Tardive dyskinesia
| Tardive dyskinesia | |
|---|---|
| Other names: Linguofacial dyskinesia, tardive dystonia, tardive oral dyskinesia[1] | |
 | |
| Tardive dyskinesia is believed to involve the neurotransmitter dopamine. | |
| Pronunciation | |
| Specialty | Neurology, psychiatry |
| Symptoms | Involuntary, repetitive body movements[1] |
| Causes | Neuroleptic medications (antipsychotics, metoclopramide)[1][2] |
| Diagnostic method | Based on symptoms after ruling out other potential causes[1] |
| Differential diagnosis | Huntington's disease, cerebral palsy, Tourette syndrome, dystonia[2] |
| Prevention | Using lowest possible dose of neuroleptic medication[3] |
| Treatment | Stopping neuroleptic medication if possible, switching to clozapine[1] |
| Medication | Valbenazine, tetrabenazine, botulinum toxin[1][4] |
| Prognosis | Variable[1] |
| Frequency | 20% (atypical antipsychotics) 30% (typical antipsychotics)[5] |
Tardive dyskinesia (TD) is a disorder that results in involuntary, repetitive body movements, which may include grimacing, sticking out the tongue, or smacking the lips.[1] Additionally, there may be rapid jerking movements or slow writhing movements.[1] In about 20% of people with TD, the disorder interferes with daily functioning.[3]
Tardive dyskinesia occurs in some people as a result of long-term use of dopamine-receptor-blocking medications such as antipsychotics and metoclopramide.[1][2] These medications are usually used for mental illness but may also be given for gastrointestinal or neurological problems.[1] The condition typically develops only after months to years of use.[1][3] The diagnosis is based on the symptoms after ruling out other potential causes.[1]
Efforts to prevent the condition include either using the lowest possible dose or discontinuing use of neuroleptics.[3] Treatment includes stopping the neuroleptic medication if possible or switching to clozapine.[1] Other medications such as valbenazine, tetrabenazine, or botulinum toxin may be used to lessen the symptoms.[1][4] With treatment, some see a resolution of symptoms, while others do not.[1]
Rates in those on atypical antipsychotics are about 20%, while those on typical antipsychotics have rates of about 30%.[5] The risk of acquiring the condition is greater in older people.[3] The term "tardive dyskinesia" first came into use in 1964.[3]
Signs and symptoms
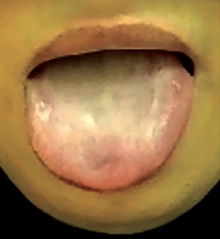
Tardive dyskinesia is characterized by repetitive, involuntary movements. Some examples of these types of involuntary movements include:[6]
- Grimacing
- Tongue movements
- Lip smacking
- Lip puckering
- Pursing of the lips
- Excessive eye blinking
- Rapid, involuntary movements of the limbs, torso, and fingers may also occur.[7]
In some cases, an individual's legs can be so affected that walking becomes difficult or impossible.[8] These symptoms are the opposite of people who are diagnosed with Parkinson's disease. People with Parkinson's have difficulty moving, whereas people with tardive dyskinesia have difficulty not moving.[9]
Respiratory irregularity, such as grunting and difficulty breathing, is another symptom associated with tardive dyskinesia, although studies have shown that the rate of people affected is relatively low.[10]
Tardive dyskinesia is often misdiagnosed as a mental illness rather than a neurological disorder,[11] and as a result, people are prescribed neuroleptic drugs, which increase the probability that the person will develop a severe and disabling case, and shortening the typical survival period.[12]
Other closely related neurological disorders have been recognized as variants of tardive dyskinesia. Tardive dystonia is similar to standard dystonia but permanent. Tardive akathisia involves painful feelings of inner tension and anxiety and a compulsive drive to move the body. In some extreme cases, afflicted individuals experience so much internal torture that they lose their ability to sit still. Tardive tourettism is a tic disorder featuring the same symptoms as Tourette syndrome. The two disorders are extremely close in nature and often can only be differentiated by the details of their respective onsets. Tardive myoclonus, a rare disorder, presents as brief jerks of muscles in the face, neck, trunk, and extremities.[9]
"AIMS Examination": This test is used when psychotropic medications have been prescribed because people sometimes develop tardive dyskinesia due to prolonged use of antipsychotic medications. The Abnormal Involuntary Movement Scale (AIMS) examination is a test used to identify the symptoms of tardive dyskinesia (TD). The test is not meant to tell whether there is an absence or presence of tardive dyskinesia. It just scales to the level of symptoms indicated by the actions observed. The levels range from none to severe. The AIMS examination was constructed in the 1970s to measure involuntary facial, trunk, and limb movements. It is best to do this test before and after the administration of the psychotropic drugs. Taking the AIMS consistently can help to track severity of TD over time.[13][14]
