Panitumumab
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | EGFR |
| Names | |
| Trade names | Vectibix |
| Other names | ABX-EGF |
| Clinical data | |
| Drug class | Monoclonal antibody[1] |
| Main uses | Colorectal cancer[1] |
| Side effects | Diarrhea, nausea, abdominal pain, tiredness, fever, rash, itchiness, dry skin[1] |
| Routes of use | intravenous |
| Typical dose | 6 mg/kg[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607066 |
| Legal | |
| License data | |
| Legal status |
|
| Pharmacokinetics | |
| Elimination half-life | ∼9.4 days (range: 4-11 days) |
| Chemical and physical data | |
| Formula | C6398H9878N1694O2016S48 |
| Molar mass | 144324.12 g·mol−1 |
Panitumumab, sold under the brand name Vectibix, is a medication used for colorectal cancer that has spread to other parts of the body.[1] It may be used alone or together with other medications for tumors that have normal RAS genes.[1] It is used when other treatments have failed.[2] It is given gradually into a vein.[1]
Common side effects include diarrhea, nausea, abdominal pain, tiredness, fever, rash, itchiness, and dry skin.[1] Other concerns include pulmonary fibrosis, infertility, and sepsis.[2][3] Use during pregnancy may harm the baby.[3] It is a monoclonal antibody that attaches to and blocks epidermal growth factor receptor (EGFR).[1]
Panitumumab was approved for medical use in the United States in 2006.[2] It was conditionally approved in Europe in 2007 and fully approved in 2015.[1] In the United Kingdom it costs the NHS about £1,500 per 400 mg as of 2021.[4] This amount in the United States costs about 5,700 USD.[5]
Medical uses
It is used for "the treatment of EGFR-expressing metastatic colorectal cancer with disease progression" despite prior treatment.[6] Panitumumab was approved by the European Medicines Agency (EMEA) in 2007, and by Health Canada in 2008 for "the treatment of refractory EGFR-expressing metastatic colorectal cancer in patients with non-mutated (wild-type) KRAS".
Panitumumab was the first monoclonal antibody to demonstrate the use of KRAS as a predictive biomarker.
Dosage
It is given at a dose of 6 mg/kg every two weeks.[2]
Versus cetuximab
Although they both target the EGFR, panitumumab (IgG2) and cetuximab (IgG1) differ in their isotype and they might differ in their mechanism of action. Monoclonal antibodies of the IgG1 isotype may activate the complement pathway and mediate antibody-dependent cellular cytotoxicity (ADCC).[7] It is not clear at this time, if one drug is superior to the other. In one of the studies, both these drugs were noted to be similar in activity.[8]
Contraindications
Panitumumab does not work in patients who have KRAS or NRAS mutations.[9]
Side effects
Panitumumab has been associated with skin rash, fatigue, nausea, diarrhea, fever, and decreased magnesium levels. Often, skin rash is noted in the sun exposed parts of the body, such as the face or chest. Oral antibiotics may be needed for worsening skin rash, such as one accompanied with blisters and ulcers. Otherwise, topical steroid creams like hydrocortisone may help.[10]
Ocular toxicity or keratitis was observed in 16% of patients on panitumumab, usually necessitating the discontinuance of therapy.[11]
In clinical trials, 90% of patients had dermatological toxicities and 15% of those were severe. Because of this, panitumumab has a boxed warning cautioning patients. Skin toxicities were typically apparent two weeks after beginning treatment. More severe skin toxicities were associated with improved progression free survival and overall survival.[11]
Pulmonary fibrosis and interstitial lung disease were observed in clinical trials.[11]
Pharmacology
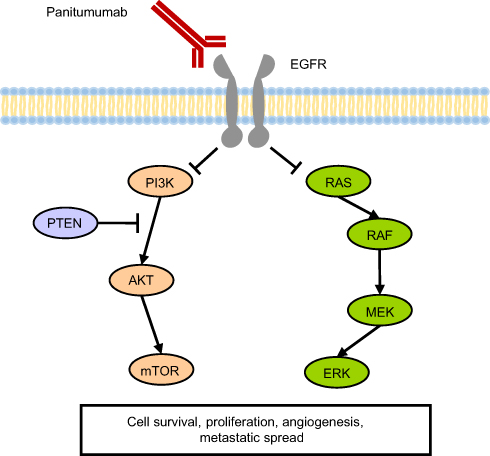
Mechanism of action
EGFR is a transmembrane protein. Panitumumab works by binding to the extracellular domain of the EGFR preventing its activation. This results in halting of the cascade of intracellular signals dependent on this receptor.[13]
Pharmacokinetics
The pharmacokinetics (PK) of panitumumab shows the so-called target-mediated disposition behavior.[14] However, the PK is approximately linear at clinical doses, and the terminal half-life for a typical male patient of 80 kg and 60 years of age with colorectal cancer is about 9.4 days.
