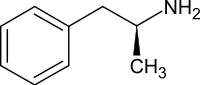
Dextroamphetamine
 | |
| Names | |
|---|---|
| Pronunciation | /ˌdɛkstroʊæmˈfɛtəmiːn/ |
| Trade names | Dexedrine, DextroStat, Metamina, Attentin, Zenzedi, others |
| Other names | D-amphetamine, dexamfetamine, dexamphetamine, (S)-amphetamine, (+)-amphetamine, dexamfetamine sulfate |
| |
| Clinical data | |
| Drug class | Amphetamine[1] |
| Main uses | ADHD, narcolepsy[2] |
| Side effects | Stomach pain, trouble sleeping, palpitations, high blood pressure, anxiety, tremor, diarrhea[1] |
| Dependence risk | Moderate-high[7][6] |
| Addiction risk | Moderate-high[7][6] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Onset of action | IR dosing: 0.5–1.5 hours[3] |
| Duration of action | IR dosing: 3–6 hours[4][5] XR dosing: 8–12 hours[6][4][5] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605027 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | By mouth: 75–100%[8] |
| Protein binding | 15–40%[9] |
| Metabolism | CYP2D6,[10] DBH,[11] FMO3[12] |
| Elimination half-life | 9–11 hours[10][13] pH-dependent: 7–34 hours[14] |
| Excretion | Kidney (45%);[15] urinary pH-dependent |
| Chemical and physical data | |
| Formula | C9H13N |
| Molar mass | 135.210 g·mol−1 |
| 3D model (JSmol) | |
| Density | 0.913 g/cm3 |
| Boiling point | 201.5 °C (394.7 °F) |
| Solubility in water | 20 mg/mL (20 °C) |
| |
| |
Dextroamphetamine (D-AMP), also known as dexamfetamine, is a medication used to treat attention deficit hyperactivity disorder (ADHD) and narcolepsy.[2] While it has been used short term for obesity, such use is no longer recommended.[1] It is taken by mouth.[2] It has also been used to improved job performance, athletic performance, and by military forces.[18]
Common side effects include stomach pain, trouble sleeping, palpitations, high blood pressure, anxiety, tremor, and diarrhea.[1] Other side effects may include cardiomyopathy, abuse, psychosis, aggression, and seizures.[1] Use in pregnancy may result in prematurity in the baby.[1] It is a central nervous system (CNS) stimulant and isomer of amphetamine.[1]
Dextroamphetamine came into medical use in 1937.[19] It is available as a generic medication.[2] In the United Kingdom 30 tablets of 20 mg costs the NHS about £80 as of 2021.[2] In the United States this amount costs about 60 USD.[20] In the United States it is a Schedule II controlled substance.[21]
Medical use
Dextroamphetamine is used to treat attention deficit hyperactivity disorder (ADHD) and narcolepsy (a sleep disorder), and is sometimes prescribed off-label for its past medical indications, such as depression and obesity.[22][23]
Dosage
In adults it is generally started at a dose of 10 mg per day and may be increased up to 60 mg per day.[2] In children a top dose of 20 to 40 mg per day is generally used.[2]
-
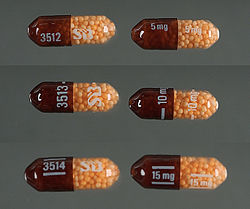
Dextroamphetamine 5mg tablets
-
Dexedrine Spansule 5, 10 and 15 mg capsules, a sustained-release dosage form of dextroamphetamine
Contraindications
Side effects
Overdose
Interactions
Many types of substances are known to interact with amphetamine, resulting in altered drug action or metabolism of amphetamine, the interacting substance, or both.[10][24] Inhibitors of the enzymes that metabolize amphetamine (e.g., CYP2D6 and FMO3) will prolong its elimination half-life, meaning that its effects will last longer.[12][24] Amphetamine also interacts with MAOIs, particularly monoamine oxidase A inhibitors, since both MAOIs and amphetamine increase plasma catecholamines (i.e., norepinephrine and dopamine);[24] therefore, concurrent use of both is dangerous.[24] Amphetamine modulates the activity of most psychoactive drugs. In particular, amphetamine may decrease the effects of sedatives and depressants and increase the effects of stimulants and antidepressants.[24] Amphetamine may also decrease the effects of antihypertensives and antipsychotics due to its effects on blood pressure and dopamine respectively.[24] Zinc supplementation may reduce the minimum effective dose of amphetamine when it is used for the treatment of ADHD.[note 1][28]
Pharmacology
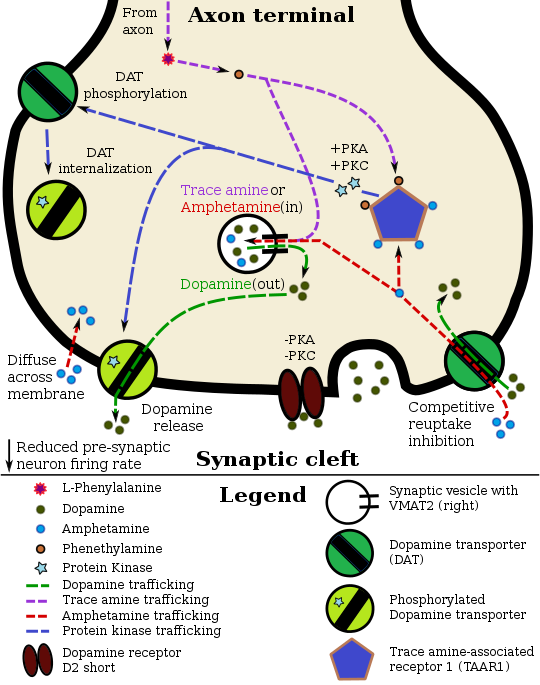
Dextroamphetamine, like other amphetamines, elicits its stimulating effects via several distinct actions: it inhibits or reverses the transporter proteins for the monoamine neurotransmitters (namely the serotonin, norepinephrine and dopamine transporters) either via trace amine-associated receptor 1 (TAAR1) or in a TAAR1 independent fashion when there are high cytosolic concentrations of the monoamine neurotransmitters[29] and it releases these neurotransmitters from synaptic vesicles via vesicular monoamine transporter 2.[30] It also shares many chemical and pharmacological properties with human trace amines, particularly phenethylamine and N-methylphenethylamine, the latter being an isomer of amphetamine produced within the human body.