Causes
Tardive dyskinesia was first described in the 1950s shortly after the introduction of chlorpromazine and other antipsychotic drugs.[15] However, the exact mechanism of the disorder remains largely uncertain. The most compelling line of evidence suggests that tardive dyskinesia may result primarily from neuroleptic-induced dopamine supersensitivity in the nigrostriatal pathway, with the D2 dopamine receptor being most affected. Neuroleptics act primarily on this dopamine system, and older neuroleptics, which have greater affinity for the D2 binding site, are associated with high risk for tardive dyskinesia.[16] The D2 hypersensitivity hypothesis is also supported by evidence of a dose–response relationship, withdrawal effects, studies on D2 agonists and antagonists, animal studies, and genetic polymorphism research.[16]
Given similar doses of the same neuroleptic, differences among individuals still exist in the likelihood of developing tardive dyskinesia. Such individual differences may be due to genetic polymorphisms, which code for D2 receptor binding site affinity, or prior exposure to environmental toxins. Decreased functional reserve or cognitive dysfunction, associated with aging, mental retardation, alcohol and drug abuse, or traumatic head injuries, has also been shown to increase risk of developing the disorder among those treated with neuroleptics.[16] Antipsychotic drugs can sometimes camouflage the signs of tardive dyskinesia from occurring in the early stages; this can happen from the individual having an increased dose of an antipsychotic drug. Often the symptoms of tardive dyskinesia are not apparent until the individual comes off of the antipsychotic drugs; however, when tardive dyskinesia worsens, the signs become visible.[17]
Other dopamine antagonists and antiemetics can cause tardive dyskinesia, such as metoclopramide and promethazine, used to treat gastrointestinal disorders. Atypical antipsychotics are considered lower-risk for causing TD than their typical counterparts with their relative rates of TD of 13.1% and 32.4% respectively in short-term trials with haloperidol being the main typical antipsychotic utilised in said trials.[18] Quetiapine and clozapine are considered the lowest risk agents for precipitating TD.[18] From 2008, there have been reported cases of the anti-psychotic medication aripiprazole, a partial agonist at D2 receptors, leading to tardive dyskinesia.[19] As of 2013, reports of tardive dyskinesia in aripiprazole have grown in number.[20] The available research seems to suggest that the concurrent prophylactic use of a neuroleptic and an antiparkinsonian drug is useless to avoid early extrapyramidal side-effects and may render the person more sensitive to tardive dyskinesia. Since 1973 the use of these drugs has been found to be associated with the development of tardive dyskinesia.[21][22]
Risk factors
An increased risk of tardive dyskinesia has been associated with smoking in some studies,[23][24] although a negative study does exist.[25] There seems to be a cigarette smoke-exposure-dependent risk for TD in people who are antipsychotic-treated .[26] Elderly peoples are also at a heightened risk for developing TD,[6] as are females and those with organic brain injuries or diabetes mellitus and those with the negative symptoms of schizophrenia.[18] TD is also more common in those that experience acute neurological side effects from antipsychotic drug treatment.[18] Racial discrepancies in TD rate also exist, with Africans and African Americans having higher rates of TD after exposure to antipsychotics.[6] Certain genetic risk factors for TD have been identified including polymorphisms in the genes encoding the D3, 5-HT2A and 5-HT2C receptors.[27]
Diagnosis
The diagnosis is based on the symptoms after ruling out other potential causes.[1] The AIMS scale (Abnormal Involuntary Movement Scale) may be used to measure the severity.[28] There is generally normal neuro imaging.[28]
Prevention
