Hepatitis C
| Hepatitis C | |
|---|---|
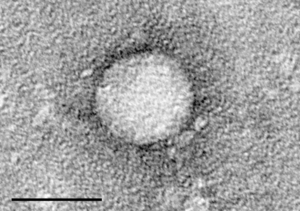 | |
| Electron micrograph of hepatitis C virus from cell culture (scale = 50 nanometers) | |
| Specialty | Gastroenterology, Infectious disease |
| Symptoms | Typically none[1] |
| Complications | Liver failure, liver cancer, esophageal and gastric varices[2] |
| Duration | Long term (80%)[1] |
| Causes | Hepatitis C virus usually spread by blood-to-blood contact[1][3] |
| Diagnostic method | Blood testing for antibodies or viral RNA[1] |
| Prevention | Clean needles, testing donated blood[4] |
| Treatment | Medications, liver transplant[5] |
| Medication | Antivirals (sofosbuvir, simeprevir, others)[1][4] |
| Frequency | 71 million (2017)[6] |
| Deaths | 399,000 (2016)[6] |
Hepatitis C is an infectious disease caused by the hepatitis C virus (HCV) that primarily affects the liver.[2] During the initial infection people often have mild or no symptoms.[1] Occasionally a fever, dark urine, abdominal pain, and yellow tinged skin occurs.[1] The virus persists in the liver in about 75% to 85% of those initially infected.[1] Early on chronic infection typically has no symptoms.[1] Over many years however, it often leads to liver disease and occasionally cirrhosis.[1] In some cases, those with cirrhosis will develop serious complications such as liver failure, liver cancer, or dilated blood vessels in the esophagus and stomach.[2]
HCV is spread primarily by blood-to-blood contact associated with intravenous drug use, poorly sterilized medical equipment, needlestick injuries in healthcare, and transfusions.[1][3] Using blood screening, the risk from a transfusion is less than one per two million.[1] It may also be spread from an infected mother to her baby during birth.[1] It is not spread by superficial contact.[4] It is one of five known hepatitis viruses: A, B, C, D, and E.[7] Diagnosis is by blood testing to look for either antibodies to the virus or its RNA.[1] Testing is recommended in all people who are at risk.[1]
There is no vaccine against hepatitis C.[1][8] Prevention includes harm reduction efforts among people who use intravenous drugs and testing donated blood.[4] Chronic infection can be cured more than 95% of the time with antiviral medications such as sofosbuvir or simeprevir.[6][1][4] Peginterferon and ribavirin were earlier generation treatments that had a cure rate of less than 50% and greater side effects.[4][9] Getting access to the newer treatments however can be expensive.[4] Those who develop cirrhosis or liver cancer may require a liver transplant.[5] Hepatitis C is the leading reason for liver transplantation, though the virus usually recurs after transplantation.[5]
An estimated 71 million people (1%) worldwide are infected with hepatitis C as of 2017[update].[6] In 2013, about eleven million new cases occurred.[10] It occurs most commonly in Africa and Central and East Asia.[4] About 167,000 deaths due to liver cancer and 326,000 deaths due to cirrhosis occurred in 2015 due to hepatitis C.[11] The existence of hepatitis C – originally identifiable only as a type of non-A non-B hepatitis – was suggested in the 1970s and proven in 1989.[12] Hepatitis C infects only humans and chimpanzees.[13]
Signs and symptoms
Acute infection
Acute symptoms develop in some 20–30% of those infected.[1] When this occurs, it is generally 4–12 weeks following infection (but it may take from 2 weeks to 6 months for acute symptoms to appear).[1][4]
Symptoms are generally mild and vague, and may include fatigue, nausea and vomiting, fever, muscle or joint pains, abdominal pain, decreased appetite and weight loss, jaundice (occurs in ~25% of those infected), dark urine, and clay-coloured stools.[1][14][15] There is no evidence that acute hepatitis C can alone cause acute liver failure, though liver injury and elevated liver enzymes may occur.[14][16] Symptoms and laboratory findings suggestive of liver disease should prompt further tests and can thus help establish a diagnosis of hepatitis C infection early on.[15]
Following the acute phase, the infection may resolve spontaneously in 10–50% of affected people; this occurs more frequently in young people, and females.[14]
Chronic infection

About 80% of those exposed to the virus develop a chronic infection.[17] This is defined as the presence of detectable viral replication for at least six months. Most experience minimal or no symptoms during the initial few decades of the infection.[18] Chronic hepatitis C can be associated with fatigue[19] and mild cognitive problems.[20] Chronic infection after several years may cause cirrhosis or liver cancer.[5] The liver enzymes are normal in 7–53%.[21] Late relapses after apparent cure have been reported, but these can be difficult to distinguish from reinfection.[21]
Fatty changes to the liver occur in about half of those infected and are usually present before cirrhosis develops.[22][23] Usually (80% of the time) this change affects less than a third of the liver.[22] Worldwide hepatitis C is the cause of 27% of cirrhosis cases and 25% of hepatocellular carcinoma.[24] About 10–30% of those infected develop cirrhosis over 30 years.[5][15] Cirrhosis is more common in those also infected with hepatitis B, schistosoma, or HIV, in alcoholics and in those of male sex.[15] In those with hepatitis C, excess alcohol increases the risk of developing cirrhosis 100-fold.[25] Those who develop cirrhosis have a 20-fold greater risk of hepatocellular carcinoma. This transformation occurs at a rate of 1–3% per year.[5][15] Being infected with hepatitis B in addition to hepatitis C increases this risk further.[26]
Liver cirrhosis may lead to portal hypertension, ascites (accumulation of fluid in the abdomen), easy bruising or bleeding, varices (enlarged veins, especially in the stomach and esophagus), jaundice, and a syndrome of cognitive impairment known as hepatic encephalopathy.[27] Ascites occurs at some stage in more than half of those who have a chronic infection.[28]
Extrahepatic complications
The most common problem due to hepatitis C but not involving the liver is mixed cryoglobulinemia (usually the type II form) – an inflammation of small and medium-sized blood vessels.[29][30] Hepatitis C is also associated with autoimmune disorders such as Sjögren's syndrome, lichen planus, a low platelet count, porphyria cutanea tarda, necrolytic acral erythema, insulin resistance, diabetes mellitus, diabetic nephropathy, autoimmune thyroiditis, and B-cell lymphoproliferative disorders.[31][32] 20–30% of people infected have rheumatoid factor – a type of antibody.[33] Possible associations include Hyde's prurigo nodularis[34] and membranoproliferative glomerulonephritis.[19] Cardiomyopathy with associated abnormal heart rhythms has also been reported.[35] A variety of central nervous system disorders has been reported.[36] Chronic infection seems to be associated with an increased risk of pancreatic cancer.[8][37] People may experience other issues in the mouth such as dryness, salivary duct stones, and crusted lesions around the mouth.[38][39][40]
Occult infection
Persons who have been infected with hepatitis C may appear to clear the virus but remain infected.[41] The virus is not detectable with conventional testing but can be found with ultra-sensitive tests.[42] The original method of detection was by demonstrating the viral genome within liver biopsies, but newer methods include an antibody test for the virus' core protein and the detection of the viral genome after first concentrating the viral particles by ultracentrifugation.[43] A form of infection with persistently moderately elevated serum liver enzymes but without antibodies to hepatitis C has also been reported.[44] This form is known as cryptogenic occult infection.
