NPH insulin
 A vial of NPH insulin with insulin syringe | |
| Names | |
|---|---|
| Trade names | Novolin N, Humulin N, Insulatard, others |
| Other names | Neutral protamine Hagedorn insulin,[1] protamine zinc insulin (slightly different),[2] isophane insulin,[2] compound insulin zinc suspension (slightly different),[2] intermediate-acting insulin |
| Clinical data | |
| Drug class | Insulin (intermediate)[3] |
| Main uses | Diabetes[3] |
| Side effects | Low blood sugar, pain or skin changes at the sites of injection, low blood potassium, allergic reactions[3] |
| Pregnancy category |
|
| Routes of use | Subcutaneous |
| Onset of action | 90 minutes[3] |
| Duration of action | 24 hours[3] |
| Defined daily dose | 40 Iu[4] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682611 |
NPH insulin, also known as isophane insulin, is an intermediate-acting insulin given to help control blood sugar levels in people with diabetes.[3] It is used by injection under the skin once to twice a day.[1] Onset of effects is typically in 90 minutes and they last for 24 hours.[3] Versions are available that come premixed with a short-acting insulin, such as regular insulin.[2]
The common side effect is low blood sugar.[3] Other side effects may include pain or skin changes at the sites of injection, low blood potassium, and allergic reactions.[3] Use during pregnancy is relatively safe for the baby.[3] NPH insulin is made by mixing regular insulin and protamine in exact proportions with zinc and phenol such that a neutral-pH is maintained and crystals form.[1] There are human and pig insulin based versions.[1]
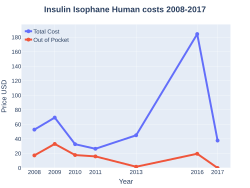
Protamine insulin was first created in 1936 and NPH insulin in 1946.[1] It is on the World Health Organization's List of Essential Medicines.[5] The wholesale cost in the developing world is about US$2.23–10.35 per 1,000 iu of NPH insulin.[6] In the United Kingdom 1,000 iu of NPH insulin costs the NHS £7.48 while in the United States this amount costs about US$134.00.[2][7] NPH is an abbreviation for "neutral protamine Hagedorn".[1]
Medical use
NPH insulin is cloudy and has an onset of 1–4 hours. Its peak is 6–10 hours and its duration is about 10–16 hours.
It has an intermediate duration of action, meaning longer than that of regular and rapid-acting insulin, and shorter than long acting insulins (ultralente, glargine or detemir).
Dosage
The defined daily dose is 40 iu (via injection).[4]
History
Hans Christian Hagedorn (1888–1971) and August Krogh (1874–1949) obtained the rights for insulin from Banting and Best in Toronto, Canada. In 1923 they formed Nordisk Insulin laboratorium, and in 1926 with August Kongsted he obtained a Danish Royal Charter as a non-profit foundation.
In 1936, Hagedorn and B. Norman Jensen discovered that the effects of injected insulin could be prolonged by the addition of protamine obtained from the "milt" or semen of river trout. The insulin would be added to the protamine, but the solution would have to be brought to pH 7 for injection. University of Toronto, Canada later licensed protamine zinc insulin (PZI),[8] to several manufacturers. This mixture only needed to be shaken before injection. The effects of PZI lasted for 24–36 h.
In 1946, Nordisk was able to form crystals of protamine and insulin and marketed it in 1950 as neutral protamine Hagedorn (NPH) insulin. NPH insulin has the advantage that it can be mixed with an insulin that has a faster onset to complement its longer lasting action, which is the primary reason NPH remains on the market today, because manufacturers sell a variety of premixed insulin formulations.
Eventually all animal insulins made by Novo Nordisk were replaced by synthetic, recombinant 'human' insulin. Synthetic 'human' insulin is also complexed with protamine to form NPH.
Timeline
- 1926 Nordisk receives Danish charter to produce insulin
- 1936 Hagedorn discovers that adding protamine to insulin prolongs the effect of insulin
- 1936 Canadians D.M. Scott and A.M. Fisher formulate zinc insulin mixture and license to Novo
- 1946 Nordisk crystallizes a protamine and insulin mixture
- 1950 Nordisk markets NPH insulin
- 1953 Nordisk markets "Lente" zinc insulin mixtures.
Names
NPH stands for neutral protamine Hagedorn, and the words refer to neutral pH (pH = 7), protamine (a protein), and Hans Christian Hagedorn (an insulin researcher).
Brand names include Humulin N, Novolin N, Novolin NPH, Gensulin N, SciLin N, Insulatard, and NPH Iletin II.
Society and culture
Cost
The wholesale cost in the developing world is about US$2.23–10.35 per 1,000 iu of NPH insulin.[6] In the United Kingdom 1,000 iu of NPH insulin costs the NHS £7.48 while in the United States this amount costs about US$134.00.[2][7]
-
NPH insulin costs (US)
-
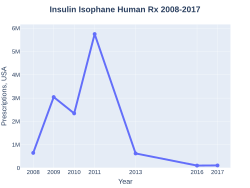
NPH insulin prescriptions (US)
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Owens, D. R. (1986). Human Insulin: Clinical Pharmacological Studies in Normal Man. Springer Science & Business Media. pp. 134–136. ISBN 9789400941618. Archived from the original on 2017-01-18.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 British national formulary: BNF 69 (69 ed.). British Medical Association. 2015. pp. 464–472. ISBN 9780857111562.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 "Insulin Human". The American Society of Health-System Pharmacists. Archived from the original on 22 October 2016. Retrieved 8 January 2017.
- ↑ 4.0 4.1 "Single Drug Information – International Medical Products Price Guide". mshpriceguide. Archived from the original on 28 August 2021. Retrieved 17 August 2020.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 6.0 6.1 "Insulin, Neutral Soluble". International Drug Price Indicator Guide. Archived from the original on 7 July 2018. Retrieved 8 December 2016.
- ↑ 7.0 7.1 "NADAC as of 2016-12-07 | Data.Medicaid.gov". Centers for Medicare and Medicaid Services. Archived from the original on 21 December 2016. Retrieved 17 January 2017.
- ↑ "The History of Insulin" (PDF). https://www.karger.com/. Basel, Switzerland: Karger Publishers. Archived (PDF) from the original on March 4, 2016. Retrieved June 10, 2015.
{{cite web}}: External link in|website=
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- CS1 errors: external links
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Chemicals that do not have a ChemSpider ID assigned
- Insulin therapies
- Human proteins
- Recombinant proteins
- Peptide hormones
- Eli Lilly and Company brands
- World Health Organization essential medicines
- RTT
- Medical mnemonics