Prevention of tardive dyskinesia is achieved by using the lowest effective dose of a neuroleptic for the shortest time. However, with diseases of chronic psychosis such as schizophrenia, this strategy must be balanced with the fact that increased dosages of neuroleptics are more beneficial in preventing recurrence of psychosis. If tardive dyskinesia is diagnosed, the causative drug should be discontinued. Tardive dyskinesia may persist after withdrawal of the drug for months, years or even permanently.[29][30] Some studies suggest that physicians should consider using atypical antipsychotics as a substitute to typical antipsychotics for people requiring medication. These agents are associated with fewer neuromotor side effects and a lower risk of developing tardive dyskinesia.[31]
Studies have tested the use of melatonin, high dosage vitamins, and different antioxidants in concurrence with antipsychotic drugs (often used to treat schizophrenia) as a way of preventing and treating tardive dyskinesia. Although further research is needed, studies reported a much lower percentage of individuals developing tardive dyskinesia than the current rate of people for those taking antipsychotic drugs.[32] Tentative evidence supports the use of vitamin E for prevention.[33]
Treatment
Valbenazine was approved by the FDA for tardive dyskinesia in April 2017.[34] Tetrabenazine, which is a dopamine depleting drug, is sometimes used to treat tardive dyskinesia and other movement disorders (e.g. Huntington's chorea).[7] Deutetrabenazine, an isotopic isomer of tetrabenazine, was approved by the FDA for tardive dyskinesia in August 2017.[35] Vitamin B6 has been reported to be an effective treatment for TD in two randomised double-blind placebo-controlled trials,[36][37] but the overall evidence for its effectiveness is considered "weak."[38] Clonidine may also be useful in the treatment of TD, although dose-limiting hypotension and sedation may hinder its usage.[39] Botox injections are used for minor focal dystonia, but not in more advanced tardive dyskinesia.[6] As of 2018 evidence is insufficient to support the use of benzodiazepines, baclofen, progabide, sodium valproate, gaboxadol, or calcium channel blockers (e.g. diltiazem).[40][41][42]
Epidemiology
Tardive dyskinesia most commonly occurs in people with psychiatric conditions who are treated with antipsychotic medications for many years. The average rate of people affected has been estimated to be around 30% for individuals taking antipsychotic medication, such as that used to treat schizophrenia.[43] A study being conducted at the Yale University School of Medicine has estimated that "32% of people develop persistent tics after 5 years on major tranquilizers, 57% by 15 years, and 68% by 25 years."[44] More drastic data was found during a longitudinal study conducted on individuals 45 years of age and older who were taking antipsychotic drugs. According to this research study, 26% of people developed tardive dyskinesia after just one year on the medication. Another 60% of this at-risk group developed the disorder after 3 years, and 23% developed severe cases of tardive dyskinesia within 3 years.[45] According to these estimates, the majority of people will eventually develop the disorder if they remain on the drugs long enough.[46]
Elderly people are more prone to develop tardive dyskinesia, and elderly women are more at-risk than elderly men. The risk is much lower for younger men and women, and also more equal across the sexes.[47] People who have undergone electroconvulsive therapy or have a history of diabetes or alcohol abuse also have a higher risk of developing tardive dyskinesia.[31]
Several studies have recently been conducted comparing the number of people affected of tardive dyskinesia with second generation, or more modern, antipsychotic drugs to that of first generation drugs. The newer antipsychotics appear to have a substantially reduced potential for causing tardive dyskinesia. However, some studies express concern that the number of people affected has decreased far less than expected, cautioning against the overestimation of the safety of modern antipsychotics.[32][48]
A physician can evaluate and diagnose a person with tardive dyskinesia by conducting a systematic examination. The physician should ask the person to relax, and look for symptoms like facial grimacing, eye or lip movements, tics, respiratory irregularities, and tongue movements. In some cases, people experience nutritional problems, so a physician can also look for a gain or loss in weight.[31]
Apart from the underlying psychiatric disorder, tardive dyskinesia may cause afflicted people to become socially isolated. It also increases the risk of body dysmorphic disorder (BDD) and can even lead to suicide. Emotional or physical stress can increase the severity of dyskinetic movements, whereas relaxation and sedation have the opposite effect.[49]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 "Tardive dyskinesia". rarediseases.info.nih.gov. 1 June 2017. Archived from the original on 18 June 2017. Retrieved 10 June 2017.
