Atrial flutter
| Atrial flutter | |
|---|---|
 | |
| Atrial flutter with varying A-V conduction (5:1 and 4:1) | |
| Specialty | Cardiology |
| Symptoms | None, palpitations, chest discomfort, lightheadedness[1][2] |
| Complications | Stroke, cardiomyopathy[2] |
| Usual onset | Sudden[3] |
| Types | Typical, atypical[2] |
| Causes | Unknown[4] |
| Risk factors | COPD, pulmonary hypertension, heart failure[2] |
| Diagnostic method | Electrocardiogram (ECG)[2] |
| Differential diagnosis | Sinus tachycardia, atrial fibrillation, multifocal atrial tachycardia[2][3] |
| Treatment | Electrical cardioversion, catheter ablation[2] |
| Medication | Diltiazem, metoprolol, blood thinners[2] |
| Frequency | 9 per 10,000 people per year[5] |
Atrial flutter (AFL) is an abnormal heart rhythm that starts in the upper chambers of the heart.[2] Generally, it is initially presents with a sudden onset of a fast heart rate.[2][3] Symptoms may be absent or include a feeling of the heart beating too fast or hard, chest discomfort, shortness of breath, or lightheadedness.[1][2] Complications can include stroke or cardiomyopathy.[2]
The cause is often unknown.[4] Risk factors include COPD, pulmonary hypertension, heart failure, and endurance sports.[2][4] Triggers can include electrolyte abnormalities, high thyroid, or low oxygen.[2] It is a type of supraventricular tachycardia.[3] Diagnosis is generally by an electrocardiogram (ECG) showing a flutter wave at 300 beats per minute with narrow QRS complexes every 2nd beat.[2][3] If the diagnosis is unclear, giving adenosine may help clarify the condition.[3] It may also convert into atrial fibrillation (AF).[4]
The recommended treatment is generally conversion to sinus rhythm.[2] If the person is unstable, this is generally done with electrical cardioversion.[2] If the person is otherwise stable medications, such as diltiazem, or electricity may be used.[2] Blood thinners may be recommended before cardioversion in stable cases to decrease the risk of stroke.[2][6] Atrial flutter is often treated more definitively with a technique known as catheter ablation.[1] Blood thinners may be stopped 6 weeks after ablation, if it is successful.[4]
Atrial flutter is the second most common supraventricular tachycardia, occurring less than one-tenth as often as atrial fibrillation.[3][5] It newly occurs in about 88 per 100,000 people per year; 5 per 100,000 in those under 50 and 587 per 100,000 in those over 80.[5] Males are affected more often than females.[2] Atrial flutter was identified as an independent condition around 1912 by the British physician Sir Thomas Lewis (1881–1945), though the ECG pattern was described a year early by Jolly and Ritchie.[7][8]
Signs and symptoms
While atrial flutter can sometimes go unnoticed, its onset is often marked by characteristic sensations of the heart feeling like it is beating too fast or hard. Such sensations usually last until the episode resolves, or until the heart rate is controlled.
Atrial flutter is usually well tolerated initially (a high heart rate is for most people just a normal response to exercise), however, people with other underlying heart disease (such as coronary artery disease) or poor exercise tolerance may rapidly develop symptoms, such as shortness of breath, chest pain, lightheadedness or dizziness, nausea and, in some patients, nervousness and feelings of impending doom.
Prolonged atrial flutter with fast heart rates may lead to decompensation with loss of normal heart function (heart failure). This may manifest as exercise intolerance (exertional breathlessness), difficulty breathing at night, or swelling of the legs and/or abdomen.
Complications
Although often regarded as a relatively benign heart rhythm problem, atrial flutter shares the same complications as the related condition atrial fibrillation. There is paucity of published data directly comparing the two, but overall mortality in these conditions appears to be very similar.[9]
Rapid heart rates may produce significant symptoms in patients with pre-existing heart disease and can lead to inadequate blood flow to the heart muscle and even a heart attack.[1] In rare situations, atrial flutter associated with a fast heart rate persists for an extended period of time without being corrected to a normal heart rhythm and leads to a tachycardia-induced cardiomyopathy.[1] Even in individuals with a normal heart, if the heart beats too quickly for a prolonged period of time, this can lead to ventricular decompensation and heart failure.
Clot formation
Because there is little if any effective contraction of the atria there is stasis (pooling) of blood in the atria. Stasis of blood in susceptible individuals can lead to the formation of a thrombus (blood clot) within the heart. A thrombus is most likely to form in the atrial appendages. A blood clot in the left atrial appendage is particularly important as the left side of the heart supplies blood to the entire body through the arteries. Thus, any thrombus material that dislodges from this side of the heart can embolize (break off and travel) to the brain's arteries, with the potentially devastating consequence of a stroke. Thrombus material can, of course, embolize to any other portion of the body, though usually with a less severe outcome.
