Durvalumab
 Antigen-binding fragment of durvalumab (pale green) in complex with PD-L1 (pink). PDB: 5X8M. | |
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | CD274 |
| Names | |
| Trade names | Imfinzi |
| Other names | MEDI4736, MEDI-4736 |
| Clinical data | |
| Drug class | Immune checkpoint inhibitor[1] |
| Main uses | Lung cancer[2] |
| Side effects | Cough, tiredness, pneumonitis, hair loss, shortness of breath, nausea, rash[3] |
| Pregnancy category | |
| Routes of use | Intravenous |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a617030 |
| Legal | |
| License data |
|
| Legal status | |
| Chemical and physical data | |
| Formula | C6502H10018N1742O2024S42 |
| Molar mass | 146322.36 g·mol−1 |
Durvalumab, sold under the brand name Imfinzi, is a medication used to treat non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC).[2] Specifically it is used for cases that produce a protein known as PD-L1.[2] It is given by injection into a vein every two weeks.[2]
Common side effects include cough, tiredness, pneumonitis, hair loss, shortness of breath, nausea, and rash.[3] Other side effects may include immune-mediated damage.[3] Use in pregnancy may harm the baby.[3] It is a monoclonal antibody that attaches to and blocking programmed death-ligand 1 (PD-L1).[2] This results in the immune system being able to attack the cells in question.[2]
Durvalumab was approved for medical use in the United States in 2017 and Europe in 2018.[7][2] In the United Kingdom 500 mg costs the NHS about £2,500 as of 2021.[8] This amount in the United States costs about 3,900 USD.[9]
Medical uses
The US Food and Drug Administration (FDA) has approved durvalumab for certain types of bladder and lung cancer:[6]
- Adults with locally advanced or metastatic urothelial carcinoma who either have disease progression during or following platinum-containing chemotherapy or have disease progression within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy.
- Adults with unresectable, Stage III non-small cell lung cancer whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy.
- In combination with etoposide and either carboplatin or cisplatin, as first-line treatment for adults with extensive-stage small cell lung cancer.
Dosage
It is used at a dose of 10 mg/kg every two weeks.[8]
Mechanism of action
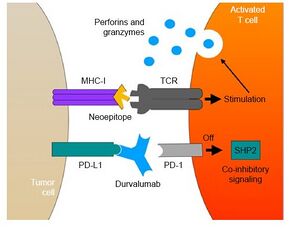
Durvalumab is a checkpoint inhibitor drug.[11]
Research
A phase IB clinical trial of durvalumab and tremelimumab showed some activity in non-small cell lung cancer (NSCLC).[12] Phase 1 data in advanced metastatic urothelial bladder (Study 1108) has led to FDA breakthrough therapy designation.[11][13] Early results of a phase I trial combining durvalumab and gefitinib in lung cancer patients "showed promise".[14] A phase I clinical trial is currently underway using durvalumab with a TLR 7/8 agonist (MEDI 9197) for solid tumors.[15] A Phase 1b/2a trial is underway combining durvalumab with an HPV DNA vaccine (MEDI 0457) in patients with HPV-associated recurrent/metastatic head and neck cancer.[16]
MYSTIC
In July 2017, AstraZeneca announced that a phase III trial of durvalumab with tremelimumab as a first-line treatment of non-small cell lung cancer failed to meet its primary endpoint of progression-free survival.[17]
PACIFIC
In November 2017, the double-blinded phase III AstraZeneca PACIFIC clinical trial demonstrated the efficacy of durvalumab in the treatment of stage III non-small cell lung cancer.[18] 709 patients with stage III NSCLC who did not have disease progression after two or more cycles of a platinum-based chemotherapy were randomly assigned to receive durvalumab or a placebo as consolidation therapy for their lung cancer. Durvalumab increased the median progression-free survival from 5.6 months (placebo) to 16.8 months (durvalumab); the 12 month progression-free survival rate was increased from 35.3% (placebo) to 55.9% (durvalumab), and the 18 month progression-free survival rate was increased from 27.0% (placebo) to 44.2% (durvalumab).[11] The median time to death or distant metastases was also increased from 14.6 months (placebo) to 23.2 months (durvalumab). Extreme side effects were also increased from 26.1% of patients (placebo) to 29.9% of patients (durvalumab).
CASPIAN
In March 2021, the open-label, sponsor-blind (AstraZeneca), randomised, controlled phase 3 trial at 209 cancer treatment centres in 23 countries worldwide (CASPIAN) demonstrated the efficacy of durvalumab in combination with platinum-based chemotherapy in the treatment of small cell lung cancer.[19] Between March 27, 2017, and May 29, 2018, 972 patients were screened and 805 were randomly assigned (268 to durvalumab plus tremelimumab plus platinum–etoposide, 268 to durvalumab plus platinum–etoposide, and 269 to platinum–etoposide). As of Jan 27, 2020, the median follow-up was 25·1 months (IQR 22·3–27·9). Durvalumab plus tremelimumab plus platinum–etoposide was not associated with a significant improvement in overall survival versus platinum–etoposide (hazard ratio [HR] 0·82 [95% CI 0·68–1·00]; p=0·045); median overall survival was 10·4 months (95% CI 9·6–12·0) versus 10·5 months (9·3–11·2). Durvalumab plus platinum–etoposide showed sustained improvement in overall survival versus platinum–etoposide (HR 0·75 [95% CI 0·62–0·91]; nominal p=0·0032); median overall survival was 12·9 months (95% CI 11·3–14·7) versus 10·5 months (9·3–11·2). The most common any-cause grade 3 or worse adverse events were neutropenia (85 [32%] of 266 patients in the durvalumab plus tremelimumab plus platinum–etoposide group, 64 [24%] of 265 patients in the durvalumab plus platinum–etoposide group, and 88 [33%] of 266 patients in the platinum–etoposide group) and anaemia (34 [13%], 24 [9%], and 48 [18%]). Any-cause serious adverse events were reported in 121 (45%) patients in the durvalumab plus tremelimumab plus platinum–etoposide group, 85 (32%) in the durvalumab plus platinum–etoposide group, and 97 (36%) in the platinum–etoposide group. Treatment-related deaths occurred in 12 (5%) patients in the durvalumab plus tremelimumab plus platinum–etoposide group (death, febrile neutropenia, and pulmonary embolism [n=2 each]; enterocolitis, general physical health deterioration and multiple organ dysfunction syndrome, pneumonia, pneumonitis and hepatitis, respiratory failure, and sudden death [n=1 each]), six (2%) patients in the durvalumab plus platinum–etoposide group (cardiac arrest, dehydration, hepatotoxicity, interstitial lung disease, pancytopenia, and sepsis [n=1 each]), and two (1%) in the platinum–etoposide group (pancytopenia and thrombocytopenia [n=1 each]).[20]
References
- ↑ "durvalumab". www.cancer.gov. 2 February 2011. Archived from the original on 28 August 2021. Retrieved 27 December 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Imfinzi". Archived from the original on 28 August 2021. Retrieved 27 December 2021.