Several clinical pictures have been associated with this type of infection.[45] It may be found in people with anti-hepatitis-C antibodies but with normal serum levels of liver enzymes; in antibody-negative people with ongoing elevated liver enzymes of unknown cause; in healthy populations without evidence of liver disease; and in groups at risk for HCV infection including those on hemodialysis or family members of people with occult HCV. The clinical relevance of this form of infection is under investigation.[46] The consequences of occult infection appear to be less severe than with chronic infection but can vary from minimal to hepatocellular carcinoma.[43]
The rate of occult infection in those apparently cured is controversial but appears to be low.[21] 40% of those with hepatitis but with both negative hepatitis C serology and the absence of detectable viral genome in the serum have hepatitis C virus in the liver on biopsy.[47] How commonly this occurs in children is unknown.[48]
Virology
The hepatitis C virus (HCV) is a small, enveloped, single-stranded, positive-sense RNA virus.[5] It is a member of the genus Hepacivirus in the family Flaviviridae.[19] There are seven major genotypes of HCV, which are known as genotypes one to seven.[49] The genotypes are divided into several subtypes with the number of subtypes depending on the genotype. In the United States, about 70% of cases are caused by genotype 1, 20% by genotype 2 and about 1% by each of the other genotypes.[15] Genotype 1 is also the most common in South America and Europe.[5]
The half life of the virus particles in the serum is around 3 hours and may be as short as 45 minutes.[50][51] In an infected person, about 1012 virus particles are produced each day.[50] In addition to replicating in the liver the virus can multiply in lymphocytes.[52]
Transmission

The primary route of transmission in the developed world is intravenous drug use (IDU), while in the developing world the main methods are blood transfusions and unsafe medical procedures.[3] The cause of transmission remains unknown in 20% of cases;[53] however, many of these are believed to be accounted for by IDU.[14]
Drug use
Intravenous drug use (IDU) is a major risk factor for hepatitis C in many parts of the world.[54] Of 77 countries reviewed, 25 (including the United States) were found to have prevalences of hepatitis C in the intravenous drug user population of between 60% and 80%.[17][54] Twelve countries had rates greater than 80%.[17] It is believed that ten million intravenous drug users are infected with hepatitis C; China (1.6 million), the United States (1.5 million), and Russia (1.3 million) have the highest absolute totals.[17] Occurrence of hepatitis C among prison inmates in the United States is 10 to 20 times that of the occurrence observed in the general population; this has been attributed to high-risk behavior in prisons such as IDU and tattooing with nonsterile equipment.[55][56] Shared intranasal drug use may also be a risk factor.[57]
Healthcare exposure
Blood transfusion, transfusion of blood products, or organ transplants without HCV screening carry significant risks of infection.[15] The United States instituted universal screening in 1992[58] and Canada instituted universal screening in 1990.[59] This decreased the risk from one in 200 units[58] to between one in 10,000 to one in 10,000,000 per unit of blood.[14][53] This low risk remains as there is a period of about 11–70 days between the potential blood donor's acquiring hepatitis C and the blood's testing positive depending on the method.[53] Some countries do not screen for hepatitis C due to the cost.[24]
Those who have experienced a needle stick injury from someone who was HCV positive have about a 1.8% chance of subsequently contracting the disease themselves.[15] The risk is greater if the needle in question is hollow and the puncture wound is deep.[24] There is a risk from mucosal exposures to blood, but this risk is low, and there is no risk if blood exposure occurs on intact skin.[24]
Hospital equipment has also been documented as a method of transmission of hepatitis C, including reuse of needles and syringes; multiple-use medication vials; infusion bags; and improperly sterilized surgical equipment, among others.[24] Limitations in the implementation and enforcement of stringent standard precautions in public and private medical and dental facilities are known to be the primary cause of the spread of HCV in Egypt, the country with highest rate of infection in the world.[60]
Sexual intercourse
Sexual transmission of hepatitis C is uncommon.[9] Studies examining the risk of HCV transmission between heterosexual partners, when one is infected and the other is not, have found very low risks.[9] Sexual practices that involve higher levels of trauma to the anogenital mucosa, such as anal penetrative sex, or that occur when there is a concurrent sexually transmitted infection, including HIV or genital ulceration, present greater risks.[9][61] The United States Department of Veterans Affairs recommends condom use to prevent hepatitis C transmission in those with multiple partners, but not those in relationships that involve only a single partner.[62]
Body modification
Tattooing is associated with two to threefold increased risk of hepatitis C.[63] This can be due to either improperly sterilized equipment or contamination of the dyes being used.[63] Tattoos or piercings performed either before the mid-1980s, "underground," or nonprofessionally are of particular concern, since sterile techniques in such settings may be lacking. The risk also appears to be greater for larger tattoos.[63] It is estimated that nearly half of prison inmates share unsterilized tattooing equipment.[63] It is rare for tattoos in a licensed facility to be directly associated with HCV infection.[64]
Personal-care items such as razors, toothbrushes, and manicuring or pedicuring equipment can be contaminated with blood. Sharing such items can potentially lead to exposure to HCV.[65][66] Appropriate caution should be taken regarding any medical condition that results in bleeding, such as cuts and sores.[66] HCV is not spread through casual contact, such as hugging, kissing, or sharing eating or cooking utensils.[66] Neither is it transmitted through food or water.[67]
Mother-to-child
Mother-to-child transmission of hepatitis C occurs in fewer than 10% of pregnancies, though this may be up to 22% if co-infection with HIV.[68] It is not clear when transmission occurs during pregnancy, but it may occur both during gestation and at delivery.[53] A long labor is associated with a greater risk of transmission.[24] There is no evidence that breastfeeding spreads HCV; however, to be cautious, an infected mother is advised to avoid breastfeeding if her nipples are cracked and bleeding,[69] or if her viral loads are high.[53]
