Insulin aspart
 | |
| Names | |
|---|---|
| Trade names | NovoLog, NovoRapid, Fiasp, others |
| Clinical data | |
| Drug class | Insulin (short acting) |
| Main uses | Diabetes[1] |
| Side effects | Low blood sugar, allergic reactions, itchiness, pain at the site of injection[1] |
| Pregnancy category | |
| Routes of use | Subcutaneous, intravenous |
| Onset of action | ~ 2 hrs (max)[1] |
| Duration of action | ~ 4 hrs[1] |
| Defined daily dose | 40 units[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605013 |
| Legal | |
| License data |
|
| Legal status | |
| Chemical and physical data | |
| Formula | C256H381N65O79S6 |
| Molar mass | 5825.60 g·mol−1 |
Insulin aspart, sold under the brand name Novolog among others, is a type of manufactured insulin used to treat type 1 and type 2 diabetes.[1] Typically it is taken just before eating.[1] It is generally used by injection under the skin but may also be used by injection into a vein.[1] Maximum effect occurs after about 1–3 hours and lasts for 3–5 hours.[1] Generally a longer-acting insulin like NPH is also needed.[1]
Common side effects include low blood sugar, allergic reactions, itchiness, and pain at the site of injection.[1] Other serious side effects may include low blood potassium.[1] Use in pregnancy and breastfeeding is generally safe.[2] It works the same as human insulin by increasing the amount of glucose that tissues take in and decreasing the amount of glucose made by the liver.[1] It is a manufactured form of human insulin; where a single amino acid has been changed, specifically a proline with an aspartic acid at the B28 position.[4]
Insulin aspart was approved for medical use in the United States in 2000.[1] In the United Kingdom it costs the NHS about £1.89 per 100 units as of 2019.[5] In the United States the wholesale cost of this amount is about US$30.00.[6] In 2017, it was the 56th most prescribed medication in the United States, with more than 14 million prescriptions.[7][8] Manufacturing involves yeast, which have had the gene for insulin aspart put into their genome.[9] This yeast then makes the insulin, which is harvested from the bioreactor.[10]
Medical uses
As of 2018[update], there is a lack of compelling evidence to conclude superiority of insulin aspart over human insulin in type 2 DM.[11] It is thus unclear why the shifting of people from human insulin to insulin aspart has occurred.[12] In type 1 DM it appears to result in slightly better blood sugar control.[13]
Onset of action
The onset of action is approximately 15 minutes, the peak action is reached in 45–90 minutes, and the duration is 3–5 hours. However, as with all insulin, these numbers are based on averages, and vary between individuals due to blood flow, injection site, temperature and exercise.[14][15]
Dosage
The defined daily dose is 40 units by injection.[3]
Side effects
The safety of insulin aspart in people with diabetes is no different than for regular insulin. The side effects that are commonly associated with insulin therapy include: allergic reactions, injection site irritation, rashes, and hypoglycemia.[16] The most common side effect is hypoglycemia. Long-term use of insulin, including insulin aspart, can cause lipodystrophy at the site of repeated injections or infusion. To reduce the risk of lipodystrophy, rotate the injection sites within the same region. Weight gain can also occur with the use of insulin aspart and it has been attributed to anabolic effects of insulin and a decrease in glucosuria. Use of insulin aspart has also been associated with sodium retention and edema.[17]
Chemical properties
The components of insulin aspart are as follows:
- Metal ion – zinc (19.6 μg/mL)
- Buffer – disodium hydrogen phosphate dihydrate (1.25 mg/mL)
- Preservatives – 3-methylphenol (m-cresol) (1.72 mg/mL) and phenol (1.50 mg/mL)
- Isotonicity agents – glycerin (16 mg/mL) and sodium chloride (0.58 mg/mL).