Pharmacodynamics
Pharmacodynamics of amphetamine in a dopamine neuron
|
Amphetamine and its enantiomers have been identified as potent full agonists of trace amine-associated receptor 1 (TAAR1), a GPCR, discovered in 2001, that is important for regulation of monoaminergic systems in the brain.[36][37] Activation of TAAR1 increases cAMP production via adenylyl cyclase activation and inhibits the function of the dopamine transporter, norepinephrine transporter, and serotonin transporter, as well as inducing the release of these monoamine neurotransmitters (effluxion).[29][36][38] Amphetamine enantiomers are also substrates for a specific neuronal synaptic vesicle uptake transporter called VMAT2.[30] When amphetamine is taken up by VMAT2, the vesicle releases (effluxes) dopamine, norepinephrine, and serotonin, among other monoamines, into the cytosol in exchange.[30]
Dextroamphetamine (the dextrorotary enantiomer) and levoamphetamine (the levorotary enantiomer) have identical pharmacodynamics, but their binding affinities to their biomolecular targets vary.[37][39] Dextroamphetamine is a more potent agonist of TAAR1 than levoamphetamine.[37] Consequently, dextroamphetamine produces roughly three to four times more central nervous system (CNS) stimulation than levoamphetamine;[37][39] however, levoamphetamine has slightly greater cardiovascular and peripheral effects.[39]
History
Racemic amphetamine was first synthesized under the chemical name "phenylisopropylamine" in Berlin, 1887 by the Romanian chemist Lazar Edeleanu. It was not widely marketed until 1932, when the pharmaceutical company Smith, Kline & French (now known as GlaxoSmithKline) introduced it in the form of the Benzedrine inhaler for use as a bronchodilator. Notably, the amphetamine contained in the Benzedrine inhaler was the liquid free-base,[note 2] not a chloride or sulfate salt.
Three years later, in 1935, the medical community became aware of the stimulant properties of amphetamine, specifically dextroamphetamine, and in 1937 Smith, Kline, and French introduced tablets under the tradename Dexedrine.[40] In the United States, Dexedrine was approved to treat narcolepsy, attention disorders, and obesity. In Canada indications once included epilepsy and parkinsonism.[41] Dextroamphetamine was marketed in various other forms in the following decades, primarily by Smith, Kline, and French, such as several combination medications including a mixture of dextroamphetamine and amobarbital (a barbiturate) sold under the tradename Dexamyl and, in the 1950s, an extended release capsule (the "Spansule").[42] Preparations containing dextroamphetamine were also used in World War II as a treatment against fatigue.[43]
It quickly became apparent that dextroamphetamine and other amphetamines had a high potential for misuse, although they were not heavily controlled until 1970, when the Comprehensive Drug Abuse Prevention and Control Act was passed by the United States Congress. Dextroamphetamine, along with other sympathomimetics, was eventually classified as Schedule II, the most restrictive category possible for a drug with a government-sanctioned, recognized medical use.[44] Internationally, it has been available under the names AmfeDyn (Italy), Curban (US), Obetrol (Switzerland), Simpamina (Italy), Dexedrine/GSK (US & Canada), Dexedrine/UCB (United Kingdom), Dextropa (Portugal), and Stild (Spain).[45] It became popular on the mod scene in England in the early 1960s, and carried through to the Northern Soul scene in the north of England to the end of the 1970s.
In October 2010, GlaxoSmithKline sold the rights for Dexedrine Spansule to Amedra Pharmaceuticals (a subsidiary of CorePharma).[46]
The U.S. Air Force uses dextroamphetamine as one of its "go pills", given to pilots on long missions to help them remain focused and alert. Conversely, "no-go pills" are used after the mission is completed, to combat the effects of the mission and "go-pills".[47][48][49][50] The Tarnak Farm incident was linked by media reports to the use of this drug on long term fatigued pilots. The military did not accept this explanation, citing the lack of similar incidents. Newer stimulant medications or awakeness promoting agents with different side effect profiles, such as modafinil, are being investigated and sometimes issued for this reason.[48]
Society and culture
Formulations
| Brand name |
United States Adopted Name |
(D:L) ratio | Dosage form |
Marketing start date |
US consumer price data |
Sources |
|---|---|---|---|---|---|---|
| Adderall | – | 3:1 (salts) | tablet | 1996 | GoodRx Archived 26 July 2020 at the Wayback Machine | [23][59] |
| Adderall XR | – | 3:1 (salts) | capsule | 2001 | GoodRx Archived 26 July 2020 at the Wayback Machine | [23][59] |
| Mydayis | – | 3:1 (salts) | capsule | 2017 | GoodRx Archived 26 July 2020 at the Wayback Machine | [60] |
| Adzenys XR-ODT | amphetamine | 3:1 (base) | ODT | 2016 | GoodRx Archived 25 July 2020 at the Wayback Machine | [61][62] |
| Dyanavel XR | amphetamine | 3.2:1 (base) | suspension | 2015 | GoodRx Archived 26 July 2020 at the Wayback Machine | [63][64] |
| Evekeo | amphetamine sulfate | 1:1 (salts) | tablet | 2012 | GoodRx Archived 25 July 2020 at the Wayback Machine | [65] [66] |
| Dexedrine | dextroamphetamine sulfate | 1:0 (salts) | capsule | 1976 | GoodRx Archived 26 July 2020 at the Wayback Machine | [23][59] |
| Zenzedi | dextroamphetamine sulfate | 1:0 (salts) | tablet | 2013 | GoodRx Archived 26 July 2020 at the Wayback Machine | [59] |
| Vyvanse | lisdexamfetamine dimesylate | 1:0 (prodrug) | capsule | 2007 | GoodRx Archived 26 July 2020 at the Wayback Machine | [23][67] |
| tablet |
Dextroamphetamine sulfate
