Brilaroxazine
 | |
| Clinical data | |
|---|---|
| Other names | RP5063; oxaripiprazole [1][2] |
| Routes of administration | By mouth |
| Drug class | Atypical antipsychotic |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >80% [3] |
| Protein binding | >99% |
| Metabolism | Liver (mostly via CYP3A4 (64%) and CYP2D6 (17%)) [4] |
| Elimination half-life | 55 hours [3] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
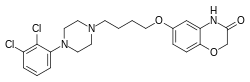
| Formula | C22H25Cl2N3O3 |
| Molar mass | 450.36 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Brilaroxazine (developmental code name RP5063), also known as oxaripiprazole,[1][2] is an investigational atypical antipsychotic which is under development by Reviva Pharmaceuticals for the treatment of neuropsychiatric and inflammatory disorders.[3][5][6][7] It has currently completed the first of two phase III clinical trials for schizophrenia. Reviva Pharmaceuticals also intends to investigate brilaroxazine for the treatment of bipolar disorder, major depressive disorder, attention deficit hyperactivity disorder (ADD/ADHD), psychosis/agitation associated with Alzheimer's disease, Parkinson's disease psychosis, as well as the inflammatory disorders pulmonary arterial hypertension (PAH), idiopathic pulmonary fibrosis (IPF), and psoriasis (topical gel).[3][8] The FDA granted brilaroxazine orphan drug designation for the treatment of PAH and IPF.
Brilaroxazine is a third-generation antipsychotic and dopamine-serotonin system modulator due to its unique actions on dopamine and serotonin neurotransmitter systems compared to other antipsychotics.[9][10][11] Clinical data from phase I, phase II (NCT01490086), and phase III (NCT05184335) trials suggest that brilaroxazine may have favorable efficacy and a significantly improved side effect profile compared to existing third-generation drugs.[9][12][13]
Pharmacology
Pharmacodynamics
Brilaroxazine acts as a potent partial agonist of D2, D3, D4 and 5-HT1A receptors, and as an antagonist of 5-HT2A, 5-HT2B, 5-HT2C, 5-HT6 and 5-HT7 receptors.[9][11] Brilaroxazine exhibits high affinity for D2S, D2L, D3, D4.4, 5-HT1A, 5-HT2A, 5-HT2B, and 5-HT7 receptors, and moderate affinity for D1, D5, 5-HT2C, 5-HT3, 5-HT6, H1 and α4β2 nicotinic receptors, the serotonin transporter, and the α1B adrenergic receptor.[9][11] It lacks significant affinity for 5-HT1B, α2 adrenergic, and muscarinic acetylcholine receptors, as well as for the norepinephrine and dopamine transporters.[11]
| Site | Ki (nM) | Action | |
|---|---|---|---|
| D1 | 100 | ND | |
| D2 | 0.40 | Partial agonist | |
| D2L | 0.45 | Partial agonist | |
| D2S | 0.28 | Partial agonist | |
| D3 | 3.7 | Partial agonist | |
| D4 | 6.0 | Partial agonist | |
| D5 | 200 | ND | |
| 5-HT1A | 1.5 | Partial agonist | |
| 5-HT2A | 2.5 | Weak partial agonist/ Antagonist | |
| 5-HT2B | 0.19 | Antagonist | |
| 5-HT2C | 39 | Antagonist | |
| 5-HT3 | 78 | ND | |
| 5-HT6 | 51 | Antagonist | |
| 5-HT7 | 2.7 | Antagonist | |
| α4β2 nicotinic | 36.3 | ND | |
| SERT | 107 | ND | |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | |||
Chemistry
Brilaroxazine is identical to aripiprazole in chemical structure except for the replacement of a methylene group in aripiprazole's quinolinone ring system with an oxygen atom, resulting instead in a benzoxazinone ring system. This structural change is within the molecule's secondary pharmacophore, which plays a major role in modulating its binding and intrinsic efficacy at dopamine and serotonin receptors.[14][15] The drug is also related structurally to brexpiprazole and cariprazine.
