Macitentan
 | |
| Names | |
|---|---|
| Trade names | Opsumit |
| Other names | ACT-064992 |
| |
| Clinical data | |
| Drug class | Endothelin receptor antagonist (ERA)[1] |
| Main uses | Pulmonary arterial hypertension (PAH)[2] |
| Side effects | Low red blood cells, headache, sore throat[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Typical dose | 10 mg OD[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a615033 |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Metabolism | Hydrolysis, oxidation (CYP3A4) |
| Excretion | 2/3 urine, 1/3 faeces |
| Chemical and physical data | |
| Formula | C19H20Br2N6O4S |
| Molar mass | 588.28 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Macitentan, sold under the brand name Opsumit, is a medication used to treat pulmonary arterial hypertension (PAH).[2] It may be used alone or with PDE5 inhibitors or prostanoids.[2] It is taken by mouth.[2]
Common side effects include low red blood cells, headache, and sore throat.[1] Other side effects include liver problems, infertility, and pulmonary edema.[2] Use in pregnancy may harm the baby.[2] It is an endothelin receptor antagonist (ERA) which help widen widen the arteries in the lungs.[1]
Macitentan was approved for medical use in the United States and Europe in 2013.[1][2] Approval for a generic version was granted in 2021.[5] In the United Kingdom a month of medication costs the NHS about £2,300 as of 2021.[3] In the United States this amount costs about 11,000 USD.[6]
Medical uses
Dosage
It is taken at a dose of 10 mg per day.[3]
Side effects
The FDA has issued a black box warning for macitentan for embryo-fetal toxicity. It is only available to females in the U.S through a risk evaluation and mitigation strategy (REMS) program.[7]
Mechanism of action
Macitentan
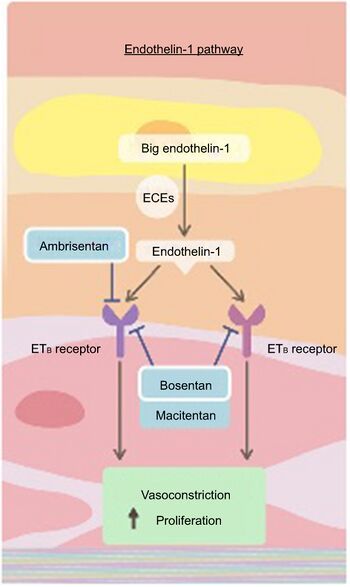
Macitentan blocks the ET1-dependent rise in intracellular calcium by inhibiting the binding of ET-1 to ET receptors. Blocking of the ETA receptor subtype seems to be of more importance in the treatment of PAH than blocking of ETB, likely because there are higher numbers of ETA receptors than ETB receptors in pulmonary arterial smooth muscle cells.[9] The blocking of Endothelin 1 leads to vasodilation and decreases the proliferation of cells in the vessels of the artries which contributes to the narrowing and leads to the pulmonary arterial hypertension.
It is a dual ERA, meaning that it acts as an antagonist of two endothelin (ET) receptor subtypes, ETA and ETB.[10] However, macitentan has a 50-fold increased selectivity for the ETA subtype compared to the ETB subtype.[11]
Endothelin
Endothelin (ET) is an extremely potent blood vessel constricting substance that is secreted by endothelial cells.[9] In the lungs, the most common ET form released is ET-1.[9] ET-1 release can occur through both constitutive and non-constitutive pathways.[9] Upon release, ET-1 can bind to the ET receptors that are expressed on arterial smooth muscle cells and fibroblasts in the lungs.[9] ET receptors are G protein coupled receptors and, when activated, lead to an increase in intracellular calcium levels via the Gαq signaling pathway.[9]There are two receptor subtypes that endothelin will bind to: ETA and ETB. ETA is associated with cell growth and vasoconstriction while ETB is responsible for anti-proliferation of cells, vasodilation and ET-1 clearance. The rise in intracellular calcium leads to contraction of the arterial smooth muscle, as well as vascular remodelling due to cell proliferation.[9] Prolonged constriction and fibrosis are factors in the pathogenesis of PAH.[10]
Pharmacokinetics

Macitentan is taken as a 10 mg oral dose once a day.[10] Its half-life in humans is about 16 hours and steady state is reached by the third day of administration.[12] It is absorbed slowly into the plasma.[13] Macitentan dealkylates into the active metabolite ACT-132577, which reaches its peak plasma concentration about 30 hours after the first dose is administered, and has a half-life of approximately 48 hours.[13] Although ACT-132577 has a lower affinity for the ET receptors than its parent compound,[11] It maintains higher plasma concentrations than macitentan.[13] Both compounds can be excreted from the body through the urine or feces.[12]
Co-administration of ciclosporin has only a slight effect on the concentrations of macitentan and its active metabolite, while rifampicin decreases the area under the curve (AUC) of the drug's blood plasma concentration by 79%, and ketoconazole approximately doubles it. This corresponds to the finding that macitentan is mainly metabolised via the liver enzyme CYP3A4.[14]
Experimental pharmacokinetics
Macitentan has slow association kinetics.[9] Its potency increases 6.3-fold when it is pre-incubated with pulmonary arterial smooth muscle cells for 120 minutes compared to 10 minutes with pulmonary arterial smooth muscle cells.[9] Macitentan also has a high receptor occupancy half-life (approximately 17 minutes) compared to bosentan (approximately 70 seconds) and ambrisentan (approximately 40 seconds).[9] This increased receptor occupancy half-life allows macitentan to act as a non-competitive antagonist of ET receptors.[9] Bosentan and ambrisentan are both competitive antagonists.[9]
Similar medications
The other two ERAs marketed as of 2014 are bosentan and ambrisentan.[10]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Opsumit". Archived from the original on 21 June 2021. Retrieved 13 November 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Macitentan Monograph for Professionals". Drugs.com. Archived from the original on 28 October 2020. Retrieved 13 November 2021.