History
Panitumumab was generated using Abgenix's XenoMouse platform technology, in which engineered mice were utilized to produce human antibodies. Abgenix partnered with Immunex Corporation to develop the antibody, and Amgen acquired Immunex in 2003. In 2006, Amgen acquired Abgenix as well. In 2013, Amgen formed an agreement with Zhejiang Beta Pharma to form Amgen Beta Pharmaceuticals and market panitumumab in China. Amgen and Takeda have an agreement under which Takeda will develop and commercialise panitumumab in Japan.[15] Panitumumab is licensed to Dr Reddys Laboratories in India and GlaxoSmithKline in the UK.
In 2014, Amgen and Illumina entered into an agreement to develop a companion diagnostic to accompany panitumumab.[16]
FDA approval
Panitumumab was initially approved on September 27, 2006 for EGFR-expressing, metastatic CRC with disease progression on or following fluoropyrimidine-, oxaliplatin-, and irinotecan-containing regimens, based on the results of a study which showed clinical benefit in metastatic colorectal cancer patients.[17] In July 2009, the FDA updated the labels of two anti-EGFR monoclonal antibody drugs (panitumumab and cetuximab) indicated for the treatment of metastatic colorectal cancer to include information about KRAS mutations.[18] This was the result of a study, which demonstrated lack of benefit with Panitumumab in patients who carried NRAS mutations.[9]
It is also approved as a first-line agent in combination with FOLFOX.[11]
Research
Panitumumab is being studied in numerous phase II and III clinical trials. Phase III clinical trials include treatment of esophageal cancer,[19] urothelial carcinoma,[20] metastatic head and neck cancer,[21] and liver metastasis in colorectal cancer.[22] Early trials showed limited efficacy in patients with malignant melanoma, bladder cancer, prostate cancer, and renal cell carcinoma.[15]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 "Vectibix". Archived from the original on 13 August 2021. Retrieved 26 October 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Panitumumab Monograph for Professionals". Drugs.com. Archived from the original on 4 March 2016. Retrieved 26 October 2021.
- ↑ 3.0 3.1 "Panitumumab (Vectibix) Use During Pregnancy". Drugs.com. Archived from the original on 1 December 2020. Retrieved 26 October 2021.
- ↑ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 923. ISBN 978-0857114105.
- ↑ "Vectibix Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 10 February 2018. Retrieved 26 October 2021.
- ↑ "U.S. Food and Drug Administration" (PDF). Archived (PDF) from the original on 2017-02-27. Retrieved 2021-08-13.
- ↑ HealthValue: IgG1 & IgG2 Archived 2019-06-05 at the Wayback Machine[unreliable medical source?]
- ↑ Price TJ, Peeters M, Kim TW, Li J, Cascinu S, Ruff P, et al. (May 2014). "Panitumumab versus cetuximab in patients with chemotherapy-refractory wild-type KRAS exon 2 metastatic colorectal cancer (ASPECCT): a randomised, multicentre, open-label, non-inferiority phase 3 study". The Lancet. Oncology. 15 (6): 569–79. doi:10.1016/S1470-2045(14)70118-4. hdl:2381/43402. PMID 24739896.
- ↑ 9.0 9.1 Douillard JY, Oliner KS, Siena S, Tabernero J, Burkes R, Barugel M, et al. (September 2013). "Panitumumab-FOLFOX4 treatment and RAS mutations in colorectal cancer". The New England Journal of Medicine. 369 (11): 1023–34. doi:10.1056/NEJMoa1305275. PMID 24024839. S2CID 14556160. Archived from the original on 2021-10-17. Retrieved 2021-08-13.
- ↑ Lacouture ME, Mitchell EP, Piperdi B, Pillai MV, Shearer H, Iannotti N, et al. (March 2010). "Skin toxicity evaluation protocol with panitumumab (STEPP), a phase II, open-label, randomized trial evaluating the impact of a pre-Emptive Skin treatment regimen on skin toxicities and quality of life in patients with metastatic colorectal cancer". Journal of Clinical Oncology. 28 (8): 1351–7. doi:10.1200/JCO.2008.21.7828. PMID 20142600. S2CID 30651519. Archived from the original on 2021-10-17. Retrieved 2021-08-13.
