Mesna
 | |
| Names | |
|---|---|
| Pronunciation | /ˈmɛznə/ |
| |
| Clinical data | |
| Drug class | Organosulfur compound[1] |
| Main uses | Prevent toxicity from cyclophosphamide or ifosfamide[2] |
| Side effects | Headache, vomiting, sleepiness, loss of appetite, cough, rash, joint pain[2] |
| Pregnancy category |
|
| Routes of use | by mouth, intravenous |
| Defined daily dose | 1.2 g [3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 45–79% (by mouth) |
| Metabolism | Oxidised in circulation |
| Elimination half-life | 0.36–8.3 hours |
| Excretion | kidney |
| Chemical and physical data | |
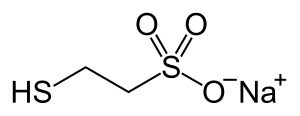
| Formula | C2H5NaO3S2 |
| Molar mass | 164.17 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Mesna, sold under the brand name Mesnex among others, is a medication used in those taking cyclophosphamide or ifosfamide to decrease the risk of bleeding from the bladder.[2] It is used either by mouth or injection into a vein.[2]
Common side effects include headache, vomiting, sleepiness, loss of appetite, cough, rash, and joint pain.[2] Serious side effects include allergic reactions.[2] Use during pregnancy appears to be safe for the baby but this use has not been well studied.[4] Mesna is an organosulfur compound.[1] It works by altering the breakdown products of cyclophosphamide and ifosfamide found in the urine making them less toxic.[2]
Mesna was approved for medical use in the United States in 1988.[2] It is on the World Health Organization's List of Essential Medicines.[5] The wholesale cost in the developing world is about US$3.50 per 400 mg vial.[6] In the United Kingdom this amount costs the NHS about £3.95.[7]
Medical uses
Chemotherapy adjuvant
Mesna is used therapeutically to reduce the incidence of haemorrhagic cystitis and haematuria when a patient receives ifosfamide or cyclophosphamide for cancer chemotherapy. These two anticancer agents, in vivo, may be converted to urotoxic metabolites, such as acrolein.
Mesna assists to detoxify these metabolites by reaction of its sulfhydryl group with α,β-unsaturated carbonyl containing compounds such as acrolein.[8] This reaction is known as a Michael addition. Mesna also increases urinary excretion of cysteine.
Other
Outside North America, mesna is also used as a mucolytic agent, working in the same way as acetylcysteine; it is sold for this indication as Mistabron[9] and Mistabronco.
Dosage
The defined daily dose is 1.2 gram (inhaled).[3]
It is administered intravenously or orally (per mouth).[10] The IV mesna infusions would be given with IV ifosfamide, while oral mesna would be given with oral cyclophosphamide. The oral doses must be double the intravenous (IV) mesna dose due to bioavailability issues. The oral preparation allows patients to leave the hospital sooner, instead of staying four to five days for all the IV mesna infusions.
Mechanism of action
Mesna reduces the toxicity of urotoxic compounds that may form after chemotherapy administration. Mesna is a water-soluble compound with antioxidant properties, and is given concomitantly with the chemotherapeutic agents cyclophosphamide and ifosfamide. Mesna concentrates in the bladder where acrolein accumulates after administration of chemotherapy and through a Michael addition, forms a conjugate with acrolein and other urotoxic metabolites.[8] This conjugation reaction inactivates the urotoxic compounds to harmless metabolites. The metabolites are then excreted in the urine.[11]
Names
It is marketed by Baxter as Uromitexan and Mesnex. The name of the substance is an acronym for 2-mercaptoethane sulfonate Na (Na being the chemical symbol for sodium).
See also
- Coenzyme M—a coenzyme with the same structure used by methanogenic bacteria
References
- ↑ 1.0 1.1 Patwardhan, Bhushan; Chaguturu, Rathnam (2016). Innovative Approaches in Drug Discovery: Ethnopharmacology, Systems Biology and Holistic Targeting. Academic Press. p. 53. ISBN 9780128018224. Archived from the original on 2016-12-21.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 "Mesna". The American Society of Health-System Pharmacists. Archived from the original on 11 May 2017. Retrieved 8 December 2016.
- ↑ 3.0 3.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 27 July 2020. Retrieved 17 September 2020.
- ↑ "Mesna (Mesnex) Use During Pregnancy". www.drugs.com. Archived from the original on 11 May 2017. Retrieved 20 December 2016.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Mesna". International Drug Price Indicator Guide. Archived from the original on 11 May 2017. Retrieved 8 December 2016.
- ↑ British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. p. 578. ISBN 9780857111562.
- ↑ 8.0 8.1 Thurston, David E. (2007). Chemistry and Pharmacology of Anticancer Drugs. Boca Raton: CRC Press/Taylor & Francis. pp. 53–54. ISBN 978-1-4200-0890-6. Archived from the original on 2016-05-19.
- ↑ "Mistabron Ampoules". South African Electronic Package Inserts. August 1973. Archived from the original on 2008-10-22. Retrieved 2008-08-12.
- ↑ Mace JR, Keohan ML, Bernardy H, et al. (December 2003). "Crossover randomized comparison of intravenous versus intravenous/oral mesna in soft tissue sarcoma treated with high-dose ifosfamide". Clin. Cancer Res. 9 (16 Pt 1): 5829–34. PMID 14676103. Archived from the original on 2013-01-12. Retrieved 2008-12-18.
- ↑ Shaw IC, Graham MI (1987). "Mesna—a short review". Cancer Treat. Rev. 14 (2): 67–86. doi:10.1016/0305-7372(87)90041-7. PMID 3119211.
External links
| Identifiers: |
|---|
- BC Cancer Agency
- NIH/MedlinePlus patient information Archived 2008-10-01 at the Wayback Machine
- Mesna at the US National Library of Medicine Medical Subject Headings (MeSH)
- Pages using duplicate arguments in template calls
- Articles with hatnote templates targeting a nonexistent page
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with changed CASNo identifier
- Webarchive template wayback links
- Chemotherapeutic adjuvants
- Thiols
- Expectorants
- Organic sodium salts
- World Health Organization essential medicines
- RTT
- Sulfonates