HIV
In terms of the mechanism of HIV/HCV coinfection, we find that liver fibrosis occurs via the relocation of microbial products originally in the intestinal lumen, now in the systemic circulation, and its direct effect via interaction with Kupffer cells or indirectly via initiation of systemic immune activation, to cause the aforementioned liver fibrosis.[70]
Diagnosis
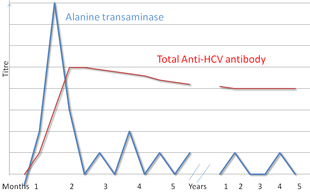
There are a number of diagnostic tests for hepatitis C, including HCV antibody enzyme immunoassay or ELISA, recombinant immunoblot assay, and quantitative HCV RNA polymerase chain reaction (PCR).[15] HCV RNA can be detected by PCR typically one to two weeks after infection, while antibodies can take substantially longer to form and thus be detected.[27]
Chronic hepatitis C is defined as infection with the hepatitis C virus persisting for more than six months based on the presence of its RNA.[18] Chronic infections are typically asymptomatic during the first few decades,[18] and thus are most commonly discovered following the investigation of elevated liver enzyme levels or during a routine screening of high-risk individuals. Testing is not able to distinguish between acute and chronic infections.[24] Diagnosis in the infant is difficult as maternal antibodies may persist for up to 18 months.[48]
Serology
Hepatitis C testing typically begins with blood testing to detect the presence of antibodies to the HCV, using an enzyme immunoassay.[15] If this test is positive, a confirmatory test is then performed to verify the immunoassay and to determine the viral load.[15] A recombinant immunoblot assay is used to verify the immunoassay and the viral load is determined by an HCV RNA polymerase chain reaction.[15] If there is no RNA and the immunoblot is positive, it means that the person tested had a previous infection but cleared it either with treatment or spontaneously; if the immunoblot is negative, it means that the immunoassay was wrong.[15] It takes about 6–8 weeks following infection before the immunoassay will test positive.[19] A number of tests are available as point of care testing which means that results are available within 30 minutes.[71]
Liver enzymes are variable during the initial part of the infection[18] and on average begin to rise at seven weeks after infection.[19] The elevation of liver enzymes does not closely follow disease severity.[19]
Biopsy
Liver biopsies are used to determine the degree of liver damage present; however, there are risks from the procedure.[5] The typical changes seen are lymphocytes within the parenchyma, lymphoid follicles in portal triad, and changes to the bile ducts.[5] There are a number of blood tests available that try to determine the degree of hepatic fibrosis and alleviate the need for biopsy.[5]
Screening
It is believed that only 5–50% of those infected in the United States and Canada are aware of their status.[63] Routine screening for those between the ages of 18 and 79 was recommended by the United States Preventive Services Task Force in 2020.[72] Previously testing was recommended for those at high risk, which includes injection drug users, those who have received blood transfusions before 1992,[73] those who have been in jail, those on long term hemodialysis,[74] and those with tattoos.[63] Screening is also recommended in those with elevated liver enzymes, as this is frequently the only sign of chronic hepatitis.[75] As of 2012[update], the U.S. Centers for Disease Control and Prevention (CDC) recommends a single screening test for those born between 1945 and 1965.[76][77][78][79] In Canada one time screening is recommended for those born between 1945 and 1975.[80]
Prevention
As of 2016[update], no approved vaccine protects against contracting hepatitis C.[81] A combination of harm reduction strategies, such as the provision of new needles and syringes and treatment of substance use, decreases the risk of hepatitis C in intravenous drug users by about 75%.[82] The screening of blood donors is important at a national level, as is adhering to universal precautions within healthcare facilities.[19] In countries where there is an insufficient supply of sterile syringes, medications should be given orally rather than via injection (when possible).[24] The use of medication to try to prevent the disease after exposure is not recommended as of 2020 by the CDC.[83]
Treatment
Those with chronic hepatitis C are advised to avoid alcohol and medications toxic to the liver.[15] They should also be vaccinated against hepatitis A and hepatitis B due to the increased risk if also infected.[15] Use of acetaminophen is generally considered safe at reduced doses.[9] Nonsteroidal anti-inflammatory drugs (NSAIDs) are not recommended in those with advanced liver disease due to an increased risk of bleeding.[9] Ultrasound surveillance for hepatocellular carcinoma is recommended in those with accompanying cirrhosis.[15] Coffee consumption has been associated with a slower rate of liver scarring in those infected with HCV.[9]
Medications
Approximately 90% of chronic cases clear with treatment.[4] Treatment with antiviral medication is recommended in all people with proven chronic hepatitis C who are not at high risk of dying from other causes.[84] People with the highest complication risk should be treated first, with the risk of complications based on the degree of liver scarring.[84] The initial recommended treatment depends on the type of hepatitis C virus, if the person has received previous hepatitis C treatment, and whether or not a person has cirrhosis.[85] Direct-acting antivirals (DAAs) may reduce the number of the infected people.[86]
No prior treatment
- HCV genotype 1a (no cirrhosis): 8 weeks of glecaprevir/pibrentasvir or ledipasvir/sofosbuvir (the latter for people who do not have HIV/AIDS, are not African-American, and have less than 6 million HCV viral copies per milliliter of blood) or 12 weeks of elbasvir/grazoprevir, ledipasvir/sofosbuvir, or sofosbuvir/velpatasvir.[87] Sofosbuvir with either daclatasvir or simeprevir may also be used.[85]
- HCV genotype 1a (with compensated cirrhosis): 12 weeks of elbasvir/grazoprevir, glecaprevir/pibrentasvir, ledipasvir/sofosbuvir, or sofosbuvir/velpatasvir. An alternative treatment regimen of elbasvir/grazoprevir with weight-based ribavirin for 16 weeks can be used if the HCV is found to have antiviral resistance mutations against NS5A protease inhibitors.[88]