In the United States, immediate release (IR) formulations of dextroamphetamine sulfate are available generically as 5 mg and 10 mg tablets, marketed by Barr (Teva Pharmaceutical Industries), Mallinckrodt Pharmaceuticals, Wilshire Pharmaceuticals, Aurobindo Pharmaceutical USA and CorePharma. Previous IR tablets sold by the brand names of Dexedrine and Dextrostat have been discontinued but in 2015 IR tablets became available by the brand name Zenzedi, offered as 2.5 mg, 5 mg, 7.5 mg, 10 mg, 15 mg, 20 mg and 30 mg tablets.[68] Dextroamphetamine sulfate is also available as a controlled-release (CR) capsule preparation in strengths of 5 mg, 10 mg, and 15 mg under the brand name Dexedrine Spansule, with generic versions marketed by Barr and Mallinckrodt. A bubblegum flavored oral solution is available under the brand name ProCentra, manufactured by FSC Pediatrics, which is designed to be an easier method of administration in children who have difficulty swallowing tablets, each 5 mL contains 5 mg dextroamphetamine.[69] The conversion rate between dextroamphetamine sulfate to amphetamine free base is .728.[70]
In Australia, dexamphetamine is available in bottles of 100 instant release 5 mg tablets as a generic drug.[71] or slow release dextroamphetamine preparations may be compounded by individual chemists.[72] In the United Kingdom, it is available in 5 mg instant release sulfate tablets under the generic name dexamfetamine sulfate as well as 10 mg and 20 mg strength tablets under the brand name Amfexa. It is also available in generic dexamfetamine sulfate 5 mg/ml oral sugar-free syrup.[73] The brand name Dexedrine was available in the United Kingdom prior to UCB Pharma disinvesting the product to another pharmaceutical company (Auden Mckenzie).[74]
Lisdexamfetamine
Dextroamphetamine is the active metabolite of the prodrug lisdexamfetamine (L-lysine-dextroamphetamine), available by the brand name Vyvanse (Elvanse in the European market) (Venvanse in the Brazil market) (lisdexamfetamine dimesylate). Dextroamphetamine is liberated from lisdexamfetamine enzymatically following contact with red blood cells. The conversion is rate-limited by the enzyme, which prevents high blood concentrations of dextroamphetamine and reduces lisdexamfetamine's drug liking and abuse potential at clinical doses.[75][76] Vyvanse is marketed as once-a-day dosing as it provides a slow release of dextroamphetamine into the body. Vyvanse is available as capsules, and chewable tablets, and in seven strengths; 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 60 mg, and 70 mg. The conversion rate between lisdexamfetamine dimesylate (Vyvanse) to dextroamphetamine base is 29.5%.[77][78][79]
Adderall

Another pharmaceutical that contains dextroamphetamine is commonly known by the brand name Adderall. It is available as immediate release (IR) tablets and extended release (XR) capsules. Adderall contains equal amounts of four amphetamine salts:
- One-quarter racemic (d,l-)amphetamine aspartate monohydrate
- One-quarter dextroamphetamine saccharate
- One-quarter dextroamphetamine sulfate
- One-quarter racemic (d,l-)amphetamine sulfate
Adderall has a total amphetamine base equivalence of 63%.[80] While the enantiomer ratio by dextroamphetamine salts to levoamphetamine salts is 3:1, the amphetamine base content is 75.9% dextroamphetamine, 24.1% levoamphetamine. [note 4]
| drug | formula | molecular mass [note 5] |
amphetamine base [note 6] |
amphetamine base in equal doses |
doses with equal base content [note 7] | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| (g/mol) | (percent) | (30 mg dose) | ||||||||
| total | base | total | dextro- | levo- | dextro- | levo- | ||||
| dextroamphetamine sulfate[82][83] | (C9H13N)2•H2SO4 | 368.49
|
270.41
|
73.38%
|
73.38%
|
—
|
22.0 mg
|
—
|
30.0 mg
| |
| amphetamine sulfate[84] | (C9H13N)2•H2SO4 | 368.49
|
270.41
|
73.38%
|
36.69%
|
36.69%
|
11.0 mg
|
11.0 mg
|
30.0 mg
| |
| Adderall | 62.57%
|
47.49%
|
15.08%
|
14.2 mg
|
4.5 mg
|
35.2 mg
| ||||
| 25% | dextroamphetamine sulfate[82][83] | (C9H13N)2•H2SO4 | 368.49
|
270.41
|
73.38%
|
73.38%
|
—
|
|||
| 25% | amphetamine sulfate[84] | (C9H13N)2•H2SO4 | 368.49
|
270.41
|
73.38%
|
36.69%
|
36.69%
|
|||
| 25% | dextroamphetamine saccharate[85] | (C9H13N)2•C6H10O8 | 480.55
|
270.41
|
56.27%
|
56.27%
|
—
|
|||
| 25% | amphetamine aspartate monohydrate[86] | (C9H13N)•C4H7NO4•H2O | 286.32
|
135.21
|
47.22%
|
23.61%
|
23.61%
|
|||
| lisdexamfetamine dimesylate[67] | C15H25N3O•(CH4O3S)2 | 455.49
|
135.21
|
29.68%
|
29.68%
|
—
|
8.9 mg
|
—
|
74.2 mg
| |
| amphetamine base suspension[63] | C9H13N | 135.21
|
135.21
|
100%
|
76.19%
|
23.81%
|
22.9 mg
|
7.1 mg
|
22.0 mg
| |
Performance
Recreational
Dextroamphetamine is also used recreationally as a euphoriant and aphrodisiac, and like other amphetamines is used as a club drug for its energetic and euphoric high. Dextroamphetamine is considered to have a high potential for misuse in a recreational manner since individuals typically report feeling euphoric, more alert, and more energetic after taking the drug.[87][88][89] Large recreational doses of dextroamphetamine may produce symptoms of dextroamphetamine overdose.[89] Recreational users sometimes open dexedrine capsules and crush the contents in order to insufflate it or subsequently dissolve it in water and inject it.[89] Injection into the bloodstream can be dangerous because insoluble fillers within the tablets can block small blood vessels.[89] Chronic overuse of dextroamphetamine can lead to severe drug dependence, resulting in withdrawal symptoms when drug use stops.[89]
Notes
- ↑ The human dopamine transporter contains a high affinity extracellular zinc binding site which, upon zinc binding, inhibits dopamine reuptake and amplifies amphetamine-induced dopamine efflux in vitro.[25][26][27] The human serotonin transporter and norepinephrine transporter do not contain zinc binding sites.[27]
- ↑ Free-base form amphetamine is a volatile oil, hence the efficacy of the inhalers.