- ↑ 2.0 2.1 2.2 "Tardive Dyskinesia". NORD (National Organization for Rare Disorders). 2015. Archived from the original on 28 August 2017. Retrieved 11 June 2017.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Vijayakumar, D; Jankovic, J (May 2016). "Drug-Induced Dyskinesia, Part 2: Treatment of Tardive Dyskinesia". Drugs. 76 (7): 779–87. doi:10.1007/s40265-016-0568-1. PMID 27091214.
- ↑ 4.0 4.1 "Tardive Dyskinesia Information Page". National Institute of Neurological Disorders and Stroke. Archived from the original on 2017-07-04.
- ↑ 5.0 5.1 Carbon, M; Hsieh, CH; Kane, JM; Correll, CU (March 2017). "Tardive Dyskinesia Prevalence in the Period of Second-Generation Antipsychotic Use: A Meta-Analysis". The Journal of Clinical Psychiatry. 78 (3): e264–e278. doi:10.4088/jcp.16r10832. PMID 28146614.
- ↑ 6.0 6.1 6.2 6.3 "Tardive Dyskinesia". Medscape Reference. WebMD. 9 February 2012. Archived from the original on 3 December 2013. Retrieved 25 November 2013.
- ↑ 7.0 7.1 National Institutes of Health, National Institute of Neurological Disorders and Stroke. (2011). Ninds tardive dyskinesia information page. Retrieved from website: "Archived copy". Archived from the original on 2012-04-27. Retrieved 2012-05-01.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Nasrallah, H. (2003, Sept). Tardive dyskinesia. Retrieved from "Archived copy". Archived from the original on 2012-04-21. Retrieved 2012-05-01.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ 9.0 9.1 Duke University Health System. (2010, May 17). Additional movement disorders. Retrieved from "Treatments for Adults | Duke Health". Archived from the original on 2012-05-24. Retrieved 2012-05-01.
- ↑ Yassa, R.; Lal, S. (1986). "Respiratory irregularity and tardive dyskinesia a prevalence study". Acta Psychiatrica Scandinavica. 73 (5): 506–10. doi:10.1111/j.1600-0447.1986.tb02717.x. PMID 2875609.
- ↑ Yassa, Ramzy; Jones, Barry D. (1985). "Complications of tardive dyskinesia: A review". Psychosomatics. 26 (4): 305–7, 310, 312–3. doi:10.1016/S0033-3182(85)72863-0. PMID 2859630.
- ↑ Dean, C. E.; Thuras, P. D. (2009). "Mortality and tardive dyskinesia: Long-term study using the US National Death Index". The British Journal of Psychiatry. 194 (4): 360–4. doi:10.1192/bjp.bp.108.049395. PMID 19336789.
- ↑ Encyclopedia of Mental Disorders
- ↑ Montana State Hospital
- ↑ Baldessarini, Ross J. (1988). "A summary of current knowledge of tardive dyskinesia". L'Encéphale. 14 (Spec Issue): 263–8. PMID 2905654.
- ↑ 16.0 16.1 16.2 Hoerger, Michael. "The primacy of neuroleptic-Induced D2 receptor hypersensitivity in tardive dyskinesia". Archived from the original on 2008-01-18.[unreliable medical source?]