Recent Developments
In October 2023, Reviva Pharmaceuticals released topline results from their pivotal RECOVER phase III clinical trial (NCT05184335). The RECOVER study was a 4-week, randomized, double-blind, placebo-controlled, multicenter trial where 411 patients received brilaroxazine 15 mg, 50 mg, or placebo once daily. The trial cohort consisted of 60% USA, 34% Indian, and 6% Bulgarian patients with balanced randomization and diverse demographic representation across all three trial arms. The primary endpoint was PANSS Total Score change from baseline vs placebo at week 4. Secondary endpoints were PANSS Positive Symptoms, PANSS Negative Symptoms, PANSS Negative Marder Factor, PANSS Social Cognition, PANSS Excitement/Agitation, CGI-S, and Personal and Social Performance (PSP) score changes from baseline vs placebo at week 4.
Brilaroxazine 50 mg successfully met all primary and secondary endpoints with statistically significant and clinically meaningful improvements over placebo across all major symptom domains. The primary endpoint PANSS Total Score change from baseline was -23.9 for brilaroxazine 50 mg vs -13.8 for placebo at week 4, resulting in a 10.1 point reduction over placebo (p < 0.001). Within the first week of dosing, brilaroxazine 50 mg already achieved separation from placebo in the primary endpoint and several secondary endpoints.
| Endpoint | Point Reduction / Improvement | Cohen's d Effect Size | P Value |
|---|---|---|---|
| PANSS Total Score | 10.1 | 0.6 | <0.001 |
| PANSS Positive Symptoms | 2.8 | 0.5 | <0.001 |
| PANSS Negative Symptoms | 2.0 | 0.4 | 0.003 |
| PANSS Negative Marder Factor | 2.1 | 0.4 | 0.002 |
| PANSS Social Cognition | 1.6 | 0.5 | <0.001 |
| PANSS Excitement/Agitation | 2.1 | 0.5 | <0.001 |
| Personal and Social Performance (PSP) | 6.3 | 0.5 | <0.001 |
| CGI-S | >= 1 | 0.5 | <0.001 |
The lower brilaroxazine 15 mg dose met two secondary endpoints at week 4, PANSS Social Cognition and Personal and Social Performance (PSP), and showed a clear improvement trend and increasing separation from placebo in the primary endpoint PANSS Total Score and PANSS Positive Symptoms score starting at week 3.
Brilaroxazine was very well tolerated and safe, with overall treatment emergent adverse event (TEAE) rates of 34.5% for brilaroxazine 15 mg, 35.5% for 50 mg, and 30% for placebo. Discontinuation rates for brilaroxazine were lower than for placebo and were 19% for brilaroxazine 15 mg, 16% for 50 mg, and 22% for placebo. Discontinuation rates due to drug side effects were also lower for brilaroxazine than for placebo and were 1% for brilaroxazine 15 mg, 0% for 50 mg, and 4% for placebo. Common TEAEs (>5%) were headache (<6%) and somnolence (<=7.5%) but these were mild-to-moderate in severity and generally transient in nature. There were no incidences of suicidal ideation.
There was no significant change in bodyweight, blood glucose, lipids, prolactin, or thyroid hormones compared to placebo. The percentage of patients who gained weight during the trial was 2.1% for brilaroxazine 15 mg, 5.9% for 50 mg, and 2.9% for placebo. This is a significant improvement over the currently prescribed third-generation antipsychotics aripiprazole (Abilify), brexpiprazole (Rexulti), and cariprazine (Vraylar). In comparable short-term (4-6 week) acute schizophrenia clinical trials, the percentage of patients taking a third-generation antipsychotic that had clinically relevant weight gain (>= 7% gain) was 9.2% for aripiprazole vs 4.3% for placebo, 10.4% for brexpiprazole vs 4.1% for placebo, and 9.2% for cariprazine vs 4.7% for placebo.[16][17] The meta-analysis studies used pooled data from each drug's entire schizophrenia dose range, therefore the results for the highest 50 mg dose of brilaroxazine would likely show even more significant improvement in weight change against comparable highest dosages of third-generation antipsychotics if the breakdown were available.