- ↑ These represent the current brands in the United States, except Dexedrine instant release tablets. Dexedrine tablets, introduced in 1937, is discontinued but available as Zenzedi and generically;[51][52] Dexedrine listed here represents the extended release "Spansule" capsule which was approved in 1976.[53][54] Amphetamine sulfate tablets, now sold as Evekeo (brand), were originally sold as Benzedrine (brand) sulfate in 1935[55][56] and discontinued sometime after 1982.[57][58]
- ↑ Calculated by dextroamphetamine base percent / total amphetamine base percent = 47.49/62.57 = 75.90% from table: Amphetamine base in marketed amphetamine medications. The remainder is levoamphetamine.
- ↑ For uniformity, molecular masses were calculated using the Lenntech Molecular Weight Calculator[81] and were within 0.01g/mol of published pharmaceutical values.
- ↑ Amphetamine base percentage = molecular massbase / molecular masstotal. Amphetamine base percentage for Adderall = sum of component percentages / 4.
- ↑ dose = (1 / amphetamine base percentage) × scaling factor = (molecular masstotal / molecular massbase) × scaling factor. The values in this column were scaled to a 30 mg dose of dextroamphetamine sulfate. Due to pharmacological differences between these medications (e.g., differences in the release, absorption, conversion, concentration, differing effects of enantiomers, half-life, etc.), the listed values should not be considered equipotent doses.
- Image legend
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 "Dextroamphetamine Monograph for Professionals". Drugs.com. Archived from the original on 11 August 2021. Retrieved 28 December 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 370. ISBN 978-0857114105.
- ↑ Green-Hernandez, Carol; Singleton, Joanne K.; Aronzon, Daniel Z. (1 January 2001). Primary Care Pediatrics. Lippincott Williams & Wilkins. p. 243. ISBN 9780781720083. Archived from the original on 23 May 2016. Retrieved 8 August 2021.
- ↑ 4.0 4.1 4.2 Millichap JG (2010). "Chapter 9: Medications for ADHD". In Millichap JG (ed.). Attention Deficit Hyperactivity Disorder Handbook: A Physician's Guide to ADHD (2nd ed.). New York, USA: Springer. p. 112. ISBN 9781441913968.
Table 9.2 Dextroamphetamine formulations of stimulant medication
Dexedrine [Peak:2–3 h] [Duration:5–6 h] ...
Adderall [Peak:2–3 h] [Duration:5–7 h]
Dexedrine spansules [Peak:7–8 h] [Duration:12 h] ...
Adderall XR [Peak:7–8 h] [Duration:12 h]
Vyvanse [Peak:3–4 h] [Duration:12 h] - ↑ 5.0 5.1 Mignot EJ (October 2012). "A practical guide to the therapy of narcolepsy and hypersomnia syndromes". Neurotherapeutics. 9 (4): 739–752. doi:10.1007/s13311-012-0150-9. PMC 3480574. PMID 23065655.
- ↑ 6.0 6.1 6.2 Stahl SM (March 2017). "Amphetamine (D)". Prescriber's Guide: Stahl's Essential Psychopharmacology (6th ed.). Cambridge, United Kingdom: Cambridge University Press. pp. 39–44. ISBN 9781108228749. Retrieved 8 August 2017.
- ↑ 7.0 7.1 Stahl SM (March 2017). "Amphetamine (D,L)". Prescriber's Guide: Stahl's Essential Psychopharmacology (6th ed.). Cambridge, United Kingdom: Cambridge University Press. pp. 45–51. ISBN 9781108228749. Archived from the original on 8 June 2019. Retrieved 5 August 2017.
- ↑ "Pharmacology". Dextromphetamine. DrugBank. Archived from the original on 6 August 2019. Retrieved 5 November 2013.
- ↑ "Pharmacology". Amphetamine. DrugBank. University of Alberta. 8 February 2013. Archived from the original on 12 October 2013. Retrieved 5 November 2013.
- ↑ 10.0 10.1 10.2 "HIGHLIGHTS OF PRESCRIBING INFORMATION" (PDF). Archived (PDF) from the original on 18 July 2018. Retrieved 28 December 2021.
- ↑ Lemke TL, Williams DA, Roche VF, Zito W (2013). Foye's Principles of Medicinal Chemistry (7th ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 648. ISBN 978-1609133450.
Alternatively, direct oxidation of amphetamine by DA β-hydroxylase can afford norephedrine.
- ↑ 12.0 12.1 Krueger, SK; Williams, DE (June 2005). "Mammalian flavin-containing monooxygenases: structure/function, genetic polymorphisms and role in drug metabolism". Pharmacology & therapeutics. 106 (3): 357–87. doi:10.1016/j.pharmthera.2005.01.001. PMID 15922018.
- ↑ "Adderall IR Prescribing Information" (PDF). United States Food and Drug Administration. Teva Pharmaceuticals USA, Inc. October 2015. pp. 1–6. Archived (PDF) from the original on 15 September 2018. Retrieved 18 May 2016.