- ↑ Ross, Colin A; Read, John (2004). "Antipsychotic medication: myths and facts". In Read, John; Mosher, Loren R.; Bentall, Richard P. (eds.). Models of Madness: Psychological, Social and Biological Approaches to Schizophrenia. London: Brunner-Routledge. pp. 101–14. ISBN 978-1-58391-906-4.
{{cite book}}:|access-date=requires|url=(help);|archive-url=requires|url=(help); Unknown parameter|chapterurl=ignored (help) - ↑ 18.0 18.1 18.2 18.3 Rana, Abdul Qayyum; Chaudry, Zishan; Blanchet, Pierre (2013). "New and emerging treatments for symptomatic tardive dyskinesia". Drug Design, Development and Therapy. 7: 1329–40. doi:10.2147/DDDT.S32328. PMC 3825689. PMID 24235816.
- ↑ Alexander, C; Power, P (2008). "A case of aripiprazole and tardive dyskinesia". Journal of Psychopharmacology. 23 (2): 214–5. doi:10.1177/0269881108089591. PMID 18515468.
- ↑ Alexander, J (Mar 2013). "Aripiprazole induced tardive dyskinesia-accruing evidence". Aust N Z J Psychiatry. 47 (3): 289–90. doi:10.1177/0004867412461058. PMID 22990431.
- ↑ Crane, GE (1973). "Is tardive dyskinesia a drug effect?". The American Journal of Psychiatry. 130 (9): 1043–44. doi:10.1176/ajp.130.9.1043-a. PMID 4727768.
- ↑ Crane, George E (1973). "Rapid Reversal of Tardive Dyskinesia". American Journal of Psychiatry. 130 (10): 1159. doi:10.1176/ajp.130.10.1159. PMID 4728916.
- ↑ Yassa, R; Lal, S; Korpassy, A; Ally, J (January 1987). "Nicotine exposure and tardive dyskinesia". Biological Psychiatry. 22 (1): 67–72. doi:10.1016/0006-3223(87)90131-4. PMID 3790642.
- ↑ Chong, SA; Tan, EC; Tan, CH; Mythily (May 2003). "Smoking and tardive dyskinesia: lack of involvement of the CYP1A2 gene". J Psychiatry Neurosci. 28 (3): 185–89. PMC 161742. PMID 12790158.
- ↑ Zhang, XY; Yu, YQ; Sun, S; Zhang, X; Li, W; Xiu, MH; Chen da, C; Yang, FD; Zhu, F; Kosten, TA; Kosten, TR (August 2011). "Smoking and tardive dyskinesia in male patients with chronic schizophrenia". Progress in Neuro-Psychopharmacology and Biological Psychiatry. 35 (7): 1765–69. doi:10.1016/j.pnpbp.2011.06.006. PMID 21723906.
- ↑ Diehl, A; Reinhard, I; Schmitt, A; Mann, K; Gattaz, WF (January 2009). "Does the degree of smoking effect the severity of tardive dyskinesia? A longitudinal clinical trial". European Psychiatry. 24 (1): 33–40. doi:10.1016/j.eurpsy.2008.07.007. PMID 18774276.
- ↑ Segman, Ronnen H; Heresco-Levy, Uriel; Finkel, Boris; Inbar, Roi; Neeman, Tal; Schlafman, Michael; Dorevitch, Abe; Yakir, Avraham; Lerner, Arturo; Goltser, Tanya; Shelevoy, Aida; Lerer, Bernard (2000). "Association between the serotonin 2C receptor gene and tardive dyskinesia in chronic schizophrenia: Additive contribution of 5-HT2C ser and DRD3 gly alleles to susceptibility". Psychopharmacology. 152 (4): 408–13. doi:10.1007/s002130000521. PMID 11140333.
- ↑ 28.0 28.1 Vasan, Sarayu; Padhy, Ranjit K. (2022). "Tardive Dyskinesia". StatPearls. StatPearls Publishing. Archived from the original on 3 October 2021. Retrieved 15 May 2022.
- ↑ Rauchverger, B; Isakov, V; Jabarin, M (2007). "Olanzapine-Induced Tardive Dystonia Successfully Treated by Tetrabenazine". Journal of Neuropsychiatry. 19 (4): 484–5. doi:10.1176/appi.neuropsych.19.4.484-a. PMID 18070868.