- ↑ 3.0 3.1 3.2 3.3 "DailyMed - IMFINZI- durvalumab injection, solution". dailymed.nlm.nih.gov. Archived from the original on 28 August 2021. Retrieved 27 December 2021.
- ↑ 4.0 4.1 "Durvalumab (Imfinzi) Use During Pregnancy". Drugs.com. 30 August 2019. Archived from the original on 29 August 2021. Retrieved 7 February 2020.
- ↑ "Imfinzi EPAR". European Medicines Agency (EMA). Archived from the original on 28 August 2021. Retrieved 30 September 2020.
- ↑ 6.0 6.1 "Imfinzi- durvalumab injection, solution". DailyMed. 5 June 2020. Archived from the original on 28 August 2021. Retrieved 30 September 2020.
- ↑ "Durvalumab Monograph for Professionals". Drugs.com. Archived from the original on 12 December 2021. Retrieved 27 December 2021.
- ↑ 8.0 8.1 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 915. ISBN 978-0857114105.
- ↑ "Imfinzi Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 10 May 2021. Retrieved 27 December 2021.
- ↑ Faiena, Izak; Cummings, Amy L.; Crosetti, Anna M.; Pantuck, Allan J.; Chamie, Karim; Drakaki, Alexandra (23 January 2018). "Durvalumab: an investigational anti-PD-L1 monoclonal antibody for the treatment of urothelial carcinoma". Drug Design, Development and Therapy. 12: 209–215. doi:10.2147/DDDT.S141491.
- ↑ 11.0 11.1 11.2 Syn, Nicholas L; Teng, Michele W L; Mok, Tony S K; Soo, Ross A (2017). "De-novo and acquired resistance to immune checkpoint targeting". The Lancet Oncology. 18 (12): e731–e741. doi:10.1016/s1470-2045(17)30607-1. PMID 29208439.
- ↑ "Astrazeneca's combination of durvalumab with tremelimumab shows clinical activity in non-small cell lung cancer irrespective of PD-L1 status. Feb 2016". Archived from the original on 15 February 2016. Retrieved 20 June 2021.
- ↑ AstraZeneca's (AZN) Durvalumab Granted FDA Breakthrough Therapy Designation Archived 28 August 2021 at the Wayback Machine. StreeInsider.com Corporate News, FDA, Management Comments. 17 February 2016.
- ↑ "Promising Drug for Lung Cancer and Mesothelioma Patients". 19 May 2016. Archived from the original on 28 August 2021. Retrieved 20 June 2021.
- ↑ Clinical trial number NCT02556463 for "A Study of MEDI9197 in Subjects With Solid Tumors or CTCL and in Combination With Durvalumab and/or Palliative Radiation in Subjects With Solid Tumors" at ClinicalTrials.gov
- ↑ Clinical trial number NCT03162224? for "Safety and Efficacy of MEDI0457 and Durvalumab in Patients With HPV Associated Recurrent/Metastatic Head and Neck Cancer" at ClinicalTrials.gov
- ↑ "AstraZeneca reports initial results from the ongoing MYSTIC trial in Stage IV lung cancer". www.astrazeneca.com. Archived from the original on 28 August 2021. Retrieved 20 June 2021.
- ↑ "AstraZeneca presents superior progression-free survival for Imfinzi in the PACIFIC trial of patients with locally-advanced unresectable lung cancer at ESMO 2017 Congress". www.astrazeneca.com. September 2017. Archived from the original on 28 August 2021. Retrieved 20 June 2021.
- ↑ "Archive copy". Archived from the original on 3 June 2021. Retrieved 20 June 2021.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ "Archive copy". Archived from the original on 28 August 2021. Retrieved 20 June 2021.
{{cite web}}: CS1 maint: archived copy as title (link)
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Durvalumab". NCI Drug Dictionary. National Cancer Institute. Archived from the original on 28 August 2021. Retrieved 20 June 2021.
- "Durvalumab". National Cancer Institute. Archived from the original on 28 August 2021. Retrieved 20 June 2021.
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- CS1 maint: archived copy as title
- Use dmy dates from November 2019
- Articles with invalid date parameter in template
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- Drugs that are a monoclonal antibody
- Chemicals that do not have a ChemSpider ID assigned
- Breakthrough therapy
- Experimental cancer drugs
- Monoclonal antibodies
- AstraZeneca brands
- Orphan drugs
- RTT