- ↑ "AMPHETAMINE". Archived from the original on 2 October 2017. Retrieved 28 December 2021.
{{cite web}}: CS1 maint: bot: original URL status unknown (link) - ↑ "dextrostat (dextroamphetamine sulfate) tablet [Shire US Inc.]". DailyMed. Wayne, PA: Shire US Inc. August 2006. Archived from the original on 13 January 2012. Retrieved 8 November 2013.
- ↑ "Dexedrine, ProCentra(dextroamphetamine) dosing, indications, interactions, adverse effects, and more". reference.medscape.com. Archived from the original on 6 November 2018. Retrieved 4 October 2015.
Onset of action: 1–1.5 hr
- ↑ Brams M, Mao AR, Doyle RL (September 2008). "Onset of efficacy of long-acting psychostimulants in pediatric attention-deficit/hyperactivity disorder". Postgrad. Med. 120 (3): 69–88. doi:10.3810/pgm.2008.09.1909. PMID 18824827. S2CID 31791162.
Onset of efficacy was earliest for d-MPH-ER at 0.5 hours, followed by d, l-MPH-LA at 1 to 2 hours, MCD at 1.5 hours, d, l-MPH-OR at 1 to 2 hours, MAS-XR at 1.5 to 2 hours, MTS at 2 hours, and LDX at approximately 2 hours. ... MAS-XR, and LDX have a long duration of action at 12 hours postdose
- ↑ Miller, Richard Lawrence (2002). The Encyclopedia of Addictive Drugs. Greenwood Publishing Group. p. 107. ISBN 978-0-313-31807-8. Archived from the original on 11 January 2022. Retrieved 28 December 2021.
- ↑ Cutler, Janis; Marcus, Eric (28 April 2010). Psychiatry. Oxford University Press. p. 569. ISBN 978-0-19-970682-2. Archived from the original on 11 January 2022. Retrieved 28 December 2021.
- ↑ "Dextroamphetamine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 28 December 2021.
- ↑ "DailyMed - DEXTROAMPHETAMINE SULFATE tablet". dailymed.nlm.nih.gov. Archived from the original on 11 January 2022. Retrieved 28 December 2021.
- ↑ "Dexedrine Prescribing Information" (PDF). United States Food and Drug Administration. Amedra Pharmaceuticals LLC. February 2015. pp. 1–7. Archived (PDF) from the original on 17 February 2017. Retrieved 4 September 2015.
- ↑ 23.0 23.1 23.2 23.3 23.4 Heal DJ, Smith SL, Gosden J, Nutt DJ (June 2013). "Amphetamine, past and present – a pharmacological and clinical perspective". J. Psychopharmacol. 27 (6): 479–496. doi:10.1177/0269881113482532. PMC 3666194. PMID 23539642.
- ↑ 24.0 24.1 24.2 24.3 24.4 24.5 "Adderall XR Prescribing Information" (PDF). United States Food and Drug Administration. Shire US Inc. December 2013. pp. 8–10. Archived (PDF) from the original on 30 December 2013. Retrieved 30 December 2013.
- ↑ Krause J (April 2008). "SPECT and PET of the dopamine transporter in attention-deficit/hyperactivity disorder". Expert Rev. Neurother. 8 (4): 611–625. doi:10.1586/14737175.8.4.611. PMID 18416663. S2CID 24589993.
Zinc binds at ... extracellular sites of the DAT [103], serving as a DAT inhibitor. In this context, controlled double-blind studies in children are of interest, which showed positive effects of zinc [supplementation] on symptoms of ADHD [105,106]. It should be stated that at this time [supplementation] with zinc is not integrated in any ADHD treatment algorithm.
- ↑ Sulzer D (February 2011). "How addictive drugs disrupt presynaptic dopamine neurotransmission". Neuron. 69 (4): 628–649. doi:10.1016/j.neuron.2011.02.010. PMC 3065181. PMID 21338876.
They did not confirm the predicted straightforward relationship between uptake and release, but rather that some compounds including AMPH were better releasers than substrates for uptake. Zinc, moreover, stimulates efflux of intracellular [3H]DA despite its concomitant inhibition of uptake (Scholze et al., 2002).
- ↑ 27.0 27.1 Scholze P, Nørregaard L, Singer EA, Freissmuth M, Gether U, Sitte HH (June 2002). "The role of zinc ions in reverse transport mediated by monoamine transporters". J. Biol. Chem. 277 (24): 21505–21513. doi:10.1074/jbc.M112265200. PMID 11940571.
- ↑ Scassellati C, Bonvicini C, Faraone SV, Gennarelli M (October 2012). "Biomarkers and attention-deficit/hyperactivity disorder: a systematic review and meta-analyses". J. Am. Acad. Child Adolesc. Psychiatry. 51 (10): 1003–1019.e20. doi:10.1016/j.jaac.2012.08.015. PMID 23021477.
- ↑ 29.0 29.1 29.2 29.3 29.4 29.5 29.6 Miller GM (January 2011). "The emerging role of trace amine-associated receptor 1 in the functional regulation of monoamine transporters and dopaminergic activity". J. Neurochem. 116 (2): 164–76. doi:10.1111/j.1471-4159.2010.07109.x. PMC 3005101. PMID 21073468.
- ↑ 30.0 30.1 30.2 30.3 30.4 Eiden LE, Weihe E (January 2011). "VMAT2: a dynamic regulator of brain monoaminergic neuronal function interacting with drugs of abuse". Ann. N. Y. Acad. Sci. 1216 (1): 86–98. Bibcode:2011NYASA1216...86E. doi:10.1111/j.1749-6632.2010.05906.x. PMC 4183197. PMID 21272013.
- ↑ Sulzer D, Cragg SJ, Rice ME (August 2016). "Striatal dopamine neurotransmission: regulation of release and uptake". Basal Ganglia. 6 (3): 123–148. doi:10.1016/j.baga.2016.02.001. PMC 4850498. PMID 27141430.
