Hydrochlorothiazide
 | |
 | |
| Names | |
|---|---|
| Trade names | Apo-hydro, others |
| |
| Clinical data | |
| Drug class | Diuretic |
| Main uses | High blood pressure, swelling[1] |
| Side effects | Poor kidney function, electrolyte imbalance, gout, high blood sugar, feeling lightheaded with standing[2] |
| Pregnancy category | |
| Routes of use | By mouth (capsules, tablets, solution) |
| Defined daily dose | 25 mg[4] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682571 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | Variable (~70% on average) |
| Metabolism | Not significant[5] |
| Elimination half-life | 5.6–14.8 h |
| Excretion | Primarily kidney (>95% as unchanged drug) |
| Chemical and physical data | |
| Formula | C7H8ClN3O4S2 |
| Molar mass | 297.73 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Hydrochlorothiazide (HCTZ or HCT) is a diuretic medication often used to treat high blood pressure and swelling due to fluid build up.[2] Other uses include diabetes insipidus, renal tubular acidosis, and to decrease the risk of kidney stones in those with a high calcium level in the urine.[2] For high blood pressure it is sometimes considered as a first-line treatment.[2][6] HCTZ is taken by mouth and may be combined with other blood pressure medications as a single pill to increase effectiveness.[2]
Potential side effects include poor kidney function, electrolyte imbalances including low blood potassium and less commonly low blood sodium, gout, high blood sugar, and feeling lightheaded with standing.[2] While allergies to HCTZ are reported to occur more often in those with allergies to sulfa drugs, this association is not well supported.[2] It may be used during pregnancy, but it is not a first-line medication in this group.[2]
It is in the thiazide medication class and acts by decreasing the kidneys' ability to retain water.[2] This initially reduces blood volume, decreasing blood return to the heart and thus cardiac output.[7] It is believed to lower peripheral vascular resistance in the long run.[7]
Two companies, Merck and Ciba, state they discovered the medication which became commercially available in 1959.[8] It is on the World Health Organization's List of Essential Medicines.[9] It is available as a generic drug[2] and is relatively affordable.[10] In 2017, it was the twelfth most commonly prescribed medication in the United States, with more than 42 million prescriptions.[11][12] In the UK, it is only available in combination form.[13]
Medical uses
Hydrochlorothiazide is frequently used for the treatment of hypertension, congestive heart failure, symptomatic edema, diabetes insipidus, renal tubular acidosis.[2] It is also used for the prevention of kidney stones in those who have high levels of calcium in their urine.[2]
A 2019 review found that it was less effective than chlortalidone but also had fewer side effects.[14] A 2022 trial however found no difference HCT and and chlortalidone.[15]
Hydrochlorothiazide is also sometimes used to prevent osteopenia and for treatment of hypoparathyroidism,[16] hypercalciuria, Dent's disease, and Ménière's disease. For diabetes insipidus, the effect of thiazide diuretics is presumably mediated by a hypovolemia-induced increase in proximal sodium and water reabsorption, thereby diminishing water delivery to the ADH-sensitive sites in the collecting tubules and increasing the urine osmolality.[medical citation needed]
Thiazides are also used in the treatment of osteoporosis. Thiazides decrease mineral bone loss by promoting calcium retention in the kidney, and by directly stimulating osteoblast differentiation and bone mineral formation.[17]
It may be given together with other antihypertensive agents in fixed-dose combination preparations, such as in losartan/hydrochlorothiazide.
Dosage
The defined daily dose of HCTZ is 25 mg (by mouth).[4] The dose however may range from 12.5 to 100 mg.[2] For high blood pressure generally 12.5 to 25 mg once per day is used.[1] For edema 25 mg once to twice per day may be used with the maximum dose being 100 mg.[1]
Side effects
- Low blood potassium is an occasional side effect. It can be usually prevented by potassium supplements or by combining hydrochlorothiazide with a potassium-sparing diuretic
- Other disturbances in electrolytes including [[hypomagnesemia|low blood magnesium, low blood sodium, and high blood calcium.