- HCV genotype 1b (no cirrhosis): 8 weeks of glecaprevir/pibrentasvir or ledipasvir/sofosbuvir (with the aforementioned limitations for the latter as above) or 12 weeks of elbasvir/grazoprevir, ledipasvir/sofosbuvir, or sofosbuvir/velpatasvir. Alternative regimens include 12 weeks of ombitasvir/paritaprevir/ritonavir with dasabuvir or 12 weeks of sofosbuvir with either daclatasvir or simeprevir.[89]
- HCV genotype 1b (with compensated cirrhosis): 12 weeks of elbasvir/grazoprevir, glecaprevir/pibrentasvir, ledipasvir/sofosbuvir, or sofosbuvir/velpatasvir. A 12-week course of paritaprevir/ritonavir/ombitasvir with dasabuvir may also be used.[90]
- HCV genotype 2 (no cirrhosis): 8 weeks of glecaprevir/pibrentasvir or 12 weeks of sofosbuvir/velpatasvir. Alternatively, 12 weeks of sofosbuvir/daclatasvir can be used.[91]
- HCV genotype 2 (with compensated cirrhosis): 12 weeks of sofosbuvir/velpatasvir or glecaprevir/pibrentasvir. An alternative regimen of sofosbuvir/daclatasvir can be used for 16–24 weeks.[92]
- HCV genotype 3 (no cirrhosis): 8 weeks of glecaprevir/pibrentasvir or 12 weeks of sofosbuvir/velpatasvir or sofosbuvir and daclatasvir.[93]
- HCV genotype 3 (with compensated cirrhosis): 12 weeks of glecaprevir/pibrentasvir, sofosbuvir/velpatasvir, or if certain antiviral mutations are present, 12 weeks of sofosbuvir/velpatasvir/voxilaprevir (when certain antiviral mutations are present), or 24 weeks of sofosbuvir and daclatasvir.[94]
- HCV genotype 4 (no cirrhosis): 8 weeks of glecaprevir/pibrentasvir or 12 weeks of sofosbuvir/velpatasvir, elbasvir/grazoprevir, or ledipasvir/sofosbuvir. A 12-week regimen of ombitasvir/paritaprevir/ritonavir is also acceptable in combination with weight-based ribavirin.[95]
- HCV genotype 4 (with compensated cirrhosis): A 12-week regimen of sofosbuvir/velpatasvir, glecaprevir/pibrentasavir, elbasvir/grazoprevir, or ledipasvir/sofosbuvir is recommended. A 12-week course of ombitasvir/paritaprevir/ritonavir with weight-based ribavirin is an acceptable alternative.[96]
- HCV genotype 5 or 6 (with or without compensated cirrhosis): If no cirrhosis is present, then 8 weeks of glecaprevir/pibrentasvir is recommended. If cirrhosis is present, then a 12-week course of glecaprevir/pibrentasvir, sofosbuvir/velpatasvir, or ledipasvir/sofosbuvir is warranted.[97]
Chronic infection can be cured in more than 90% of people with medications.[98] Getting access to these treatments however can be expensive.[4] The combination of sofosbuvir, velpatasvir, and voxilaprevir may be used in those who have previously been treated with sofosbuvir or other drugs that inhibit NS5A and were not cured.[99]
Prior to 2011, treatments consisted of a combination of pegylated interferon alpha and ribavirin for a period of 24 or 48 weeks, depending on HCV genotype.[15] This produces cure rates of between 70 and 80% for genotype 2 and 3, respectively, and 45 to 70% for genotypes 1 and 4.[100] Adverse effects with these treatments were common, with half of people getting flu-like symptoms and a third experiencing emotional problems.[15] Treatment during the first six months is more effective than once hepatitis C has become chronic.[27] In those with chronic hepatitis B, treatment for hepatitis C results in reactivation of hepatitis B in about 25%.[101]
Surgery
Cirrhosis due to hepatitis C is a common reason for liver transplantation[27] though the virus usually (80–90% of cases) recurs afterwards.[5][102] Infection of the graft leads to 10–30% of people developing cirrhosis within five years.[103] Treatment with pegylated interferon and ribavirin post-transplant decreases the risk of recurrence to 70%.[104] A 2013 review found unclear evidence regarding if antiviral medication was useful if the graft became reinfected.[105]
Alternative medicine
Several alternative therapies are claimed by their proponents to be helpful for hepatitis C including milk thistle, ginseng, and colloidal silver.[106] However, no alternative therapy has been shown to improve outcomes in hepatitis C, and no evidence exists that alternative therapies have any effect on the virus at all.[106][107][108]
Prognosis

| no data <10 10–15 15–20 20–25 25–30 30–35 | 35–40 40–45 45–50 50–75 75–100 >100 |
The responses to treatment is measured by sustained viral response (SVR), defined as the absence of detectable RNA of the hepatitis C virus in blood serum for at least 24 weeks after discontinuing the treatment,[109] and rapid virological response (RVR) defined as undetectable levels achieved within four weeks of treatment. Successful treatment decreases the future risk of hepatocellular carcinoma by 75%.[110]
Prior to 2012 sustained response occurs in about 40–50% in people with HCV genotype 1 given 48 weeks of treatment.[5] A sustained response is seen in 70–80% of people with HCV genotypes 2 and 3 with 24 weeks of treatment.[5] A sustained response occurs about 65% in those with genotype 4 after 48 weeks of treatment. Finally for genotype 6, a 48 weeks treatment with pegylated interferon and ribavirin results in a higher rate in sustained responses than for genotype 1 (86% vs. 52%). Further studies are needed for a 24 weeks treatment and at lower dosages.[111]
Spontaneous resolution
About 15–45% of those infected spontaneously clear the virus within 6 months, with the rest going on the develop chronic infection.[4] Spontaneous resolution following acute infection appears more common in females, and younger persons, and appears to also be influenced by genetic factors.[14] Chronic infection may also resolve spontaneously months or years after the acute phase, though this is unusual.[14]
Epidemiology

It is estimated that 143 million people (2%) of people globally are living with chronic hepatitis C.[112] About 1.75 million people are infected per year, and about 400,000 people die yearly from hepatitis C-related diseases.[4] During 2010 it is estimated that 16,000 people died from acute infections while 196,000 deaths occurred from liver cancer secondary to the infection.[113] Rates have increased substantially in the 20th century due to a combination of intravenous drug abuse and reused but poorly sterilized medical equipment.[24]
Rates are high (>3.5% population infected) in Central and East Asia, North Africa and the Middle East, they are intermediate (1.5–3.5%) in South and Southeast Asia, sub-Saharan Africa, Andean, Central and Southern Latin America, Caribbean, Oceania, Australasia and Central, Eastern and Western Europe; and they are low (<1.5%) in Asia-Pacific, Tropical Latin America and North America.[114]
Among those chronically infected, the risk of cirrhosis after 20 years varies between studies but has been estimated at ~10–15% for men and ~1–5% for women. The reason for this difference is not known. Once cirrhosis is established, the rate of developing hepatocellular carcinoma is ~1–4% per year.[115] Rates of new infections have decreased in the Western world since the 1990s due to improved screening of blood before transfusion.[27]
