345 (23): 1667–1675. doi:10.1056/NEJMoa010713. ISSN 0028-4793. PMID 11759645. Archived from the original on 2021-10-31. Retrieved 2021-10-24.
- ↑ Healey, Jeff S., Adrian Baranchuk, Eugene Crystal, Carlos A. Morillo, Michael Garfinkle, Salim Yusuf, and Stuart J. Connolly. "Prevention of atrial fibrillation with angiotensin-converting enzyme inhibitors and angiotensin receptor blockers: a meta-analysis." Journal of the American College of Cardiology 45, no. 11 (2005): 1832-1839.
- ↑ Ducharme, Anique; Swedberg, Karl; Pfeffer, Marc A.; Cohen-Solal, Alain; Granger, Christopher B.; Maggioni, Aldo P.; Michelson, Eric L.; McMurray, John J.V.; Olsson, Lars; Rouleau, Jean L.; Young, James B. (July 2006). "Prevention of atrial fibrillation in patients with symptomatic chronic heart failure by candesartan in the Candesartan in Heart failure: Assessment of Reduction in Mortality and morbidity (CHARM) program". American Heart Journal. 152 (1): 86–92. doi:10.1016/j.ahj.2005.06.036. Archived from the original on 2020-09-01. Retrieved 2021-10-24.
- ↑ Elkjaer AS, Lynge SK, Grauslund J (February 2020). "Evidence and indications for systemic treatment in diabetic retinopathy: a systematic review". Acta Ophthalmol. 98 (4): 329–336. doi:10.1111/aos.14377. PMID 32100477. S2CID 211523341.
- ↑ 14.0 14.1 14.2 Chaturvedi N, Porta M, Klein R, Orchard T, Fuller J, Parving HH, et al. Effect of candesartan on prevention (DIRECT-Prevent 1) and progression (DIRECT-Protect 1) of retinopathy in type 1 diabetes: randomised, placebo-controlled trials. The Lancet. 2008;372(9647):1394-402.
- ↑ 15.0 15.1 15.2 Sjølie AK, Klein R, Porta M, Orchard T, Fuller J, Parving HH, et al. Effect of candesartan on progression and regression of retinopathy in type 2 diabetes (DIRECT-Protect 2): a randomised placebo-controlled trial. The Lancet. 2008;372(9647):1385-93.
- ↑ Dorosch T, Ganzer CA, Lin M, Seifan A (September 2019). "Efficacy of Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers in the Preventative Treatment of Episodic Migraine in Adults". Curr Pain Headache Rep. 23 (11): 85. doi:10.1007/s11916-019-0823-8. PMID 31515634. S2CID 202557362.
- ↑ Cernes R, Mashavi M, Zimlichman R (2011). "Differential clinical profile of candesartan compared to other angiotensin receptor blockers". Vasc Health Risk Manag. 7: 749–59. doi:10.2147/VHRM.S22591. PMC 3253768. PMID 22241949.
- ↑ Cheungpasitporn, W; Thongprayoon, C; Chiasakul, T; Korpaisarn, S; Erickson, SB (November 2015). "Renin-angiotensin system inhibitors linked to anemia: a systematic review and meta-analysis". QJM : Monthly Journal of the Association of Physicians. 108 (11): 879–84. doi:10.1093/qjmed/hcv049. PMID 25697787.

- ↑ Patti R, Sinha A, Sharma S, Yoon TS, Kupfer Y (May 2019). "Losartan-induced Severe Hepatic Injury: A Case Report and Literature Review". Cureus. 11 (5): e4769. doi:10.7759/cureus.4769. PMC 6663042. PMID 31363450.
- ↑ Mizuno, K.; et al. (1992). "Hypotensive activity of TCV-116, a newly developed angiotensin II receptor antagonist, in spontaneously hypertensive rats". Life Sci. 51 (20): PL183-187. doi:10.1016/0024-3205(92)90627-2. PMID 1435062.
- ↑ Ogihara, T.; et al. (Jul–Aug 1993). "Pilot study of a new angiotensin II receptor antagonist, TCV-116: effects of a single oral dose on blood pressure in patients with essential hypertension". Clin. Ther. 15 (4): 684–91. PMID 8221818.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with hatnote templates targeting a nonexistent page
- Articles with changed KEGG identifier
- Angiotensin II receptor antagonists
- Tetrazoles
- Benzimidazoles
- Ethers
- Carboxylic acids
- Biphenyls
- AstraZeneca brands
- Takeda Pharmaceutical Company brands
- RTT