- Hyperuricemia (high levels of uric acid in the blood). All thiazide diuretics including hydrochlorothiazide can inhibit excretion of uric acid by the kidneys, thereby increasing serum concentrations of uric acid. This may increase the chance of gout.[18]
- Hyperglycemia, high blood sugar
- Hyperlipidemia, high cholesterol and triglycerides
- Headache
- Nausea/vomiting
- Photosensitivity
- Weight gain
- Pancreatitis
There is also a slight increase in non melanoma skin cancer attributed to the fact that HCTZ makes a person more sensitive to the sun.[19]
The frequency of side effects increase as the medication dose is increased and the highest occurrence of side effects occur at doses greater than 25 mg per day.[medical citation needed]
Package inserts, based on case reports and observational studies, have reported that an allergy to a sulfa drug predisposes the patient to cross sensitivity to a thiazide diuretic. A 2005 review of the literature did not find support for this cross-sensitivity.[20]
Mechanism of action
Hydrochlorothiazide belongs to thiazide class of diuretics. It reduces blood volume by acting on the kidneys to reduce sodium (Na+) reabsorption in the distal convoluted tubule. The major site of action in the nephron appears on an electroneutral NaCl co-transporter by competing for the chloride site on the transporter. By impairing Na+ transport in the distal convoluted tubule, hydrochlorothiazide induces a natriuresis and concomitant water loss. Thiazides increase the reabsorption of calcium in this segment in a manner unrelated to sodium transport.[21] Additionally, by other mechanisms, HCTZ is believed to lower peripheral vascular resistance.[7]
Society and culture

Cost
In 2017, it was the twelfth most commonly prescribed medication in the United States, with more than 42 million prescriptions.[11][12]
-
Hydrochlorothiazide costs (US)
-
Hydrochlorothiazide prescriptions (US)
Trade names
Hydrochlorothiazide is available as a generic drug under a large number of brand names, including Apo-Hydro, Aquazide, BPZide, Dichlotride, Esidrex, Hydrochlorot, Hydrodiuril, HydroSaluric, Hypothiazid, Microzide, Oretic and many others.[medical citation needed]
To reduce pill burden and in order to reduce side effects, hydrochlorothiazide is often used in fixed-dose combinations with many other classes of antihypertensive drugs such as:
- ACE inhibitors — e.g. Prinzide or Zestoretic (with lisinopril), Co-Renitec (with enalapril), Capozide (with captopril), Accuretic (with quinapril), Monopril HCT (with fosinopril), Lotensin HCT (with benazepril), etc.
- Angiotensin receptor blockers — e.g. Hyzaar (with losartan), Co-Diovan or Diovan HCT (with valsartan), Teveten Plus (with eprosartan), Avalide or CoAprovel (with irbesartan), Atacand HCT or Atacand Plus (with candesartan), etc.
- Beta blockers — e.g. Ziac or Lodoz (with bisoprolol), Nebilet Plus or Nebilet HCT (with nebivolol), Dutoprol or Lopressor HCT (with metoprolol), etc.
- Direct renin inhibitors — e.g. Co-Rasilez or Tekturna HCT (with aliskiren)
- Potassium sparing diuretics: Dyazide and Maxzide triamterene[22]
Sport
Use of hydrochlorothiazide is prohibited by the World Anti-Doping Agency for its ability to mask the use of performance-enhancing drugs.[23]
Combination medicine
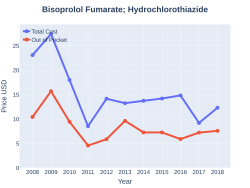
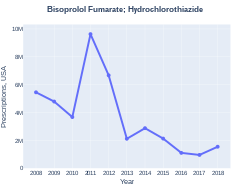
Bisoprolol fumarate and hydrochlorothiazide is a combination medicine. This medication is used for individuals with hypertension (using a benzothiadiazine diuretic and a adrenoceptor blocking agent)[24] This combination medicine has a cost in the U.S. of $33.49(USD) for 90 tablets (5-6.25 mg)[25]
-
Bisoprolol fumarate/hydrochlorothiazide costs (US)
-
Bisoprolol fumarate/hydrochlorothiazide prescriptions (US)
See also
References
- ↑ 1.0 1.1 1.2 "HYDROCHLOROTHIAZIDE oral - Essential drugs". medicalguidelines.msf.org. Archived from the original on 29 August 2021. Retrieved 30 August 2020.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 "Hydrochlorothiazide". The American Society of Health-System Pharmacists. Archived from the original on 10 May 2017. Retrieved 30 November 2016.
- ↑ 3.0 3.1 "Hydrochlorothiazide Use During Pregnancy". Drugs.com. 30 July 2019. Archived from the original on 21 October 2020. Retrieved 19 January 2020.
- ↑ 4.0 4.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 2 July 2020. Retrieved 22 September 2020.
- ↑ Beermann B, Groschinsky-Grind M, Rosén A (1976). "Absorption, metabolism, and excretion of hydrochlorothiazide". Clin Pharmacol Ther. 19 (5 (Pt 1)): 531–37. doi:10.1002/cpt1976195part1531. PMID 1277708.
- ↑ Wright, JM; Musini, VM; Gill, R (18 April 2018). "First-line drugs for hypertension". The Cochrane Database of Systematic Reviews. 4: CD001841. doi:10.1002/14651858.CD001841.pub3. PMC 6513559. PMID 29667175.
- ↑ 7.0 7.1 7.2 Duarte JD, Cooper-DeHoff RM (June 2010). "Mechanisms for blood pressure lowering and metabolic effects of thiazide and thiazide-like diuretics". Expert Rev Cardiovasc Ther. 8 (6): 793–802. doi:10.1586/erc.10.27. PMC 2904515. PMID 20528637. NIHMSID: NIHMS215063.
