Etilefrine
 | |
| Clinical data | |
|---|---|
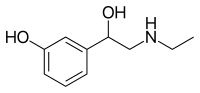
| Other names | (2-ethylamino-1-(3'-hydroxy-phenyl)ethanol |
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.010.829 |
| Chemical and physical data | |
| Formula | C10H15NO2 |
| Molar mass | 181.235 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Etilefrine is a cardiac stimulant used as an antihypotensive. It is a sympathomimetic amine of the 3-hydroxy-phenylethanolamine series used in treating orthostatic hypotension of neurological, cardiovascular, endocrine or metabolic origin. Intravenous infusion of this compound increases cardiac output, stroke volume, venous return and blood pressure in man and experimental animals, suggesting stimulation of both α and β adrenergic receptors.[1][2][3][4][5] However, in vitro studies indicate that etilefrine has a much higher affinity for β1 (cardiac) than for β2 adrenoreceptors.[6]
Intravenous etilefrine increases the pulse rate, cardiac output, stroke volume, central venous pressure and mean arterial pressure of healthy individuals. Peripheral vascular resistance falls during the infusion of 1–8 mg etilefrine but begins to rise at higher dosage. Marked falls in pulse rate, cardiac output, stroke volume and peripheral bloodflow, accompanied by rises in mean arterial pressure, occur when etilefrine is infused after administration of intravenous propranolol 2,5 mg. These findings indicate that etilefrine has both β1 and α1 adrenergic effects in man.
References
- ^ Nusser E, Donath H, Russ W (August 1965). "[On the circulatory action of depot-Effortil in patients with hypotonic regulation circulator disorders]". Die Medizinische Welt (in German). 32: 1824–7. PMID 5320529.
- ^ Mellander S (1966). "Comparative effects of acetylcholine, butyl-nor-synephrine (Vasculat), noradrenaline, and ethyl-adrainol (Effonti) on resistance, capacitance, and precapillary sphincter vessels and capillary filtration in cat skeletal muscle". Angiologica. 3 (2): 77–99. doi:10.1159/000157650. PMID 4380206.
- ^ von Limbourg P, Just H, Lang KF (1973). "Positive inotrope Wirkung von Etilefrinhydrochlorid (EffortilR)". Kardiol. 586: 1.
- ^ Tarnow J, Brückner JB, Eberlein HG, Patschke D, Reinecke A, Schmicke P (1973). "Experimentelle Untersuchungen zur Beeinflussung der Hämodynamik in tiefer Halothannarkose durch Dopamin, Glucagon, Effortil, Noradrenalin und Dextran". Der Anaesthesist. 22: 8–15.
- ^ Carrera AL, Aguilera AM (1973). "Algunos effectos circulatorios de la m− oxifenil etanol etilmaina y sus modificaciones por el bloqueo α y β adrenergico". Arch. Inst. Cardiol. 43. Mexico: 279–287.
- ^ Offermeier J, Dreyer AC (March 1971). "A comparison of the effects of noradrenaline, adrenaline and some phenylephrine derivatives on alpha-, beta- and beta- adrenergic receptors". South African Medical Journal = Suid-Afrikaanse Tydskrif vir Geneeskunde. 45 (10): 265–7. PMID 4396765.
External links
- "Etilefrine". PubChem.
- "Etilefrine". Kegg.
- CS1 German-language sources (de)
- Articles with short description
- Short description matches Wikidata
- ECHA InfoCard ID from Wikidata
- Drugs with no legal status
- Drugboxes which contain changes to watched fields
- Alpha-1 adrenergic receptor agonists
- Alpha-2 adrenergic receptor agonists
- Beta-adrenergic agonists
- Cardiac stimulants
- Phenylethanolamines
- Phenols