Akathisia and extrapyramidal symptom (EPS) rates were very low and were 0% for brilaroxazine 15mg, 0.7% for 50 mg, and 0% for placebo. The percentage of patients with elevated LDL cholesterol compared to baseline was 0% for brilaroxazine 15 and 50 mg vs 2.9% for placebo, and the percentage of patients with low HDL cholesterol compared to baseline was 0.7% for brilaroxazine 15 mg, 1.4% for 50 mg, and 1.4% for placebo.
Reviva also has an ongoing 52-week, single-arm, phase III open-label extension (OLE) study of stable schizophrenia patients to further evaluate the long-term safety and tolerability of brilaroxazine as part of the RECOVER program (NCT05184335). In this study, rollover patients from the RECOVER 4-week double-blind trial as well as de novo stable schizophrenia patients receive brilaroxazine in a 15-30-50 mg flexible dosing schedule. Completion is expected in Q4 2024. In Q1 2024 Reviva plans to initiate RECOVER-2 in Q2 2024. RECOVER-2 is a confirmatory 4-week, randomized, double-blind, placebo-controlled, multicenter phase III clinical trial of 450 acute schizophrenia patients, where patients will receive brilaroxazine 30 mg, 50 mg, or placebo once daily. Completion is expected in Q2 2025 and brilaroxazine FDA new drug application (NDA) submission is expected in Q3 2025. If successful, commercial availability is anticipated by 2026.
See also
References
- ^ a b Modica MN, Lacivita E, Intagliata S, Salerno L, Romeo G, Pittalà V, Leopoldo M (October 2018). "Structure-Activity Relationships and Therapeutic Potentials of 5-HT7 Receptor Ligands: An Update". J Med Chem. 61 (19): 8475–8503. doi:10.1021/acs.jmedchem.7b01898. hdl:11586/218985. PMID 29767995.
- ^ a b Jordan AW (14 September 2018). Antidepressants: History, Science, and Issues. Abc-Clio. ISBN 9781440839276.
- ^ a b c d "Reviva Corporate Presentation". Reviva Pharmaceuticals. Retrieved 2023-07-19.
- ^ Cantillon M, Ings R, Prakash A, Bhat L (2018). "A Population Pharmacokinetic and Pharmacodynamic Analysis of RP5063 Phase 2 Study Data in Patients with Schizophrenia or Schizoaffective Disorder". European Journal of Drug Metabolism and Pharmacokinetics. 43 (5): 573–585. doi:10.1007/s13318-018-0472-z. PMC 6133081. PMID 29619682.
- ^ Medicines in Development for Mental Health (PDF) (Report). Pharmaceutical Research and Manufacturers of America. 2014. p. 20. Retrieved 2015-05-19.
- ^ Köster LS, Carbon M, Correll CU (December 2014). "Emerging drugs for schizophrenia: an update". Expert Opinion on Emerging Drugs. 19 (4): 511–31. doi:10.1517/14728214.2014.958148. PMID 25234340. S2CID 42729570.
- ^ "Drug Development in Schizophrenia: Summary and Table". Pharmaceutical Medicine. 28 (5): 265–271. 2014. doi:10.1007/s40290-014-0070-6. ISSN 1178-2595. S2CID 8513976.
- ^ "Reviva Product Pipeline". Reviva Pharmaceuticals. Retrieved 2023-07-19.
- ^ a b c d e Cantillon M, Prakash A, Alexander A, Ings R, Sweitzer D, Bhat L (2017). "Dopamine serotonin stabilizer RP5063: A randomized, double-blind, placebo-controlled multicenter trial of safety and efficacy in exacerbation of schizophrenia or schizoaffective disorder". Schizophrenia Research. 189: 126–133. doi:10.1016/j.schres.2017.01.043. PMID 28215471. S2CID 19499381.
