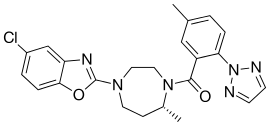
Suvorexant
 | |
 | |
| Names | |
|---|---|
| Trade names | Belsomra |
| Other names | MK-4305 |
| |
| Clinical data | |
| Drug class | Dual orexin receptor antagonist (DORA)[1] |
| Main uses | Trouble sleeping[1] |
| Side effects | Sleepiness, headache, unusual dreams, cough, dry mouth[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Typical dose | 10 to 20 mg OD[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614046 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 82% (at 10 mg) |
| Protein binding | >99% |
| Metabolism | hepatic, CYP3A, CYP2C19 |
| Elimination half-life | ~12 hours |
| Excretion | Feces (66%), urine (23%) |
| Chemical and physical data | |
| Formula | C23H23ClN6O2 |
| Molar mass | 450.93 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Suvorexant, sold under the trade name Belsomra, is a medication used to treat trouble sleeping, including trouble falling asleep and staying asleep.[1] It may be effective for at least a year.[1] It is taken by mouth, half an hour before bed.[1]
Common side effects include sleepiness, headache, unusual dreams, cough, and dry mouth.[1] Other side effects include abuse, sleep paralysis, suicide, anxiety, and a decreased ability to drive.[1] Safety in pregnancy and breastfeeding is unclear.[3] It is a dual orexin receptor antagonist (DORA).[1]
Suvorexant was approved for medical use in the United States in 2014,[1] and Canada in 2018.[4] In the United States it costs about 380 USD per month as of 2021.[5] In the United States it is a Schedule IV controlled substance.[6]
Medical uses
Suvorexant is used for the treatment of insomnia, characterized by difficulties with sleep onset and/or sleep maintenance.[7]
It is unclear how the medication compares to others used for insomnia as no comparisons have been done.[8] It is also unclear if this medication is safe among people with a history of addiction, as they were excluded from the clinical trials of suvorexant.[8]
Dosage
It is generally started at a dose of 10 mg per day and may be increased to 20 mg per day.[1]
Contraindications
Suvorexant is not recommended in people with liver impairment.[7] Suvorexant pregnancy category is classified as Category C.[7] Based on animal testing, this medication may cause fetal harm during pregnancy and should only be given in pregnancy if the potential benefit justifies the potential harm to the fetus. Evidence is inconclusive about whether using this medication while breastfeeding puts the infant at risk of harm.[7]
Suvorexant is contraindicated in people diagnosed with narcolepsy.[7]
Side effects
The most common complaint about the drug is from users who report that it did not help them to sleep.[9] Some people reported that the drug caused a sleep disturbance such as a nightmare, sleep terror, or abnormal dream.[9][10] Others reported that the drug caused them to be more awake.[9]
Issues include sleepiness the next day and issues with driving.[10] Other concerns include thoughts of suicide.[10]
Misuse
According to the U.S. Drug Enforcement Administration (DEA), suvorexant produces similar reinforcing effects to those of zolpidem and thus may have a similar abuse liability.[11] As such, suvorexant has been designated a schedule IV controlled substance in the U.S. under the Controlled Substances Act.[11]
Interactions
Suvorexant is not recommended if people are also taking medications that strongly inhibit the liver enzyme CYP3A like itraconazole, lopinavir/ritonavir, clarithromycin, ritonavir, ketoconazole, indinavir/ritonavir, or conivaptan.[7][12] If suvorexant is used with a medication that moderately inhibits the liver enzyme CYP3A, like verapamil, erythromycin, diltiazem, or dronedarone, it is recommended that the dose of suvorexant be adjusted.[7][12]
Pharmacology
Pharmacodynamics
Suvorexant exerts its therapeutic effect in insomnia through antagonism of orexin receptors. The orexin neuropeptide signaling system is a central promoter of wakefulness. Blocking the binding of wake-promoting neuropeptides orexin A and orexin B to receptors orexin receptor type 1 (OX1) and orexin receptor type 2 (OX2) is thought to suppress wake drive.[7] Animal studies report the binding affinities for OX1 (0.55 nM) and OX2 (0.35 nM).[13]
Pharmacokinetics
The bioavailability of suvorexant is at 82%. It is highly protein-bound. Food delays the time to max concentration. The primary route of elimination is through the feces, with approximately 66% of radiolabeled dose recovered in the feces compared to 23% in the urine. The elimination half-life is reported to be 12 hours.[7]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 "Suvorexant Monograph for Professionals". Drugs.com. Archived from the original on 7 March 2016. Retrieved 15 October 2021.
- ↑ "Suvorexant (Belsomra) Use During Pregnancy". Drugs.com. 9 September 2019. Archived from the original on 24 January 2021. Retrieved 30 January 2020.
- ↑ "Suvorexant (Belsomra) Use During Pregnancy". Drugs.com. Archived from the original on 24 January 2021. Retrieved 15 October 2021.
- ↑ "Regulatory Decision Summary - Belsomra - Health Canada". hpr-rps.hres.ca. Government of Canada. Archived from the original on 6 July 2020. Retrieved 6 February 2020.
- ↑ "Suvorexant Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 15 October 2021.
- ↑ "Rules - 2014 - Final Rule: Placement of Suvorexant into Schedule IV". www.deadiversion.usdoj.gov. Archived from the original on 2016-04-17. Retrieved 2016-04-03.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 "Belsomra- suvorexant tablet, film coated". DailyMed. 20 November 2019. Archived from the original on 2 July 2019. Retrieved 30 January 2020.
- ↑ 8.0 8.1 Patel KV, Aspesi AV, Evoy KE (April 2015). "Suvorexant: a dual orexin receptor antagonist for the treatment of sleep onset and sleep maintenance insomnia". The Annals of Pharmacotherapy. 49 (4): 477–83. doi:10.1177/1060028015570467. PMID 25667197.
- ↑ 9.0 9.1 9.2 Carr T (5 February 2016). "FDA Fields Complaints on Sleeping Pill Suvorexant". Consumer Reports. Archived from the original on 2 December 2020. Retrieved 4 December 2020.
- ↑ 10.0 10.1 10.2 Jacobson LH, Callander GE, Hoyer D (November 2014). "Suvorexant for the treatment of insomnia". Expert Review of Clinical Pharmacology. 7 (6): 711–30. doi:10.1586/17512433.2014.966813. PMID 25318834.
- ↑ 11.0 11.1 "Schedules of controlled substances: placement of suvorexant into Schedule IV. Final rule" (PDF). Federal Register. 79 (167): 51243–7. August 2014. PMID 25167596. Archived (PDF) from the original on 2016-08-11. Retrieved 2020-12-04.
- ↑ 12.0 12.1 "U.S. Food and Drug Administration." Drug Development and Drug Interactions: Table of Substrates, Inhibitors and Inducers. U.S. Food and Drug Administration, 27 Oct. 2014. Web. 30 Oct. 2014.
- ↑ "Suvorexant Advisory Committee Meeting Briefing Document" (PDF). May 22, 2013. Archived (PDF) from the original on February 1, 2015. Retrieved Feb 7, 2015.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Parker I (9 December 2013). "The Big Sleep". The New Yorker. Archived from the original on 3 December 2020. Retrieved 4 December 2020.
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Articles with changed InChI identifier
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with changed CASNo identifier
- Articles with changed ChemSpider identifier
- Articles with changed EBI identifier
- Benzoxazoles
- Chloroarenes
- Diazepanes
- Hypnotics
- Merck & Co. brands
- Orexin antagonists
- Sedatives
- Triazoles
- RTT