Sudden cardiac death
Sudden death is not directly associated with atrial flutter. However, in individuals with a pre-existing accessory conduction pathway, such as the bundle of Kent in Wolff-Parkinson-White syndrome, the accessory pathway may conduct activity from the atria to the ventricles at a rate that the AV node would usually block. Bypassing the AV node, the atrial rate of 300 beats/minute leads to a ventricular rate of 300 beats/minute (1:1 conduction). Even if the ventricles are able to sustain a cardiac output at such a high rates, 1:1 flutter with time may degenerate into ventricular fibrillation, causing hemodynamic collapse and death.
Pathophysiology
Atrial flutter is caused by a re-entrant rhythm. This usually occurs along the cavo-tricuspid isthmus of the right atrium though atrial flutter can originate from the left atrium as well. Typically initiated by a premature electrical impulse arising in the atria, atrial flutter is propagated due to differences in refractory periods of atrial tissue. This creates electrical activity that moves in a localized self-perpetuating loop, which usually lasts about 200 milliseconds for the complete circuit. For each cycle around the loop, an electric impulse results and propagates through the atria.
The impact and symptoms of atrial flutter depend on the heart rate of the affected person. Heart rate is a measure of the ventricular rather than atrial activity. Impulses from the atria are conducted to the ventricles through the atrio-ventricular node (AV node). In a person with atrial flutter, a 12-lead electrocardiogram (ECG) will demonstrate the atrial chambers of the heart contracting at a rate of 280–300 beats per minute whereas the ventricular chambers of the heart typically beat at a rate of 140–150 beats per minute.[3] Due primarily to its longer refractory period, the AV node exerts a protective effect on heart rate by blocking atrial impulses in excess of about 180 beats/minute, for the example of a resting heart rate. (This block is dependent on the age of the patient, and can be calculated roughly by subtracting patient age from 220). If the flutter rate is 300/minute only half of these impulses will be conducted, giving a ventricular rate of 150/minute, or a 2:1 heart block. The addition of rate-controlling drugs or conduction system disease can increase this block substantially (see image below).
Diagnosis
Typical atrial flutter is recognized on an electrocardiogram by presence of characteristic "flutter waves" at a regular rate of 200 to 300 beats per minute. Flutter waves may not be evident on an ECG in atypical forms of atrial flutter. Individual flutter waves may be symmetrical, resembling p-waves, or may be asymmetrical with a "sawtooth" shape, rising gradually and falling abruptly or vice versa. If atrial flutter is suspected clinically but is not clearly evident on ECG, acquiring a Lewis lead ECG may be helpful in revealing flutter waves.
Classification
There are two types of atrial flutter, the common type I and rarer type II.[10] Most individuals with atrial flutter will manifest only one of these. Rarely someone may manifest both types; however, they can manifest only one type at a time.
Type I
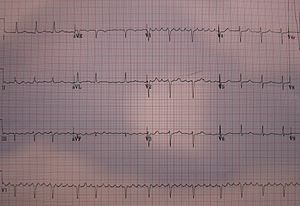

Type I atrial flutter, also known as common atrial flutter or typical atrial flutter, has an atrial rate of 240 to 340 beats/minute. However, this rate may be slowed by antiarrhythmic agents.
The reentrant loop circles the right atrium, passing through the cavo-tricuspid isthmus – a body of fibrous tissue in the lower atrium between the inferior vena cava, and the tricuspid valve.[1] Type I flutter is further divided into two subtypes, known as counterclockwise atrial flutter and clockwise atrial flutter depending on the direction of current passing through the loop.[1]
- Counterclockwise atrial flutter (known as cephalad-directed atrial flutter) is more commonly seen. The flutter waves in this rhythm are inverted in ECG leads II, III, and aVF.[1]
- The re-entry loop cycles in the opposite direction in clockwise atrial flutter, thus the flutter waves are upright in II, III, and aVF.[1]
Type II
Type II (atypical) atrial flutter follows a significantly different re-entry pathway to type I flutter, and is typically faster, usually 340–350 beats/minute.[11] Atypical atrial flutter rarely occurs in people who have not undergone previous heart surgery or previous catheter ablation procedures. Left atrial flutter is considered atypical and is common after incomplete left atrial ablation procedures.[12] Atypical atrial flutter originating from the right atrium and heart's septum have also been described.
Management
In general, atrial flutter should be managed the same as atrial fibrillation. Because both rhythms can lead to the formation of a blood clot in the atrium, individuals with atrial flutter usually require some form of anticoagulation or antiplatelet agent. Both rhythms can be associated with dangerously fast heart rates and thus require medication to control the heart rate (such as beta blockers or calcium channel blockers) and/or rhythm control with class III antiarrhythmics (such as ibutilide or dofetilide). However, atrial flutter is more resistant to correction with such medications than atrial fibrillation.[1] For example, although the class III antiarrhythmic agent ibutilide is an effective treatment for atrial flutter, rates of recurrence after treatment are quite high (70–90%).[1] Additionally, there are some specific considerations particular to treatment of atrial flutter.
