Rapacuronium bromide
Jump to navigation
Jump to search
This article relies largely or entirely on a single source. (November 2007) |
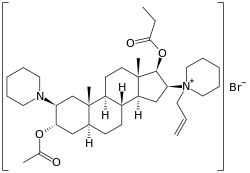 Rapacuronium bromide | |
| Clinical data | |
|---|---|
| Other names | [(2S, 3S, 5S, 8R, 9S, 10S, 13S, 14S, 16S, 17S)-3-acetyloxy-10,13-dimethyl-2-(1-piperidyl)-16-(1-prop-2-enyl-3,4,5,6-tetrahydro-2H-pyridin-1-yl)-2 ,3 ,4 ,5 ,6 ,7 ,8 ,9 ,11 ,12 ,14, 15, 16, 17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]propanoate |
| Routes of administration | Intravenous |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Not applicable |
| Protein binding | Variable |
| Metabolism | Hydrolyzed to active metabolites CYP system not involved |
| Elimination half-life | 141 minutes (mean) |
| Excretion | Renal and fecal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.211.226 |
| Chemical and physical data | |
| Formula | C37H61N2O4+ |
| Molar mass | 597.905 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Rapacuronium bromide (brand name Raplon) is a rapidly acting, non-depolarizing aminosteroid neuromuscular blocker formerly used in modern anaesthesia, to aid and enable endotracheal intubation, which is often necessary to assist in the controlled ventilation of unconscious patients during surgery and sometimes in intensive care. As a non-depolarizing agent it did not cause initial stimulation of muscles before weakening them.[1]
Due to risk of fatal bronchospasm it was withdrawn from the United States market by Organon on March 27, 2001, less than 2 years after its FDA approval in 1999.[2]
References
- ^ Onrust SV, Foster RH (November 1999). "Rapacuronium bromide: a review of its use in anaesthetic practice". Drugs. 58 (5): 887–918. doi:10.2165/00003495-199958050-00011. PMID 10595867. S2CID 46984904.
- ^ Shapse D, et al. (Organon International) (27 March 2001). "Voluntary Market Withdrawal" (PDF). Food and Drug Administration. Archived from the original (PDF) on 24 September 2008.
Categories:
- Articles with short description
- Short description matches Wikidata
- Articles needing additional references from November 2007
- All articles needing additional references
- Drugs not assigned an ATC code
- Drugs with non-standard legal status
- Articles with changed CASNo identifier
- Articles with changed EBI identifier
- ECHA InfoCard ID from Wikidata
- Articles without KEGG source
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Muscle relaxants
- Nicotinic antagonists
- Withdrawn drugs
- Quaternary ammonium compounds
- 1-Piperidinyl compounds
- Acetate esters
- Propionate esters
- Androstanes
- Neuromuscular blockers
- All stub articles
- Musculoskeletal system drug stubs