- ↑ Fernandez, Hubert H; Friedman, Joseph H (2003). "Classification and Treatment of Tardive Syndromes". The Neurologist. 9 (1): 16–27. doi:10.1097/01.nrl.0000038585.58012.97. PMID 12801428.
- ↑ 31.0 31.1 31.2 Saltz, Bruce L.; Robinson, Delbert G.; Woerner, Margaret G. (2004). "Recognizing and Managing Antipsychotic Drug Treatment Side Effects in the Elderly". Primary Care Companion to the Journal of Clinical Psychiatry. 6 (Suppl 2): 14–9. PMC 487007. PMID 16001096.
- ↑ 32.0 32.1 Lerner, Vladimir (2011). "Antioxidants as a Treatment and Prevention of Tardive Dyskinesia". In Ritsner, Michael S. (ed.). Handbook of Schizophrenia Spectrum Disorders, Volume III. pp. 109–34. doi:10.1007/978-94-007-0834-1_6. ISBN 978-94-007-0833-4.
{{cite book}}:|access-date=requires|url=(help);|archive-url=requires|url=(help); Unknown parameter|chapterurl=ignored (help) - ↑ Soares-Weiser, K; Maayan, N; Bergman, H (17 January 2018). "Vitamin E for antipsychotic-induced tardive dyskinesia". The Cochrane Database of Systematic Reviews. 1: CD000209. doi:10.1002/14651858.CD000209.pub3. PMC 6491331. PMID 29341067.
- ↑ Commissioner, Office of the. "Press Announcements - FDA approves first drug to treat tardive dyskinesia". www.fda.gov. Archived from the original on 12 April 2017. Retrieved 12 April 2017.
- ↑ "Drug Approval Package: Austedo (deutetrabenazine) Tablets". www.accessdata.fda.gov. Archived from the original on 2020-07-08. Retrieved 2018-09-14.
- ↑ Lerner, V; Miodownik, C; Kaptsan, A; Cohen, H; Matar, M; Loewenthal, U; Kotler, M (September 2001). "Vitamin B6 in the Treatment of Tardive Dyskinesia: A Double-Blind, Placebo-Controlled, Crossover Study" (PDF). The American Journal of Psychiatry. 158 (9): 1511–14. doi:10.1176/appi.ajp.158.9.1511. PMID 11532741. Archived (PDF) from the original on 2013-12-03. Retrieved 2013-11-25.
- ↑ Lerner, V; Miodownik, C; Kaptsan, A; Bersudsky, Y; Libov, I; Sela BA; Witztum, E (November 2007). "Vitamin B6 treatment for tardive dyskinesia: a randomized, double-blind, placebo-controlled, crossover study". Journal of Clinical Psychiatry. 68 (11): 1648–54. doi:10.4088/jcp.v68n1103. PMID 18052557.
- ↑ Adelufosi, Adegoke Oloruntoba; Abayomi, Olukayode; Ojo, Tunde Massey-Ferguson (13 April 2015). "Pyridoxal 5 phosphate for neuroleptic-induced tardive dyskinesia". Cochrane Database of Systematic Reviews (4): CD010501. doi:10.1002/14651858.CD010501.pub2. PMID 25866243.
- ↑ Aia, Pratibha G; Revuelta, Gonzalo J; Cloud, Leslie J; Factor, Stewart A (2011). "Tardive Dyskinesia". Current Treatment Options in Neurology. 13 (3): 231–41. doi:10.1007/s11940-011-0117-x. PMID 21365202.
- ↑ Bergman, H; Bhoopathi, PS; Soares-Weiser, K (20 January 2018). "Benzodiazepines for antipsychotic-induced tardive dyskinesia". The Cochrane Database of Systematic Reviews. 1: CD000205. doi:10.1002/14651858.CD000205.pub3. PMC 6491128. PMID 29352477.