Despite the challenges in determining synaptic vesicle pH, the proton gradient across the vesicle membrane is of fundamental importance for its function. Exposure of isolated catecholamine vesicles to protonophores collapses the pH gradient and rapidly redistributes transmitter from inside to outside the vesicle. ... Amphetamine and its derivatives like methamphetamine are weak base compounds that are the only widely used class of drugs known to elicit transmitter release by a non-exocytic mechanism. As substrates for both DAT and VMAT, amphetamines can be taken up to the cytosol and then sequestered in vesicles, where they act to collapse the vesicular pH gradient.
- ↑ Ledonne A, Berretta N, Davoli A, Rizzo GR, Bernardi G, Mercuri NB (July 2011). "Electrophysiological effects of trace amines on mesencephalic dopaminergic neurons". Front. Syst. Neurosci. 5: 56. doi:10.3389/fnsys.2011.00056. PMC 3131148. PMID 21772817.
Three important new aspects of TAs action have recently emerged: (a) inhibition of firing due to increased release of dopamine; (b) reduction of D2 and GABAB receptor-mediated inhibitory responses (excitatory effects due to disinhibition); and (c) a direct TA1 receptor-mediated activation of GIRK channels which produce cell membrane hyperpolarization.
- ↑ "TAAR1". GenAtlas. University of Paris. 28 January 2012. Retrieved 29 May 2014.
• tonically activates inwardly rectifying K(+) channels, which reduces the basal firing frequency of dopamine (DA) neurons of the ventral tegmental area (VTA)
- ↑ Underhill SM, Wheeler DS, Li M, Watts SD, Ingram SL, Amara SG (July 2014). "Amphetamine modulates excitatory neurotransmission through endocytosis of the glutamate transporter EAAT3 in dopamine neurons". Neuron. 83 (2): 404–416. doi:10.1016/j.neuron.2014.05.043. PMC 4159050. PMID 25033183.
AMPH also increases intracellular calcium (Gnegy et al., 2004) that is associated with calmodulin/CamKII activation (Wei et al., 2007) and modulation and trafficking of the DAT (Fog et al., 2006; Sakrikar et al., 2012). ... For example, AMPH increases extracellular glutamate in various brain regions including the striatum, VTA and NAc (Del Arco et al., 1999; Kim et al., 1981; Mora and Porras, 1993; Xue et al., 1996), but it has not been established whether this change can be explained by increased synaptic release or by reduced clearance of glutamate. ... DHK-sensitive, EAAT2 uptake was not altered by AMPH (Figure 1A). The remaining glutamate transport in these midbrain cultures is likely mediated by EAAT3 and this component was significantly decreased by AMPH
- ↑ Vaughan RA, Foster JD (September 2013). "Mechanisms of dopamine transporter regulation in normal and disease states". Trends Pharmacol. Sci. 34 (9): 489–496. doi:10.1016/j.tips.2013.07.005. PMC 3831354. PMID 23968642.
AMPH and METH also stimulate DA efflux, which is thought to be a crucial element in their addictive properties [80], although the mechanisms do not appear to be identical for each drug [81]. These processes are PKCβ– and CaMK–dependent [72, 82], and PKCβ knock-out mice display decreased AMPH-induced efflux that correlates with reduced AMPH-induced locomotion [72].
- ↑ 36.0 36.1 Bunzow JR, Sonders MS, Arttamangkul S, Harrison LM, Zhang G, Quigley DI, Darland T, Suchland KL, Pasumamula S, Kennedy JL, Olson SB, Magenis RE, Amara SG, Grandy DK (December 2001). "Amphetamine, 3,4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor". Mol. Pharmacol. 60 (6): 1181–8. doi:10.1124/mol.60.6.1181. PMID 11723224. S2CID 14140873.
- ↑ 37.0 37.1 37.2 37.3 Lewin AH, Miller GM, Gilmour B (December 2011). "Trace amine-associated receptor 1 is a stereoselective binding site for compounds in the amphetamine class". Bioorg. Med. Chem. 19 (23): 7044–7048. doi:10.1016/j.bmc.2011.10.007. PMC 3236098. PMID 22037049.
- ↑ Borowsky B, Adham N, Jones KA, Raddatz R, Artymyshyn R, Ogozalek KL, Durkin MM, Lakhlani PP, Bonini JA, Pathirana S, Boyle N, Pu X, Kouranova E, Lichtblau H, Ochoa FY, Branchek TA, Gerald C (July 2001). "Trace amines: identification of a family of mammalian G protein-coupled receptors". Proc. Natl. Acad. Sci. U.S.A. 98 (16): 8966–71. Bibcode:2001PNAS...98.8966B. doi:10.1073/pnas.151105198. PMC 55357. PMID 11459929.
- ↑ 39.0 39.1 39.2 Westfall DP, Westfall TC (2010). "Miscellaneous Sympathomimetic Agonists". In Brunton LL, Chabner BA, Knollmann BC (eds.). Goodman & Gilman's Pharmacological Basis of Therapeutics (12th ed.). New York: McGraw-Hill. ISBN 9780071624428. Archived from the original on 10 November 2013. Retrieved 8 August 2021.
- ↑ "Dexedrine". Medic8. Archived from the original on 19 December 2009. Retrieved 27 November 2013.
- ↑ "Dextroamphetamine [monograph]". Internet Mental Health. Archived from the original on 27 April 2006. Retrieved 6 September 2015.
- ↑ "Information on Dexedrine: A Quick Review | Weitz & Luxenberg". Weitzlux.com. 31 August 2013. Archived from the original on 14 February 2015. Retrieved 5 January 2017.
- ↑ Heal DJ, Smith SL, Gosden J, Nutt DJ (June 2013). "Amphetamine, past and present—a pharmacological and clinical perspective". Journal of Psychopharmacology. 27 (6): 479–96. doi:10.1177/0269881113482532. PMC 3666194. PMID 23539642.