The pH of insulin aspart is 7.2–7.6.[14]
Formulations
Insulin aspart can be used in an insulin pump and insulin pen for subcutaneous injection. Additionally, it can be used with an injection port such as the I-port.[18]
Insulin aspart has a more rapid onset, and a shorter duration of activity than normal human insulin. Therefore, insulin aspart given by injection should normally be used in a regimen with long-acting or intermediate insulin.[19] Insulin aspart can also be used with external insulin pumps. The insulin in reservoirs of insulin pumps and infusion sets should be changed every 48 hours to avoid insulin degradation and loss of preservative.[15]
Temperature
It has been debated whether or not insulin aspart should be kept refrigerated at all times. The manufacturer claims it can last 28 days without refrigeration, as long as it is kept below 86 °F / 30 °C.[20] Above these temperatures, the potency of the insulin quickly degrades, rendering it less effective.
Variations
NovoLog Mix 70/30 is a product which contains 30% insulin aspart and 70% insulin aspart protamine. The insulin aspart protamine portion is a crystalline form of insulin aspart, which delays the action of the insulin, giving it a prolonged absorption profile after injection. The combination of the fast-acting form and the long-acting form allows the patient to receive fewer injections over the course of the day.[21]
The components of NovoLog Mix 70/30 are as follows:
- Metal ion – zinc (19.6 μg/mL)
- Buffer – dibasic sodium phosphate (1.25 mg/mL)
- Preservatives – m-cresol (1.72 mg/mL) and phenol (1.50 mg/mL)
- Isotonicity agents – sodium chloride (0.58 mg/mL) and mannitol (36.4 mg/mL)
- Modifying protein – protamine (0.33 mg/mL)
The pH is 7.2–7.44.[14]
NovoLog Mix is marketed to be used with the Novo Nordisk FlexPen.[22] The onset of action is less than 30 minutes, the peak action is reached in 1–4 hours, and the duration is less than 24 hours.[14] NovoLog Mix is marketed in some countries as NovoMix 30.[23][24]
Society and culture
Cost
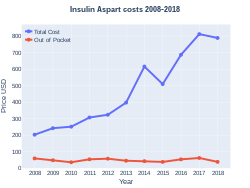
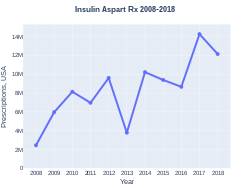
Insulin aspart was approved for medical use in the United States in 2000.[1] In the United Kingdom it costs the NHS about £1.89 per 100 units as of 2019.[5] In the United States the wholesale cost of this amount is about US$30.00.[6] In 2017, it was the 56th most prescribed medication in the United States, with more than 14 million prescriptions.[7][8]
-
Insulin aspart costs (US)
-
Insulin aspart prescriptions (US)
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 "Insulin Aspart Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 6 March 2019. Retrieved 3 March 2019.
- ↑ 2.0 2.1 2.2 "Insulin aspart Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 6 March 2019. Retrieved 3 March 2019.
- ↑ 3.0 3.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 8 August 2020. Retrieved 6 September 2020.
- ↑ Turner JR (2010). New Drug Development: An Introduction to Clinical Trials: Second Edition. Springer Science & Business Media. p. 32. ISBN 9781441964182. Archived from the original on 2019-03-06. Retrieved 2019-03-06.
- ↑ 5.0 5.1 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 697. ISBN 9780857113382.
- ↑ 6.0 6.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019. Cite error: Invalid
<ref>tag; name "NADAC2019" defined multiple times with different content - ↑ 7.0 7.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 8.0 8.1 "Insulin Aspart - Drug Usage Statistics". ClinCalc. Archived from the original on 12 April 2020. Retrieved 11 April 2020.
- ↑ Banga, Ajay K. (2005). Therapeutic Peptides and Proteins: Formulation, Processing, and Delivery Systems, Second Edition. CRC Press. p. 13. ISBN 9781420039832. Archived from the original on 2019-03-06. Retrieved 2019-03-06.