- ↑ Alabed, S; Latifeh, Y; Mohammad, HA; Bergman, H (17 April 2018). "Gamma-aminobutyric acid agonists for antipsychotic-induced tardive dyskinesia". The Cochrane Database of Systematic Reviews. 4: CD000203. doi:10.1002/14651858.CD000203.pub4. PMC 6513215. PMID 29663328.
- ↑ Essali, Adib; Soares-Weiser, Karla; Bergman, Hanna; Adams, Clive E (26 March 2018). "Calcium channel blockers for antipsychotic-induced tardive dyskinesia". Cochrane Database of Systematic Reviews. 3: CD000206. doi:10.1002/14651858.CD000206.pub4. PMC 6494274. PMID 29578611.
- ↑ Llorca, Pierre-Michel; Chereau, Isabelle; Bayle, Frank-Jean; Lancon, Christophe (2002). "Tardive dyskinesias and antipsychotics: A review". European Psychiatry. 17 (3): 129–38. doi:10.1016/S0924-9338(02)00647-8. PMID 12052573.
{{cite journal}}: Unknown parameter|displayauthors=ignored (help) - ↑ Glenmullen, Joseph (2001). Prozac Backlash: Overcoming the Dangers of Prozac, Zoloft, Paxil, and Other Antidepressants With Safe, Effective Alternatives. New York: Simon & Schuster. p. 38. ISBN 978-0-7432-0062-2.[unreliable medical source?] ::referring to Glazer, William M.; Morgenstern, Hal; Doucette, John T. (1993). "Predicting the long-term risk of tardive dyskinesia in outpatients maintained on neuroleptic medications". Journal of Clinical Psychiatry. 54 (4): 133–9. PMID 8098030.
- ↑ Jeste, Dilip V.; Caligiuri, Michael P.; Paulsen, Jane S.; Heaton, Robert K.; Lacro, Jonathan P.; Harris, M. Jackuelyn; Bailey, Anne; Fell, Robert L.; McAdams, Lou Ann (1995). "Risk of Tardive Dyskinesia in Older Patients: A Prospective Longitudinal Study of 266 Outpatients". Archives of General Psychiatry. 52 (9): 756–65. doi:10.1001/archpsyc.1995.03950210050010. PMID 7654127.
{{cite journal}}: Unknown parameter|displayauthors=ignored (help) - ↑ Whitaker, Robert (2002). Mad in America: Bad Science, Bad Medicine, and the Enduring Mistreatment of the Mentally Ill. Perseus.[page needed]
- ↑ Marshall, DL; Hazlet, TK; Gardner, JS; Blough, DK (2002). "Neuroleptic drug exposure and incidence of tardive dyskinesia: A records-based case-control study". Journal of Managed Care Pharmacy. 8 (4): 259–65. doi:10.18553/jmcp.2002.8.4.259. PMID 14613418.
{{cite journal}}: Unknown parameter|displayauthors=ignored (help) - ↑ Tarsy, Daniel; Lungu, Codrin; Baldessarini, Ross J. (2011). "Epidemiology of tardive dyskinesia before and during the era of modern antipsychotic drugs". In Vinken, P. J.; Bruyn, G. W. (eds.). Handbook of Clinical Neurology. Hyperkinetic Movement Disorders. Vol. 100. pp. 601–16. doi:10.1016/B978-0-444-52014-2.00043-4. ISBN 978-0-444-52014-2. PMID 21496610.
- ↑ Jeste, Dilip V.; Caligiuri, Michael P. (1993). "Tardive Dyskinesia". Schizophrenia Bulletin. 19 (2): 303–15. doi:10.1093/schbul/19.2.303. PMID 8100643.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 maint: archived copy as title
- All articles lacking reliable references
- Articles lacking reliable references from June 2012
- Articles with invalid date parameter in template
- CS1 errors: unsupported parameter
- CS1 errors: access-date without URL
- CS1 errors: archive-url
- Wikipedia articles needing page number citations from June 2012
- Tardive dyskinesia
- Neurological disorders
- Symptoms and signs: Nervous system
- Syndromes
- RTT
- RTTNEURO