- ↑ King, Diana G (4 January 2017) Prescription Forgery. Handwriting Services International
- ↑ Pharmaceutical Manufacturing Encyclopedia Archived 2 December 2013 at the Wayback Machine (2nd ed.), Marshall Sittig, Volume 1, Noyes Publications ISBN 978-0-8155-1144-1
- ↑ "Dexedrine FAQs". Archived from the original on 17 June 2011.
- ↑ "'Go pills': A war on drugs? – US news – Only – January 2003: BRIDGING THE GULF". NBC News. 9 January 2003. Archived from the original on 11 February 2017. Retrieved 5 January 2017.
- ↑ 48.0 48.1 This story was written by Tech. Sgt. J.C. Woodring. "Air Force scientists battle aviator fatigue". Archived from the original on 14 October 2012. Retrieved 5 January 2017.
{{cite web}}: CS1 maint: bot: original URL status unknown (link) - ↑ Emonson DL, Vanderbeek RD (1995). "The use of amphetamines in U.S. Air Force tactical operations during Desert Shield and Storm". Aviation, Space, and Environmental Medicine. 66 (3): 260–3. PMID 7661838.
- ↑ ‘Go pills’: A war on drugs? Archived 4 September 2020 at the Wayback Machine, NBC News, 9 January 2003
- ↑ Heal, David J; Smith, Sharon L; Gosden, Jane; Nutt, David J (1 June 2013). "Amphetamine, past and present – a pharmacological and clinical perspective". Journal of Psychopharmacology (Oxford, England). 27 (6): 479–496. doi:10.1177/0269881113482532. ISSN 0269-8811. PMC 3666194. PMID 23539642.
Smith, Kline and French synthesised both isomers, and in 1937 commenced marketing of d-amphetamine, which was the more potent of the two isomers, under the trade name of Dexedrine®.
- ↑ "Drugs@FDA: FDA Approved Drug Products: Dexedrine". U.S. Food and Drug Administration. Archived from the original on 28 August 2021. Retrieved 30 December 2015.
DEXEDRINE ... TABLET;ORAL 5MG Discontinued
- ↑ "DEXEDRINE – Official Site For DEXEDRINE – DEXEDRINE Spansule – DEXEDRINE Spansules – DEXEDRINE For ADHD". Official Site For DEXEDRINE. Archived from the original on 17 February 2003. Retrieved 30 December 2015.
- ↑ "Drugs@FDA: Dexedrine: Label and Approval History". U.S. Food and Drug Administration. Archived from the original on 28 August 2021. Retrieved 30 December 2015.
08/02/1976 ... Approval
- ↑ Strohl, Madeleine P. (1 March 2011). "Bradley's Benzedrine Studies on Children with Behavioral Disorders". The Yale Journal of Biology and Medicine. 84 (1): 27–33. ISSN 0044-0086. PMC 3064242. PMID 21451781.
Bradley experimented with Benzedrine sulfate, a drug marketed to doctors by the company Smith, Kline & French (SKF) between 1935 and 1937...
- ↑ Heal, David J; Smith, Sharon L; Gosden, Jane; Nutt, David J (1 June 2013). "Amphetamine, past and present – a pharmacological and clinical perspective". Journal of Psychopharmacology (Oxford, England). 27 (6): 479–496. doi:10.1177/0269881113482532. ISSN 0269-8811. PMC 3666194. PMID 23539642.
Smith, Kline and French introduced Benzedrine onto the market in 1935 as a treatment for narcolepsy (for which it is still used today), mild depression, post-encephalitic Parkinsonism and a raft of other disorders.
- ↑ Heal, David J; Smith, Sharon L; Gosden, Jane; Nutt, David J (1 June 2013). "Amphetamine, past and present – a pharmacological and clinical perspective". Journal of Psychopharmacology (Oxford, England). 27 (6): 479–496. doi:10.1177/0269881113482532. ISSN 0269-8811. PMC 3666194. PMID 23539642.
The use of Benzedrine to treat ADHD declined dramatically after Gross (1976) reported that the racemate was significantly less clinically effective than Dexedrine. Currently, the only use of l-amphetamine in ADHD medications is in mixed salts/mixed enantiomers amphetamine...
- ↑ "FDA Approved Drug Products: Label and Approval History (Benzedrine)". www.accessdata.fda.gov. Archived from the original on 6 May 2016. Retrieved 11 March 2016.
Action Date 5/11/1982, Supplement Number 007, Approval Type Chemistry
- ↑ 59.0 59.1 59.2 59.3 "National Drug Code Amphetamine Search Results". National Drug Code Directory. United States Food and Drug Administration. Archived from the original on 16 December 2013. Retrieved 16 December 2013.
- ↑ "Mydayis Prescribing Information" (PDF). United States Food and Drug Administration. Shire US Inc. June 2017. pp. 1–21. Archived (PDF) from the original on 9 June 2019. Retrieved 8 August 2017.
- ↑ "Adzenys XR-ODT Prescribing Information" (PDF). United States Food and Drug Administration. Neos Therapeutics, Inc. January 2017. p. 16. Archived (PDF) from the original on 23 December 2019. Retrieved 10 August 2017.
ADZENYS XR-ODT (amphetamine extended-release orally disintegrating tablet) contains a 3 to 1 ratio of d- to l-amphetamine, a central nervous system stimulant.
- ↑ "Adzenys XR-ODT". United States Food and Drug Administration. Archived from the original on 25 January 2017. Retrieved 7 March 2016.
- ↑ "Evekeo- amphetamine sulfate tablet". DailyMed. Arbor Pharmaceuticals, LLC. 14 August 2019. Archived from the original on 10 July 2018. Retrieved 22 December 2019.
- ↑ "Evekeo". United States Food and Drug Administration. Archived from the original on 28 June 2017. Retrieved 11 August 2015.
- ↑ 67.0 67.1 "Vyvanse Prescribing Information" (PDF). United States Food and Drug Administration. Shire US Inc. May 2017. pp. 17–21. Retrieved 10 July 2017.