- ↑ 3.0 3.1 3.2 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 198. ISBN 978-0857114105.
- ↑ "Opsumit- macitentan tablet, film coated". DailyMed. 22 September 2020. Archived from the original on 27 October 2020. Retrieved 24 October 2020.
- ↑ "Macitentan: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 24 June 2021. Retrieved 19 June 2021.
- ↑ "Opsumit Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 18 April 2021. Retrieved 13 November 2021.
- ↑ Opsumit [package insert] Whippany, NJ: Bayer HealthCare Pharmaceuticals 2017
- ↑ Wong, Alexandra K.; Channick, Richard N. (3 September 2019). "Safety and tolerability of macitentan in the management of pulmonary arterial hypertension: an update". Drug, Healthcare and Patient Safety. 11: 71–85. doi:10.2147/DHPS.S173050.
- ↑ 9.00 9.01 9.02 9.03 9.04 9.05 9.06 9.07 9.08 9.09 9.10 9.11 Gatfield J, Mueller Grandjean C, Sasse T, Clozel M, Nayler O (2012). "Slow receptor dissociation kinetics differentiate macitentan from other endothelin receptor antagonists in pulmonary arterial smooth muscle cells". PLOS ONE. 7 (10): e47662. Bibcode:2012PLoSO...747662G. doi:10.1371/journal.pone.0047662. PMC 3471877. PMID 23077657.
- ↑ 10.0 10.1 10.2 10.3 Hong IS, Coe HV, Catanzaro LM (April 2014). "Macitentan for the treatment of pulmonary arterial hypertension". The Annals of Pharmacotherapy. 48 (4): 538–47. doi:10.1177/1060028013518900. PMID 24458948. S2CID 24720486.
- ↑ 11.0 11.1 Iglarz M, Binkert C, Morrison K, Fischli W, Gatfield J, Treiber A, et al. (December 2008). "Pharmacology of macitentan, an orally active tissue-targeting dual endothelin receptor antagonist". The Journal of Pharmacology and Experimental Therapeutics. 327 (3): 736–45. doi:10.1124/jpet.108.142976. PMID 18780830. S2CID 6315900.
- ↑ 12.0 12.1 Bruderer S, Hopfgartner G, Seiberling M, Wank J, Sidharta PN, Treiber A, Dingemanse J (September 2012). "Absorption, distribution, metabolism, and excretion of macitentan, a dual endothelin receptor antagonist, in humans". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 42 (9): 901–10. doi:10.3109/00498254.2012.664665. PMID 22458347. S2CID 38365884.
- ↑ 13.0 13.1 13.2 Sidharta PN, van Giersbergen PL, Halabi A, Dingemanse J (October 2011). "Macitentan: entry-into-humans study with a new endothelin receptor antagonist". European Journal of Clinical Pharmacology. 67 (10): 977–84. doi:10.1007/s00228-011-1043-2. PMC 3169777. PMID 21541781.
- ↑ Bruderer S, Aänismaa P, Homery MC, Häusler S, Landskroner K, Sidharta PN, et al. (March 2012). "Effect of cyclosporine and rifampin on the pharmacokinetics of macitentan, a tissue-targeting dual endothelin receptor antagonist". The AAPS Journal. 14 (1): 68–78. doi:10.1208/s12248-011-9316-3. PMC 3282010. PMID 22189899.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Articles containing unverified chemical infoboxes
- Endothelin receptor antagonists
- Pyrimidines
- Sulfamides
- Johnson & Johnson brands
- Bromoarenes
- RTT