In the United States, about 2% of people have chronic hepatitis C.[15] In 2014, an estimated 30,500 new acute hepatitis C cases occurred (0.7 per 100,000 population), an increase from 2010–2012.[116] The number of deaths from hepatitis C has increased to 15,800 in 2008[117] having overtaken HIV/AIDS as a cause of death in the US in 2007.[118] In 2014 it was the single greatest cause of infectious death in the United States.[119] This mortality rate is expected to increase, as those infected by transfusion before HCV testing become apparent.[120] In Europe the percentage of people with chronic infections has been estimated to be between 0.13 and 3.26%.[121]
In England about 160,000 people are chronically infected.[122] Between 2006 and 2011 28,000, about 3%, received treatment.[122] About half of people using a needle exchange in London in 2017/8 tested positive for hepatitis C of which half were unaware of they had it.[123] As part of a bid to eradicate hepatitis C by 2025 NHS England conducted a large procurement exercise in 2019. Merck Sharp & Dohme, Gilead Sciences, and Abbvie were awarded contracts, which, together, are worth up to £1 billion over five years.[124]
The total number of people with this infection is higher in some countries in Africa and Asia.[125] Countries with particularly high rates of infection include Egypt (22%), Pakistan (4.8%) and China (3.2%).[126] It is believed that the high prevalence in Egypt is linked to a now-discontinued mass-treatment campaign for schistosomiasis, using improperly sterilized glass syringes.[24]
Since 2014, extremely effective medication have been available to eradication the disease in 8–12 weeks in most people.[127] In 2015 about 950,000 people were treated while 1.7 million new infections occurred, meaning that overall the number of people with HCV increased.[127] These numbers differ by country and improved in 2016, with some countries achieving higher cure rates than new infection rates (mostly high income countries).[127] By 2018, twelve countries are on track to achieve HCV elimination.[127] While antiviral agents will curb new infections, it is less clear whether they impact overall deaths and morbidity.[127] Furthermore, for them to be effective, people need to be aware of their infection – it is currently estimated that worldwide only 20% of infected people are aware of their infection (in the US fewer than half were aware).[127]
History

In the mid-1970s, Harvey J. Alter, Chief of the Infectious Disease Section in the Department of Transfusion Medicine at the National Institutes of Health, and his research team demonstrated how most post-transfusion hepatitis cases were not due to hepatitis A or B viruses. Despite this discovery, international research efforts to identify the virus, initially called non-A, non-B hepatitis (NANBH), failed for the next decade. In 1987, Michael Houghton, Qui-Lim Choo, and George Kuo at Chiron Corporation, collaborating with Daniel W. Bradley at the Centers for Disease Control and Prevention, used a novel molecular cloning approach to identify the unknown organism and develop a diagnostic test.[128] In 1988, Alter confirmed the virus by verifying its presence in a panel of NANBH specimens. In April 1989, the discovery of HCV was published in two articles in the journal Science.[129][130] The discovery led to significant improvements in diagnosis and improved antiviral treatment.[8][128] In 2000, Drs. Alter and Houghton were honored with the Lasker Award for Clinical Medical Research for "pioneering work leading to the discovery of the virus that causes hepatitis C and the development of screening methods that reduced the risk of blood transfusion-associated hepatitis in the U.S. from 30% in 1970 to virtually zero in 2000."[131] In 2020 Harvey J. Alter, Michael Houghton and Charles M. Rice received the Nobel Prize in Physiology and Medicine for this work.[132]
Chiron filed for several patents on the virus and its diagnosis.[133] A competing patent application by the CDC was dropped in 1990 after Chiron paid $1.9 million to the CDC and $337,500 to Bradley. In 1994, Bradley sued Chiron, seeking to invalidate the patent, have himself included as a coinventor, and receive damages and royalty income. He dropped the suit in 1998 after losing before an appeals court.[134]
Society and culture
World Hepatitis Day, held on July 28, is coordinated by the World Hepatitis Alliance.[135] The economic costs of hepatitis C are significant both to the individual and to society. In the United States the average lifetime cost of the disease was estimated at US$33,407 in 2003[136] with the cost of a liver transplant as of 2011[update] costing approximately US$200,000.[137] In Canada the cost of a course of antiviral treatment is as high as 30,000 CAD in 2003,[138] while the United States costs are between 9,200 and 17,600 in 1998 USD.[136] In many areas of the world, people are unable to afford treatment with antivirals as they either lack insurance coverage or the insurance they have will not pay for antivirals.[139] In the English National Health Service treatment rates for hepatitis C are higher among wealthier groups per 2010–2012 data.[122] Spanish anaesthetist Juan Maeso infected 275 patients between 1988 and 1997 as he used the same needles to give both himself and the patients opioids.[140] For this he was jailed.[141]
Special populations
Children and pregnancy
Compared with adults, infection in children is much less well understood. Worldwide the prevalence of hepatitis C virus infection in pregnant women and children has been estimated to 1–8% and 0.05–5% respectively.[142] The vertical transmission rate has been estimated to be 3–5% and there is a high rate of spontaneous clearance (25–50%) in the children. Higher rates have been reported for both vertical transmission (18%, 6–36% and 41%).[143][144] and prevalence in children (15%).[145]
In developed countries transmission around the time of birth is now the leading cause of HCV infection. In the absence of virus in the mother's blood transmission seems to be rare.[144] Factors associated with an increased rate of infection include membrane rupture of longer than 6 hours before delivery and procedures exposing the infant to maternal blood.[146] Cesarean sections are not recommended. Breastfeeding is considered safe if the nipples are not damaged. Infection around the time of birth in one child does not increase the risk in a subsequent pregnancy. All genotypes appear to have the same risk of transmission.