- ↑ "Zenzedi® (dextroamphetamine sulfate, USP)". Zenzedi.com. Archived from the original on 5 January 2017. Retrieved 5 January 2017.
- ↑ ProCentra (dextroamphetamine sulfate 5 mg/5 mL Oral Solution). FSC Laboratories
- ↑ Mickle, Travis et al. (2010) "Abuse-resistant amphetamine prodrugs" U.S. Patent 7,655,630
- ↑ Hazell, Philip (1995). "Stimulant treatment for attention deficit hyperactivity disorder". Australian Prescriber. 18 (3): 60–63. doi:10.18773/austprescr.1995.064.
- ↑ "Pharmaceutical Services". .health.nsw.gov.au. Archived from the original on 5 May 2013. Retrieved 5 January 2017.
- ↑ "Dexamfetamine sulphate - Medicinal forms". British National Formulary. BMJ Group and Pharmaceutical Press (Royal Pharmaceutical Society). Archived from the original on 28 August 2021. Retrieved 9 November 2019.
- ↑ "Red/Amber News Iss. 22 Archived 18 May 2013 at the Wayback Machine", p2. Interface Pharmacist Network Specialist Medicines (IPNSM). www.ipnsm.hscni.net. Retrieved 20 April 2012.
- ↑ Hutson, Peter H.; Pennick, Michael; Secker, Roger (1 December 2014). "Preclinical pharmacokinetics, pharmacology and toxicology of lisdexamfetamine: a novel d-amphetamine pro-drug". Neuropharmacology. 87: 41–50. doi:10.1016/j.neuropharm.2014.02.014. ISSN 1873-7064. PMID 24594478. S2CID 37893582.
- ↑ PHARMACOLOGY/TOXICOLOGY REVIEW AND EVALUATION. U.S. FDA (2006), pp. 18–19
- ↑ Mohammadi M, Akhondzadeh S (17 September 2011). "Advances and considerations in attention-deficit/hyperactivity disorder pharmacotherapy". Acta Medica Iranica. 49 (8): 487–98. PMID 22009816. Archived from the original on 12 March 2014. Retrieved 12 March 2014.
- ↑ Heal, David J.; Buckley, Niki W.; Gosden, Jane; Slater, Nigel; France, Charles P.; Hackett, David (1 October 2013). "A preclinical evaluation of the discriminative and reinforcing properties of lisdexamfetamine in comparison to D-amfetamine, methylphenidate and modafinil". Neuropharmacology. 73: 348–358. doi:10.1016/j.neuropharm.2013.05.021. ISSN 1873-7064. PMID 23748096. S2CID 25343254.
- ↑ Rowley, H. L.; Kulkarni, R.; Gosden, J.; Brammer, R.; Hackett, D.; Heal, D. J. (1 November 2012). "Lisdexamfetamine and immediate release d-amfetamine – differences in pharmacokinetic/pharmacodynamic relationships revealed by striatal microdialysis in freely-moving rats with simultaneous determination of plasma drug concentrations and locomotor activity". Neuropharmacology. 63 (6): 1064–1074. doi:10.1016/j.neuropharm.2012.07.008. ISSN 1873-7064. PMID 22796358. S2CID 29702399.
- ↑ "dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate and amphetamine sulfate (Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate and amphetamine sulfate) tablet". Dailymed.nlm.nih.gov. Archived from the original on 6 January 2017. Retrieved 5 January 2017.
- ↑ "Molecular Weight Calculator". Lenntech. Retrieved 19 August 2015.
- ↑ 82.0 82.1 "Dextroamphetamine Sulfate USP". Mallinckrodt Pharmaceuticals. March 2014. Retrieved 19 August 2015.
- ↑ 83.0 83.1 "D-amphetamine sulfate". Tocris. 2015. Retrieved 19 August 2015.
- ↑ 84.0 84.1 "Amphetamine Sulfate USP". Mallinckrodt Pharmaceuticals. March 2014. Retrieved 19 August 2015.
- ↑ "Dextroamphetamine Saccharate". Mallinckrodt Pharmaceuticals. March 2014. Retrieved 19 August 2015.
- ↑ "Amphetamine Aspartate". Mallinckrodt Pharmaceuticals. March 2014. Retrieved 19 August 2015.
- ↑ "Commonly Abused Prescription Drugs Chart". National Institute on Drug Abuse. Archived from the original on 1 May 2012. Retrieved 7 May 2012.
- ↑ "Stimulant ADHD Medications – Methylphenidate and Amphetamines". National Institute on Drug Abuse. Archived from the original on 2 May 2012. Retrieved 7 May 2012.
- ↑ 89.0 89.1 89.2 89.3 89.4 "National Institute on Drug Abuse. 2009. Stimulant ADHD Medications – Methylphenidate and Amphetamines". National Institute on Drug Abuse. Archived from the original on 12 March 2013. Retrieved 27 February 2013.
External links
| Identifiers: |
|
|---|
- Dextroamphetamine consumer information from Drugs.com Archived 21 April 2021 at the Wayback Machine
- Poison Information Monograph Archived 11 November 2020 at the Wayback Machine (PIM 178: Dexamphetamine Sulphate)
- Pages using duplicate arguments in template calls
- CS1 maint: bot: original URL status unknown
- Webarchive template wayback links
- Use dmy dates from November 2018
- Articles with invalid date parameter in template
- Articles with changed InChI identifier
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with hatnote templates targeting a nonexistent page
- Portal templates with all redlinked portals
- Amphetamine
- Anorectics
- Aphrodisiacs
- Drugs acting on the cardiovascular system
- Drugs acting on the nervous system
- Ergogenic aids
- Euphoriants
- Excitatory amino acid reuptake inhibitors
- Nootropics
- Norepinephrine-dopamine releasing agents
- Phenethylamines
- Stimulants
- Substituted amphetamines
- TAAR1 agonists
- Treatment and management of attention deficit hyperactivity disorder
- VMAT inhibitors
- World Anti-Doping Agency prohibited substances
- RTT