Cardioversion
Atrial flutter is considerably more sensitive to electrical direct current cardioversion than atrial fibrillation, with a shock of only 20 to 50 Joules commonly being enough to cause a return to a normal heart rhythm (sinus rhythm). Exact placement of the pads does not appear important.[13]
Ablation
Due to the reentrant nature of atrial flutter, it is often possible to ablate the circuit that causes atrial flutter with radiofrequency catheter ablation. Catheter ablation is considered to be a first-line treatment method for many people with typical atrial flutter due to its high rate of success (>90%) and low incidence of complications.[1] This is done in the cardiac electrophysiology lab by causing a ridge of scar tissue in the cavotricuspid isthmus that crosses the path of the circuit that causes atrial flutter. Eliminating conduction through the isthmus prevents reentry, and if successful, prevents the recurrence of the atrial flutter. Atrial fibrillation often occurs (30% within 5 years) after catheter ablation for atrial flutter.[1]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 Sawhney, NS; Anousheh, R; Chen, WC; Feld, GK (February 2009). "Diagnosis and management of typical atrial flutter". Cardiology Clinics (Review). 27 (1): 55–67, viii. doi:10.1016/j.ccl.2008.09.010. PMID 19111764.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 2.17 2.18 2.19 Rodriguez Ziccardi, M; Goyal, A; Maani, CV (January 2020). "Atrial Flutter". PMID 31082029.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 Link, MS (October 2012). "Clinical practice. Evaluation and initial treatment of supraventricular tachycardia". New England Journal of Medicine. 367 (15): 1438–48. doi:10.1056/NEJMcp1111259. PMID 23050527.
- ↑ 4.0 4.1 4.2 4.3 4.4 Bennett, David H. (2012). Bennett's Cardiac Arrhythmias: Practical Notes on Interpretation and Treatment. John Wiley & Sons. p. 49. ISBN 978-1-118-43240-2. Archived from the original on 2016-10-02. Retrieved 2021-02-08.
- ↑ 5.0 5.1 5.2 Bun, SS; Latcu, DG; Marchlinski, F; Saoudi, N (September 2015). "Atrial flutter: more than just one of a kind". European Heart Journal. 36 (35): 2356–63. doi:10.1093/eurheartj/ehv118. PMID 25838435.
- ↑ Vadmann, H; Nielsen, PB; Hjortshøj, SP; Riahi, S; Rasmussen, LH; Lip, GY; Larsen, TB (September 2015). "Atrial flutter and thromboembolic risk: a systematic review". Heart. 101 (18): 1446–55. doi:10.1136/heartjnl-2015-307550. PMID 26149627.
- ↑ Lewis T, Feil HS, Stroud WD (1920). "Observations upon flutter, fibrillation, II: the nature of auricular flutter". Heart. 7: 191.
- ↑ Silverman, Mark E.; Fleming, Peter R.; Hollman, Arthur; Julian, Desmond G.; Krikler, Dennis M. (2012). British Cardiology in the 20th Century. Springer Science & Business Media. p. PT206. ISBN 978-1-4471-0773-6. Archived from the original on 2021-08-27. Retrieved 2021-02-08.
- ↑ Vidaillet H, Granada JF, Chyou PH, Maassen K, Ortiz M, Pulido JN, et al. (2002). "A Population-Based Study of Mortality among Patients with Atrial Fibrillation or Flutter". The American Journal of Medicine. 113 (5): 365–70. doi:10.1016/S0002-9343(02)01253-6. PMID 12401530.
- ↑ Surawicz, Borys; Knilans, Timothy K.; Chou, Te-Chuan (2001). Chou's electrocardiography in clinical practice: adult and pediatric. Philadelphia: Saunders. ISBN 978-0-7216-8697-4.[page needed]
- ↑ "Atrial Flutter: Overview". eMedicine Cardiology. Archived from the original on 26 February 2009. Retrieved 2009-03-06.
- ↑ Garan, H (April 2008). "Atypical atrial flutter". Heart Rhythm. 5 (4): 618–21. doi:10.1016/j.hrthm.2007.10.031. PMID 18325846.
- ↑ Kirkland, S; Stiell, I; AlShawabkeh, T; Campbell, S; Dickinson, G; Rowe, BH (July 2014). "The efficacy of pad placement for electrical cardioversion of atrial fibrillation/flutter: a systematic review". Academic Emergency Medicine. 21 (7): 717–26. doi:10.1111/acem.12407. PMID 25117151.
External links
| Classification | |
|---|---|
| External resources |