HCV infection is frequently found in children who have previously been presumed to have non-A, non-B hepatitis and cryptogenic liver disease.[147] The presentation in childhood may be asymptomatic or with elevated liver function tests.[148] While infection is commonly asymptomatic both cirrhosis with liver failure and hepatocellular carcinoma may occur in childhood.
Immunosuppressed
The rate of hepatitis C in immunosuppressed people is higher. This is particularly true in those with human immunodeficiency virus infection, recipients of organ transplants, and those with hypogammaglobulinemia.[149] Infection in these people is associated with an unusually rapid progression to cirrhosis. People with stable HIV who never received medication for HCV, may be treated with a combination of peginterferon plus ribavirin with caution to the possible side effects.[150]
Research
As of 2011[update], there are about one hundred medications in development for hepatitis C.[137] These include vaccines to treat hepatitis, immunomodulators, and cyclophilin inhibitors, among others.[151] These potential new treatments have come about due to a better understanding of the hepatitis C virus.[152] There are a number of vaccines under development and some have shown encouraging results.[81]
The combination of sofosbuvir and velpatasvir in one trial (reported in 2015) resulted in cure rates of 99%.[153] More studies are needed to investigate the role of the preventive antiviral medication against HCV recurrence after transplantation.[154]
Animal models
One barrier to finding treatments for hepatitis C is the lack of a suitable animal model. Despite moderate success, current research highlights the need for pre-clinical testing in mammalian systems such as mouse, particularly for the development of vaccines in poorer communities. Currently, chimpanzees remain the available living system to study, yet their use has ethical concerns and regulatory restrictions. While scientists have made use of human cell culture systems such as hepatocytes, questions have been raised about their accuracy in reflecting the body's response to infection.[155]
One aspect of hepatitis research is to reproduce infections in mammalian models. A strategy is to introduce liver tissues from humans into mice, a technique known as xenotransplantation. This is done by generating chimeric mice, and exposing the mice HCV infection. This engineering process is known to create humanized mice, and provide opportunities to study hepatitis C within the 3D architectural design of the liver and evaluating antiviral compounds.[155] Alternatively, generating inbred mice with susceptibility to HCV would simplify the process of studying mouse models.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 "Hepatitis C FAQs for Health Professionals". CDC. January 8, 2016. Archived from the original on 21 January 2016. Retrieved 4 February 2016.
- ↑ 2.0 2.1 2.2 Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 551–52. ISBN 978-0-8385-8529-0.
- ↑ 3.0 3.1 3.2 Maheshwari, A; Thuluvath, PJ (February 2010). "Management of acute hepatitis C". Clinics in Liver Disease. 14 (1): 169–76, x. doi:10.1016/j.cld.2009.11.007. PMID 20123448.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 "Hepatitis C Fact sheet N°164". WHO. July 2015. Archived from the original on 31 January 2016. Retrieved 4 February 2016.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 Rosen, HR (2011-06-23). "Clinical practice. Chronic hepatitis C infection". The New England Journal of Medicine. 364 (25): 2429–38. doi:10.1056/NEJMcp1006613. PMID 21696309. Archived from the original on 2021-08-28. Retrieved 2020-07-28.
- ↑ 6.0 6.1 6.2 6.3 "Hepatitis C". World Health Organization. 9 July 2019. Archived from the original on 2020-05-26. Retrieved 2020-05-26.
- ↑ "Viral Hepatitis: A through E and Beyond". National Institute of Diabetes and Digestive and Kidney Diseases. April 2012. Archived from the original on 2 February 2016. Retrieved 4 February 2016.
- ↑ 8.0 8.1 8.2 Webster, Daniel P; Klenerman, Paul; Dusheiko, Geoffrey M (2015). "Hepatitis C". The Lancet. 385 (9973): 1124–35. doi:10.1016/S0140-6736(14)62401-6. ISSN 0140-6736. PMC 4878852. PMID 25687730.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 Kim, A (September 2016). "Hepatitis C Virus". Annals of Internal Medicine (Review). 165 (5): ITC33–ITC48. doi:10.7326/AITC201609060. PMID 27595226.
- ↑ Global Burden of Disease Study 2013, Collaborators (22 August 2015). "Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013". Lancet. 386 (9995): 743–800. doi:10.1016/s0140-6736(15)60692-4. PMC 4561509. PMID 26063472.
- ↑ GBD 2015 Mortality and Causes of Death, Collaborators. (8 October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/S0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
- ↑ Houghton M (November 2009). "The long and winding road leading to the identification of the hepatitis C virus". Journal of Hepatology. 51 (5): 939–48. doi:10.1016/j.jhep.2009.08.004. PMID 19781804. Archived from the original on 2021-12-17. Retrieved 2020-07-28.
- ↑ Shors, Teri (2011). Understanding viruses (2nd ed.). Burlington, MA: Jones & Bartlett Learning. p. 535. ISBN 978-0-7637-8553-6. Archived from the original on 2016-05-15.
- ↑ 14.0 14.1 14.2 14.3 14.4 14.5 14.6 Chronic Hepatitis C Virus Advances in Treatment, Promise for the Future. Springer Verlag. 2011. p. 14. ISBN 978-1-4614-1191-8. Archived from the original on 2016-06-17.
- ↑ 15.00 15.01 15.02 15.03 15.04 15.05 15.06 15.07 15.08 15.09 15.10 15.11 15.12 15.13 15.14 15.15 15.16 15.17 15.18 Wilkins, T; Malcolm JK; Raina D; Schade RR (2010-06-01). "Hepatitis C: diagnosis and treatment" (PDF). American Family Physician. 81 (11): 1351–57. PMID 20521755. Archived (PDF) from the original on 2013-05-21.
