Toceranib
Jump to navigation
Jump to search
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Palladia |
| AHFS/Drugs.com | Veterinary Use |
| License data | |
| Routes of administration | By mouth |
| Drug class | Antineoplastic agent |
| ATCvet code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 77% |
| Protein binding | 91%-93% |
| Elimination half-life | 16 h |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL |
|
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
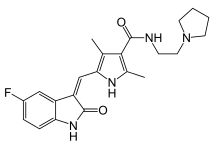
| Formula | C22H25FN4O2 |
| Molar mass | 396.466 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Toceranib is a receptor tyrosine kinase inhibitor and is used in the treatment[1] of canine mast cell tumor also called mastocytoma. Together with masitinib (Kinavet (US)/Masivet (EU/ROW) by AB Science), toceranib is the only dog-specific anti-cancer drug[2] approved by the U.S. Food and Drug Administration (FDA).[3][4] It is sold under the brand name Palladia as its phosphate salt, toceranib phosphate (INN) by Pfizer. It was developed by SUGEN as SU11654,[5] a sister compound to sunitinib, which was later approved for human therapies. Toceranib is likely to act mostly through inhibition of the kit tyrosine kinase, though it may also have an anti-angiogenic effect.
References
- ^ London CA, Malpas PB, Wood-Follis SL, et al. (June 2009). "Multi-center, Placebo-controlled, Double-blind, Randomized Study of Oral Toceranib Phosphate (SU11654), a Receptor Tyrosine Kinase Inhibitor, for the Treatment of Dogs with Recurrent (Either Local or Distant) Mast Cell Tumor Following Surgical Excision". Clin Cancer Res. 15 (11): 3856–65. doi:10.1158/1078-0432.CCR-08-1860. PMID 19470739.
- ^ CBS News FDA Approves First-Ever Dog Cancer Drug
- ^ "FDA: First Drug to Treat Cancer in Dogs Approved". U.S. Food and Drug Administration (FDA) (Press release). 3 June 2009. Archived from the original on 22 July 2010. Retrieved 2 October 2021.
- ^ "Palladia New Animal Drug Application" (PDF). U.S. Food and Drug Administration (FDA). 22 May 2009. Archived from the original (PDF) on 16 November 2010. Retrieved 2 October 2021.
- ^ "In Trials for New Cancer Drugs, Family Pets Are Benefiting, Too". The New York Times. 24 November 2006. Retrieved 2 October 2021.
External links
- "Toceranib". Drug Information Portal. U.S. National Library of Medicine.
Categories:
- Articles with short description
- Short description matches Wikidata
- Chemical pages without DrugBank identifier
- Multiple chemicals in Infobox drug
- Chemicals using indexlabels
- Chemical articles with multiple CAS registry numbers
- Chemical articles with multiple PubChem CIDs
- Articles containing unverified chemical infoboxes
- Receptor tyrosine kinase inhibitors
- Fluoroarenes
- Indoles
- Drugs developed by Pfizer
- Pyrroles
- Carboxamides
- 1-Pyrrolidinyl compounds
- Dog medications
- All stub articles
- Antineoplastic and immunomodulating drug stubs
- Veterinary medicine stubs