- ↑ Schmid, Rolf D.; Schmidt-Dannert, Claudia (2016). Biotechnology: An Illustrated Primer. John Wiley & Sons. p. 222. ISBN 9783527677566. Archived from the original on 2019-03-06. Retrieved 2019-03-06.
- ↑ Fullerton B, Siebenhofer A, Jeitler K, Horvath K, Semlitsch T, Berghold A, Gerlach FM (December 2018). "Short-acting insulin analogues versus regular human insulin for adult, non-pregnant persons with type 2 diabetes mellitus". The Cochrane Database of Systematic Reviews. 12: CD013228. doi:10.1002/14651858.CD013228. PMC 6517032. PMID 30556900.
- ↑ Cohen D, Carter P (December 2010). "How small changes led to big profits for insulin manufacturers". BMJ. 341: c7139. doi:10.1136/bmj.c7139. PMID 21159773. Archived from the original on 2021-08-29. Retrieved 2019-11-29.
- ↑ Fullerton B, Siebenhofer A, Jeitler K, Horvath K, Semlitsch T, Berghold A, et al. (June 2016). "Short-acting insulin analogues versus regular human insulin for adults with type 1 diabetes mellitus". The Cochrane Database of Systematic Reviews (6): CD012161. doi:10.1002/14651858.CD012161. PMC 6597145. PMID 27362975.
- ↑ 14.0 14.1 14.2 14.3 Crommelin DJA, Sindelar RD, Meibohm B. 2008. Pharmaceutical Biotechnology: Fundamentals and Applications. New York, NY: Informa Healthcare USA, Inc. p 270.
- ↑ 15.0 15.1 "NovoLog Insulin Aspart (rDNA origin) Injection" (PDF). FDA. 7 June 2000. Archived (PDF) from the original on 4 March 2016. Retrieved 1 September 2013.
- ↑ "Novolog: most FAQ". Archived from the original on 2011-10-03. Retrieved 2011-04-18.
- ↑ "Novolog: insulin aspart (rDNA origin) injection" (PDF). Novo Nordisk. Archived (PDF) from the original on 2011-03-22. Retrieved 2011-04-18.
- ↑ "Aspart insulin (rDNA origin) injection". Archived from the original on 2007-06-10. Retrieved 2007-06-08.
- ↑ "Novolog: insulin aspart" (PDF). Archived (PDF) from the original on 2017-05-17.
- ↑ "insulin aspart Storage". Novolog.com. Archived from the original on 2016-03-04. Retrieved 2016-04-23.
- ↑ Rx List: NovoLog Mix 70/30. 6 August 2008. http://www.rxlist.com/novolog-mix-70-30-drug.htm Archived 2009-04-20 at the Wayback Machine
- ↑ Novo Nordisk: NovoLog Mix 70/30. 2008. http://www.novologmix70-30.com/starting-with-novolog-mix.asp Archived 2009-05-08 at the Wayback Machine
- ↑ "NovoMix 30 FlexPen 100 units/ml - Summary of Product Characteristics (SmPC)". (emc). 27 March 2019. Archived from the original on 13 April 2020. Retrieved 12 April 2020.
- ↑ "NovoMix". European Medicines Agency. 17 September 2018. Archived from the original on 13 April 2020. Retrieved 12 April 2020.
External links
| Identifiers: |
|
|---|
- "Insulin aspart". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-04-13. Retrieved 2020-04-13.
- Pages using duplicate arguments in template calls
- Pages with reference errors
- Webarchive template wayback links
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Articles containing potentially dated statements from 2018
- Articles with invalid date parameter in template
- All articles containing potentially dated statements
- Articles with changed CASNo identifier
- Chemicals that do not have a ChemSpider ID assigned
- Articles with changed EBI identifier
- Insulin receptor agonists
- Insulin therapies
- Diabetes
- Human proteins
- Recombinant proteins
- Peptide hormones
- RTT