- ↑ Manka, Paul; Verheyen, Jens; Gerken, Guido; Canbay, Ali (April 2016). "Liver Failure due to Acute Viral Hepatitis (A–E)". Visceral Medicine. 32 (2): 80–85. doi:10.1159/000444915. ISSN 2297-4725. PMC 4926881. PMID 27413724.
- ↑ 17.0 17.1 17.2 17.3 Nelson, PK; Mathers BM; Cowie B; Hagan H; Des Jarlais D; Horyniak D; Degenhardt L (2011-08-13). "Global epidemiology of hepatitis B and hepatitis C in people who inject drugs: results of systematic reviews". Lancet. 378 (9791): 571–83. doi:10.1016/S0140-6736(11)61097-0. PMC 3285467. PMID 21802134.
- ↑ 18.0 18.1 18.2 18.3 Chronic Hepatitis C Virus Advances in Treatment, Promise for the Future. Springer Verlag. 2011. pp. 103–04. ISBN 978-1-4614-1191-8. Archived from the original on 2016-05-29.
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 Ray, Stuart C.; Thomas, David L. (2009). "Chapter 154: Hepatitis C". In Mandell, Gerald L.; Bennett, John E.; Dolin, Raphael (eds.). Mandell, Douglas, and Bennett's principles and practice of infectious diseases (7th ed.). Philadelphia, PA: Churchill Livingstone. ISBN 978-0-443-06839-3.
- ↑ Forton, DM; Allsop, JM; Cox, IJ; Hamilton, G; Wesnes, K; Thomas, HC; Taylor-Robinson, SD (October 2005). "A review of cognitive impairment and cerebral metabolite abnormalities in patients with hepatitis C infection". AIDS. 19 (Suppl 3): S53–63. doi:10.1097/01.aids.0000192071.72948.77. PMID 16251829.
- ↑ 21.0 21.1 21.2 Nicot, F (2004). "Chapter 19. Liver biopsy in modern medicine.". Occult hepatitis C virus infection: Where are we now?. ISBN 978-953-307-883-0.
- ↑ 22.0 22.1 El-Zayadi, AR (2008-07-14). "Hepatic steatosis: a benign disease or a silent killer". World Journal of Gastroenterology. 14 (26): 4120–26. doi:10.3748/wjg.14.4120. PMC 2725370. PMID 18636654.
- ↑ Paradis, V; Bedossa, P (December 2008). "Definition and natural history of metabolic steatosis: histology and cellular aspects". Diabetes & Metabolism. 34 (6 Pt 2): 638–42. doi:10.1016/S1262-3636(08)74598-1. PMID 19195624.
- ↑ 24.0 24.1 24.2 24.3 24.4 24.5 24.6 24.7 24.8 24.9 Alter, MJ (2007-05-07). "Epidemiology of hepatitis C virus infection". World Journal of Gastroenterology. 13 (17): 2436–41. doi:10.3748/wjg.v13.i17.2436. PMC 4146761. PMID 17552026.
- ↑ Mueller, S; Millonig G; Seitz HK (2009-07-28). "Alcoholic liver disease and hepatitis C: a frequently underestimated combination". World Journal of Gastroenterology. 15 (28): 3462–71. doi:10.3748/wjg.15.3462. PMC 2715970. PMID 19630099. Archived from the original on 2020-07-11. Retrieved 10 July 2020.
- ↑ Fattovich, G; Stroffolini, T; Zagni, I; Donato, F (November 2004). "Hepatocellular carcinoma in cirrhosis: incidence and risk factors". Gastroenterology. 127 (5 Suppl 1): S35–50. doi:10.1053/j.gastro.2004.09.014. PMID 15508101.
- ↑ 27.0 27.1 27.2 27.3 27.4 Ozaras, R; Tahan, V (April 2009). "Acute hepatitis C: prevention and treatment". Expert Review of Anti-infective Therapy. 7 (3): 351–61. doi:10.1586/eri.09.8. PMID 19344247.
- ↑ Zaltron, S; Spinetti, A; Biasi, L; Baiguera, C; Castelli, F (2012). "Chronic HCV infection: epidemiological and clinical relevance". BMC Infectious Diseases. 12 Suppl 2: S2. doi:10.1186/1471-2334-12-S2-S2. PMC 3495628. PMID 23173556.
- ↑ Dammacco F, Sansonno D (September 12, 2013). "Review Article: Therapy for Hepatitis C Virus–Related Cryoglobulinemic Vasculitis". N Engl J Med. 369 (11): 1035–45. doi:10.1056/NEJMra1208642. PMID 24024840. Archived from the original on August 28, 2021. Retrieved July 28, 2020.
- ↑ Iannuzzella, F; Vaglio, A; Garini, G (May 2010). "Management of hepatitis C virus-related mixed cryoglobulinemia". Am. J. Med. 123 (5): 400–08. doi:10.1016/j.amjmed.2009.09.038. PMID 20399313.
- ↑ Zignego, AL; Ferri, C; Pileri, SA; et al. (January 2007). "Extrahepatic manifestations of Hepatitis C Virus infection: a general overview and guidelines for a clinical approach". Digestive and Liver Disease. 39 (1): 2–17. doi:10.1016/j.dld.2006.06.008. PMID 16884964.
- ↑ Ko, HM; Hernandez-Prera, JC; Zhu, H; Dikman, SH; Sidhu, HK; Ward, SC; Thung, SN (2012). "Morphologic features of extrahepatic manifestations of hepatitis C virus infection". Clinical & Developmental Immunology. 2012: 1–9. doi:10.1155/2012/740138. PMC 3420144. PMID 22919404.
- ↑ Dammacco, F; Sansonno, D; Piccoli, C; Racanelli, V; D'Amore, FP; Lauletta, G (2000). "The lymphoid system in hepatitis C virus infection: autoimmunity, mixed cryoglobulinemia, and Overt B-cell malignancy". Seminars in Liver Disease. 20 (2): 143–57. doi:10.1055/s-2000-9613. PMID 10946420.