- ^ a b Rajagopal L, Kwon S, Huang M, Michael E, Bhat L, Cantillon M, Meltzer HY (2017). "RP5063, an atypical antipsychotic drug with a unique pharmacologic profile, improves declarative memory and psychosis in mouse models of schizophrenia". Behavioural Brain Research. 332: 180–199. doi:10.1016/j.bbr.2017.02.036. PMID 28373127. S2CID 13047310.
- ^ a b c d Marc Cantillon, M.D., Mike Li, MS, Sarath Kanekal, Ph.D., DABT, RAC, Robert M.J. Ings, Ph.D., Grace Tung, RAC, Laxminarayan Bhat (2013). "Refresh: A Phase 2 RP5063 Efficacy and Safety in Schizophrenia and Schizoaffective Disorder" (PDF). American Society of Clinical Psychopharmacology. Retrieved 2015-05-19.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ^ Cantillon M, Ings R, Bhat L (2018). "Initial Clinical Experience of RP5063 Following Single Doses in Normal Healthy Volunteers and Multiple Doses in Patients with Stable Schizophrenia". Clinical & Translational Science. 11 (4): 387–396. doi:10.1111/cts.12545. PMC 6039200. PMID 29637739.
- ^ "Reviva RECOVER Phase III Trial Topline Results" (PDF). Reviva Pharmaceuticals. Retrieved 2023-11-02.
- ^ Klein Herenbrink C, Verma R, Lim HD, Kopinathan A, Keen A, Shonberg J, Draper-Joyce CJ, Scammells PJ, Christopoulos A, Javitch JA, Capuano B, Shi L, Lane JR (2019). "Molecular Determinants of the Intrinsic Efficacy of the Antipsychotic Aripiprazole". ACS Chemical Biology. 14 (8): 1780–1792. doi:10.1021/acschembio.9b00342. PMC 7365685. PMID 31339684.
- ^ Chen Z, Fan L, Wang H, Yu J, Lu D, Qi J, Nie F, Luo Z, Liu Z, Cheng J, Wang S (2021). "Structure-based design of a novel third-generation antipsychotic drug lead with potential antidepressant properties". Nature Neuroscience. 25 (1): 39–49. doi:10.1038/s41593-021-00971-w. PMID 34887590. S2CID 256840074.
- ^ Weiss C, Weiller E, Baker RA, Duffy RA, Gwin KK, Zhang P, McQuade RD (2018). "The effects of brexpiprazole and aripiprazole on body weight as monotherapy in patients with schizophrenia and as adjunctive treatment in patients with major depressive disorder: an analysis of short-term and long-term studies". Int Clin Psychopharmacol. 33 (5): 255–260. doi:10.1097/YIC.0000000000000226. PMC 6078484. PMID 29878915.
- ^ Earley W, Durgam S, Lu K, Laszlovszky I, Debelle M, Kane JM (2017). "Safety and tolerability of cariprazine in patients with acute exacerbation of schizophrenia: a pooled analysis of four phase II/III randomized, double-blind, placebo-controlled studies". Int Clin Psychopharmacol. 32 (6): 319–328. doi:10.1097/YIC.0000000000000187. PMC 5625952. PMID 28692485.
External links
- CS1 maint: multiple names: authors list
- Articles with short description
- Short description is different from Wikidata
- Drugs with non-standard legal status
- Articles without EBI source
- Multiple chemicals in Infobox drug
- Multiple chemicals in an infobox that need indexing
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- D2-receptor agonists
- Dopamine agonists
- 5-HT1A agonists
- 5-HT2A agonists
- 5-HT2B antagonists
- 5-HT2C antagonists
- 5-HT6 antagonists
- 5-HT7 antagonists
- Atypical antipsychotics
- Benzoxazines
- Chloroarenes
- Experimental drugs developed for schizophrenia
- Piperazines
- Serotonin reuptake inhibitors
- Experimental antidepressants