Solabegron
 | |
| Clinical data | |
|---|---|
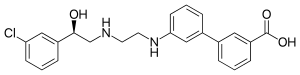
| Other names | 3-[3-[2-[[(2R)-2-(3-chlorophenyl)-2-hydroxyethyl]amino]ethylamino]phenyl]benzoic acid |
| ATC code |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C23H23ClN2O3 |
| Molar mass | 410.90 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Solabegron (code name GW-427,353) is a drug which acts as a selective agonist for the β3 adrenergic receptor. It is being developed for the treatment of overactive bladder and irritable bowel syndrome.[1][2][3] It has been shown to produce visceral analgesia by releasing somatostatin from adipocytes.[4][5]
Solabegron was discovered by GlaxoSmithKline and acquired by AltheRx in March 2011. Solabegron relaxes the bladder smooth muscle by stimulating β3 adrenoceptors, a novel mechanism compared with older established drug treatments for overactive bladder syndrome such as the anticholinergic agents. Astellas Pharma has developed the first commercially available β3 adrenergic receptor, mirabegron, which is now licensed in Japan[6] and the U.S.[7] exclusively for treatment of overactive bladder syndrome.
A Phase II study of solabegron for overactive bladder (OAB) looked at 258 patients with moderate-to-severe incontinence experiencing an average of 4.5 wet episodes per day. Results demonstrated a statistically significant improvement with solabegron as compared with placebo, as measured by the percentage reduction of the number of wet episodes and the absolute number of daily voids.
A Phase II study for irritable bowel syndrome (IBS) evaluated 102 patients with IBS. Solabegron demonstrated significant reduction in pain associated with the disorder and a trend for greater improvement in the quality of life, when compared with a placebo.
Both Phase II studies indicated a tolerability profile for solabegron that was similar to placebo. This drug does not bind to acetylcholine receptors so side effects are expected to be minor.
AltheRx is currently preparing to advance solabegron into a large clinical study in OAB.
References
- ^ Hicks A, McCafferty GP, Riedel E, Aiyar N, Pullen M, Evans C, et al. (October 2007). "GW427353 (solabegron), a novel, selective beta3-adrenergic receptor agonist, evokes bladder relaxation and increases micturition reflex threshold in the dog". The Journal of Pharmacology and Experimental Therapeutics. 323 (1): 202–9. doi:10.1124/jpet.107.125757. PMID 17626794. S2CID 19281697.
- ^ Grudell AB, Camilleri M, Jensen KL, Foxx-Orenstein AE, Burton DD, Ryks MD, et al. (May 2008). "Dose-response effect of a beta3-adrenergic receptor agonist, solabegron, on gastrointestinal transit, bowel function, and somatostatin levels in health". American Journal of Physiology. Gastrointestinal and Liver Physiology. 294 (5): G1114-9. doi:10.1152/ajpgi.00051.2008. PMID 18372395.
- ^ Kelleher DL, Hicks KJ, Cox DS, et al. (2008). "Randomized, double-blind, placebo (PLA)-controlled, crossover study to evaluate efficacy and safety of the beta 3-adrenergic receptor agonist solabegron (SOL) in patients with irritable bowel syndrome (IBS)". Neurogastroenterol Motil. 20 (Suppl 2): 131.
- ^ Cellek S, Thangiah R, Bassil AK, Campbell CA, Gray KM, Stretton JL, et al. (July 2007). "Demonstration of functional neuronal beta3-adrenoceptors within the enteric nervous system". Gastroenterology. 133 (1): 175–83. doi:10.1053/j.gastro.2007.05.009. PMID 17631141.
- ^ Schemann M, Hafsi N, Michel K, Kober OI, Wollmann J, Li Q, et al. (January 2010). "The beta3-adrenoceptor agonist GW427353 (Solabegron) decreases excitability of human enteric neurons via release of somatostatin". Gastroenterology. 138 (1): 266–74. doi:10.1053/j.gastro.2009.09.046. PMID 19786030.
- ^ Gras J (January 2012). "Mirabegron for the treatment of overactive bladder". Drugs of Today. 48 (1): 25–32. doi:10.1358/dot.2012.48.1.1738056. PMID 22384458.
- ^ "New Drug Approvals 2012 - Pt. XIV - Mirabegron (Myrbetriq)". The ChEMBL-og.
- Articles with short description
- Short description matches Wikidata
- Drugs not assigned an ATC code
- Articles with changed ChemSpider identifier
- Articles with changed InChI identifier
- Chemical pages without DrugBank identifier
- Multiple chemicals in Infobox drug
- Chemicals using indexlabels
- Chemical articles with multiple CAS registry numbers
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Anilines
- Benzoic acids
- Beta3-adrenergic agonists
- Biphenyls
- Chloroarenes
- Phenylethanolamines