- ↑ 11.0 11.1 11.2 11.3 "UpToDate". Archived from the original on 2017-02-02. Retrieved 2021-08-13.
- ↑ Battaglin, Francesca; Puccini, Alberto; Djaballah, Selma Ahcene; Lenz, Heinz-Josef (28 June 2019). "The impact of panitumumab treatment on survival and quality of life in patients with RAS wild-type metastatic colorectal cancer". Cancer Management and Research. 11: 5911–5924. doi:10.2147/CMAR.S186042.
- ↑ Plunkett, Jack W. (September 30, 2005). Plunkett's Biotech & Genetics Industry Almanac 2006. Plunkett Research. ISBN 978-1-59392-033-3.[page needed]
- ↑ Ma P, Yang BB, Wang YM, Peterson M, Narayanan A, Sutjandra L, et al. (October 2009). "Population pharmacokinetic analysis of panitumumab in patients with advanced solid tumors". Journal of Clinical Pharmacology. 49 (10): 1142–56. doi:10.1177/0091270009344989. PMID 19723673. S2CID 25766549.
- ↑ 15.0 15.1 "Panitumumab - Amgen". AdisInsight. Archived from the original on 2017-11-03. Retrieved 2021-08-13.
- ↑ "Illumina, Amgen to Develop CDx for Colorectal Cancer". News: Molecular Diagnostics. Gen. Eng. Biotechnol. News (paper). Vol. 34, no. 4. February 15, 2014. p. 32. Archived from the original on September 19, 2016. Retrieved August 13, 2021.
- ↑ Gibson TB, Ranganathan A, Grothey A (May 2006). "Randomized phase III trial results of panitumumab, a fully human anti-epidermal growth factor receptor monoclonal antibody, in metastatic colorectal cancer". Clinical Colorectal Cancer. 6 (1): 29–31. doi:10.3816/CCC.2006.n.01. PMID 16796788.
- ↑ "FDA updates Vectibix and Erbitux labels with KRAS testing info". Archived from the original on 2016-03-03. Retrieved 2021-08-13.[unreliable medical source?]
- ↑ Clinical trial number NCT01627379 for "Cisplatin and 5-FU +/- Panitumumab for Patients With Nonresectable,Advanced or Metastatic Esophageal Squamous Cell Cancer" at ClinicalTrials.gov
- ↑ Clinical trial number NCT00460265 NCT00460265 for "I-MVAC +/- Panitumumab as First-line Treatment of Advanced Urothelial Carcinoma Without H-Ras Nor K-Ras Mutations" at ClinicalTrials.gov
- ↑ Clinical trial number NCT00460265 NCT00460265 for "Study of Panitumumab Efficacy in Patients With Recurrent and/or Metastatic Head and Neck Cancer" at ClinicalTrials.gov
- ↑ Clinical trial number NCT02162563 for "Treatment Strategies in Colorectal Cancer Patients With Initially Unresectable Liver-only Metastases" at ClinicalTrials.gov
Further reading
- Amado RG, Wolf M, Peeters M, Van Cutsem E, Siena S, Freeman DJ, et al. (April 2008). "Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer" (PDF). Journal of Clinical Oncology. 26 (10): 1626–34. doi:10.1200/JCO.2007.14.7116. hdl:2434/349676. PMID 18316791. Archived (PDF) from the original on 2021-10-17. Retrieved 2021-08-13.
- Van Cutsem E, Peeters M, Siena S, Humblet Y, Hendlisz A, Neyns B, et al. (May 2007). "Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer". Journal of Clinical Oncology. 25 (13): 1658–64. doi:10.1200/JCO.2006.08.1620. PMID 17470858.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- All articles lacking reliable references
- Articles lacking reliable references from December 2014
- Articles with invalid date parameter in template
- Wikipedia articles needing page number citations from December 2014
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugs that are a monoclonal antibody
- Articles with hatnote templates targeting a nonexistent page
- Chemicals that do not have a ChemSpider ID assigned
- Articles with changed EBI identifier
- Monoclonal antibodies for tumors
- Amgen
- Takeda Pharmaceutical Company brands
- RTT