Linagliptin
 | |
| Names | |
|---|---|
| Pronunciation | /ˌlɪnəˈɡlɪptɪn/ LIN-ə-GLIP-tin |
| Trade names | Tradjenta, Trajenta, others |
| Other names | BI-1356 |
| |
| Clinical data | |
| Drug class | DPP-4 inhibitors[1] |
| Main uses | Diabetes mellitus type 2[1] |
| Side effects | Inflammation of the nose and throat[1] |
| Pregnancy category |
|
| Routes of use | By mouth (tablets) |
| Defined daily dose | 5 mg[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a611036 |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | ~30% (Tmax = 1.5 hours) |
| Protein binding | 75–99% (concentration-dependent) |
| Metabolism | Minimal (~10% metabolized) |
| Metabolites | Pharmacologically inactive |
| Elimination half-life | ~24 hours |
| Excretion | Feces (80%), urine (5%)[3] |
| Chemical and physical data | |
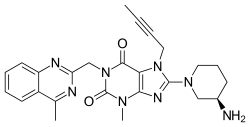
| Formula | C25H28N8O2 |
| Molar mass | 472.553 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Linagliptin, sold under the brand name Tradjenta among others, is a medication used to treat diabetes mellitus type 2.[1] It is generally less preferred than metformin and sulfonylureas as an initial treatment.[1][4] It is used together with exercise and diet.[1] It is not recommended in type 1 diabetes.[1] It is taken by mouth.[1]
Common side effects include inflammation of the nose and throat.[1] Serious side effects may include angioedema, pancreatitis, joint pain.[4][1] Use in pregnancy and breastfeeding is not recommended.[4] Linagliptin is a dipeptidyl peptidase-4 inhibitor (DPP-4 inhibitors).[1] It works by increasing the production of insulin and decreasing the production of glucagon by the pancreas.[1]
Linagliptin was approved for medical use in the United States in 2011.[1] A month supply in the United Kingdom costs the NHS about £33 as of 2019.[4] In the United States the wholesale cost of this amount is about 391 USD.[5] In 2017, it was the 200th most commonly prescribed medication in the United States, with more than two million prescriptions.[6][7]
Medical uses
Results in 2010 from a Phase III clinical trial of linagliptin showed that the drug can effectively reduce blood sugar.[8]
Dosage
The defined daily dose is 5 mg by mouth.[2]
Side effects
Linagliptin may cause severe joint pain.[3][9]
The U.S. Food and Drug Administration (FDA) is warning that the type 2 diabetes medicines like sitagliptin, saxagliptin, linagliptin, and alogliptin may cause joint pain that can be severe and disabling. FDA has added a new Warning and Precaution about this risk to the labels of all medicines in this drug class, called dipeptidyl peptidase-4 (DPP-4) inhibitors.
Trajenta's Prescribing Information[10] states the drug is contraindicated for people with bronchial hyperreactivity. Asthma is a form of bronchial hyperreactivity[11][circular reference].
Mechanism of action
Linagliptin belongs to a class of drugs called DPP-4 inhibitors.
Terminology
Society and culture
Cost
A month supply in the United Kingdom costs the NHS about £33 as of 2019.[4] In the United States the wholesale cost of this amount is about 391 USD.[5] In 2017, it was the 200th most commonly prescribed medication in the United States, with more than two million prescriptions.[6][7]
-
Linagliptin costs (US)
-
Linagliptin prescriptions (US)
See also
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 "Linagliptin Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 6 April 2019. Retrieved 6 April 2019.
- ↑ 2.0 2.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 1 November 2020. Retrieved 8 September 2020.
- ↑ 3.0 3.1 "Tradjenta (linagliptin) Tablets. Full Prescribing Information" (PDF). Boehringer Ingelheim Pharmaceuticals, Inc. Ridgefield, CT 06877 USA. Archived (PDF) from the original on 6 November 2016. Retrieved 10 November 2016.
- ↑ 4.0 4.1 4.2 4.3 4.4 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 680. ISBN 9780857113382.
- ↑ 5.0 5.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 6.0 6.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 7.0 7.1 "Linagliptin - Drug Usage Statistics". ClinCalc. Archived from the original on 10 July 2020. Retrieved 11 April 2020.
- ↑ "Four Phase III Trials Confirm Benefits of BI's Oral, Once-Daily Type 2 Diabetes Therapy". Genetic Engineering & Biotechnology News. 28 June 2010. Archived from the original on 31 October 2018. Retrieved 12 September 2010.
- ↑ "DPP-4 Inhibitors for Type 2 Diabetes: Drug Safety Communication - May Cause Severe Joint Pain". FDA. 2015-08-28. Archived from the original on 2017-04-06. Retrieved 1 September 2015.
- ↑ https://docs.boehringer-ingelheim.com/Prescribing%20Information/PIs/Tradjenta/Tradjenta.pdf?DMW_FORMAT=pdf Archived 2020-06-01 at the Wayback Machine)
- ↑ Bronchial hyperresponsiveness
- ↑ "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary names: List 61" (PDF). World Health Organization. p. 66. Archived (PDF) from the original on 3 October 2020. Retrieved 10 November 2016.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Trajenta (Australia) Archived 2015-03-31 at the Wayback Machine
- "Trajenta EPAR". European Medicines Agency (EMA). Archived from the original on 2019-12-29. Retrieved 2020-02-11.
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- Drugs with non-standard legal status
- Articles with changed InChI identifier
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- All articles lacking reliable references
- Articles lacking reliable references from May 2019
- Articles with invalid date parameter in template
- Articles with changed ChemSpider identifier
- Articles with changed EBI identifier
- Alkyne derivatives
- Boehringer Ingelheim
- Dipeptidyl peptidase-4 inhibitors
- Eli Lilly and Company brands
- Piperidines
- Quinazolines
- Xanthines
- RTT