- ↑ Ravina, Enrique (2011). The evolution of drug discovery: from traditional medicines to modern drugs (1 ed.). Weinheim: Wiley-VCH. p. 74. ISBN 9783527326693. Archived from the original on 10 January 2015.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Best drugs to treat high blood pressure The least expensive medications may be the best for many people". November 2014. Archived from the original on 3 January 2015. Retrieved 10 January 2015.
- ↑ 11.0 11.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 12.0 12.1 "Hydrochlorothiazide - Drug Usage Statistics". ClinCalc. 1 December 1981. Archived from the original on 11 April 2020. Retrieved 11 April 2020.
- ↑ "2. Cardiovascular system". British National Formulary (BNF) (82 ed.). London: BMJ Group and the Pharmaceutical Press. September 2021 – March 2022. ISBN 978-0-85711-413-6.
{{cite book}}: Unknown parameter|pagse=ignored (help) - ↑ Acelajado MC, Hughes ZH, Oparil S, Calhoun DA (March 2019). "Treatment of Resistant and Refractory Hypertension". Circ. Res. 124 (7): 1061–1070. doi:10.1161/CIRCRESAHA.118.312156. PMC 6469348. PMID 30920924.
A long-acting thiazide-like diuretic, specifically chlorthalidone, if available, is recommended over hydrochlorothiazide (HCTZ) given its superior efficacy and clear benefit demonstrated in multiple outcome studies of hypertension... However, chlorthalidone use is more frequently associated with adverse metabolic effects, particularly hypokalemia and hyponatremia, compared with HCTZ.
- ↑ Ishani, Areef; Cushman, William C.; Leatherman, Sarah M.; Lew, Robert A.; Woods, Patricia; Glassman, Peter A.; Taylor, Addison A.; Hau, Cynthia; Klint, Alison; Huang, Grant D.; Brophy, Mary T.; Fiore, Louis D.; Ferguson, Ryan E. (14 December 2022). "Chlorthalidone vs. Hydrochlorothiazide for Hypertension–Cardiovascular Events". New England Journal of Medicine: NEJMoa2212270. doi:10.1056/NEJMoa2212270.
- ↑ Mitchell DM, Regan S, Cooley MR, Lauter KB, Vrla MC, Becker CB, Burnett-Bowie SA, Mannstadt M (December 2012). "Long-term follow-up of patients with hypoparathyroidism". J. Clin. Endocrinol. Metab. 97 (12): 4507–14. doi:10.1210/jc.2012-1808. PMC 3513540. PMID 23043192.
- ↑ Dvorak MM; De Joussineau C; Carter DH; et al. (2007). "Thiazide diuretics directly induce osteoblast differentiation and mineralized nodule formation by targeting a NaCl cotransporter in bone". J. Am. Soc. Nephrol. 18 (9): 2509–16. doi:10.1681/ASN.2007030348. PMC 2216427. PMID 17656470.
- ↑ Rheumatology (Oxford) 2017;56(5):679-688
- ↑ Research, Center for Drug Evaluation and (20 August 2020). "FDA approves label changes to hydrochlorothiazide to describe small risk of non-melanoma skin cancer". FDA. Archived from the original on 26 November 2020. Retrieved 24 August 2020.
- ↑ Johnson, KK; Green, DL; Rife, JP; Limon, L (February 2005). "Sulfonamide cross-reactivity: fact or fiction?". The Annals of Pharmacotherapy. 39 (2): 290–301. doi:10.1345/aph.1E350. PMID 15644481.
- ↑ Uniformed Services University Pharmacology Note Set #3 2010, Lectures #39 & #40, Eric Marks
- ↑ "Triamterene and Hydrochlorothiazide". MedlinePlus. 1 January 2020. Archived from the original on 2 January 2020. Retrieved 1 January 2020.
- ↑ "Prohibited List" (PDF). World Anti-Doping Agency. January 2018. Archived (PDF) from the original on 22 October 2017. Retrieved 30 May 2018.
- ↑ "Bisoprolol Hydrochlorothiazide - FDA prescribing information, side effects and uses". Drugs.com. Archived from the original on 18 April 2019. Retrieved 19 April 2021.
- ↑ "Bisoprolol fumarate/hydrochlorothiazide". Rx Saver. Retrieved 18 April 2021.
{{cite web}}: CS1 maint: url-status (link)
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 errors: unsupported parameter
- CS1 maint: url-status
- Use dmy dates from January 2020
- Articles with invalid date parameter in template
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from January 2020
- AMPA receptor positive allosteric modulators
- Antihypertensive agents
- Benzothiadiazines
- Merck brands
- Carbonic anhydrase inhibitors
- Diuretics
- IARC Group 2B carcinogens
- Sulfonamides
- Thiazides
- World Anti-Doping Agency prohibited substances
- World Health Organization essential medicines
- RTT