- ↑ Lee, MR; Shumack, S (November 2005). "Prurigo nodularis: a review". The Australasian Journal of Dermatology. 46 (4): 211–18, quiz 219–20. doi:10.1111/j.1440-0960.2005.00187.x. PMID 16197418.
- ↑ Matsumori, A (2006). Role of hepatitis C virus in cardiomyopathies. Ernst Schering Research Foundation Workshop. Vol. 55. pp. 99–120. doi:10.1007/3-540-30822-9_7. ISBN 978-3-540-23971-0. PMID 16329660.
- ↑ Monaco, S; Ferrari, S; Gajofatto, A; Zanusso, G; Mariotto, S (2012). "HCV-related nervous system disorders". Clinical & Developmental Immunology. 2012: 1–9. doi:10.1155/2012/236148. PMC 3414089. PMID 22899946.
- ↑ Xu, JH; Fu, JJ; Wang, XL; Zhu, JY; Ye, XH; Chen, SD (2013-07-14). "Hepatitis B or C viral infection and risk of pancreatic cancer: A meta-analysis of observational studies". World Journal of Gastroenterology. 19 (26): 4234–41. doi:10.3748/wjg.v19.i26.4234. PMC 3710428. PMID 23864789.
- ↑ Lodi, G.; Porter, S.R; Scully, C. (1998-07-01). "Hepatitis C virus infection: Review and implications for the dentist". Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 86 (1): 8–22. doi:10.1016/S1079-2104(98)90143-3. ISSN 1079-2104. PMID 9690239.
- ↑ Carrozzo, M.; Gandolfo, S. (2003-03-01). "Oral Diseases Possibly Associated with Hepatitis C Virus". Critical Reviews in Oral Biology & Medicine. 14 (2): 115–27. doi:10.1177/154411130301400205. ISSN 1045-4411. PMID 12764074.
- ↑ Little, James W.; Falace, Donald A.; Miller, Craig; Rhodus, Nelson L. (2013). Dental Management of the Medically Compromised Patient. p. 151. ISBN 978-0323080286.
- ↑ Sugden, PB; Cameron, B; Bull, R; White, PA; Lloyd, AR (September 2012). "Occult infection with hepatitis C virus: friend or foe?". Immunology and Cell Biology. 90 (8): 763–73. doi:10.1038/icb.2012.20. PMID 22546735.
- ↑ Carreño, V (2006-11-21). "Occult hepatitis C virus infection: a new form of hepatitis C." World Journal of Gastroenterology. 12 (43): 6922–25. doi:10.3748/wjg.12.6922. PMC 4087333. PMID 17109511.
- ↑ 43.0 43.1 Carreño García, V; Nebreda, JB; Aguilar, IC; Quiroga Estévez, JA (March 2011). "[Occult hepatitis C virus infection]". Enfermedades Infecciosas y Microbiologia Clinica. 29 Suppl 3: 14–19. doi:10.1016/S0213-005X(11)70022-2. PMID 21458706.
- ↑ Pham, TN; Coffin, CS; Michalak, TI (April 2010). "Occult hepatitis C virus infection: what does it mean?". Liver International. 30 (4): 502–11. doi:10.1111/j.1478-3231.2009.02193.x. PMID 20070513.
- ↑ Carreño, V; Bartolomé, J; Castillo, I; Quiroga, JA (2012-06-21). "New perspectives in occult hepatitis C virus infection". World Journal of Gastroenterology. 18 (23): 2887–94. doi:10.3748/wjg.v18.i23.2887. PMC 3380315. PMID 22736911.
- ↑ Carreño, V; Bartolomé, J; Castillo, I; Quiroga, JA (May–June 2008). "Occult hepatitis B virus and hepatitis C virus infections". Reviews in Medical Virology. 18 (3): 139–57. doi:10.1002/rmv.569. PMID 18265423.
- ↑ Scott, JD; Gretch, DR (2007-02-21). "Molecular diagnostics of hepatitis C virus infection: a systematic review". JAMA: The Journal of the American Medical Association. 297 (7): 724–32. doi:10.1001/jama.297.7.724. PMID 17312292.
- ↑ 48.0 48.1 Robinson, JL (July 4, 2008). "Vertical transmission of the hepatitis C virus: Current knowledge and issues". Paediatr Child Health. 13 (6): 529–34. doi:10.1093/pch/13.6.529. PMC 2532905. PMID 19436425.
- ↑ Nakano, T; Lau, GM; Lau, GM; et al. (December 2011). "An updated analysis of hepatitis C virus genotypes and subtypes based on the complete coding region". Liver Int. 32 (2): 339–45. doi:10.1111/j.1478-3231.2011.02684.x. PMID 22142261.
- ↑ 50.0 50.1 Lerat, H; Hollinger, FB (2004-01-01). "Hepatitis C virus (HCV) occult infection or occult HCV RNA detection?". The Journal of Infectious Diseases. 189 (1): 3–6. doi:10.1086/380203. PMID 14702146.
- ↑ Pockros, Paul (2011). Novel and Combination Therapies for Hepatitis C Virus, An Issue of Clinics in Liver Disease. p. 47. ISBN 978-1-4557-7198-1. Archived from the original on 2016-05-21.
- ↑ Zignego, AL; Giannini, C; Gragnani, L; Piluso, A; Fognani, E (2012-08-03). "Hepatitis C virus infection in the immunocompromised host: a complex scenario with variable clinical impact". Journal of Translational Medicine. 10 (1): 158. doi:10.1186/1479-5876-10-158. PMC 3441205. PMID 22863056.
- ↑ 53.0 53.1 53.2 53.3 53.4 Pondé, RA (February 2011). "Hidden hazards of HCV transmission". Medical Microbiology and Immunology. 200 (1): 7–11. doi:10.1007/s00430-010-0159-9. PMID 20461405.
- ↑ 54.0 54.1 Xia, X; Luo J; Bai J; Yu R (October 2008). "Epidemiology of HCV infection among injection drug users in China: systematic review and meta-analysis". Public Health. 122 (10): 990–1003. doi:10.1016/j.puhe.2008.01.014. PMID 18486955.
- ↑ Imperial, JC (June 2010). "Chronic hepatitis C in the state prison system: insights into the problems and possible solutions". Expert Review of Gastroenterology & Hepatology. 4 (3): 355–64. doi:10.1586/egh.10.26. PMID 20528122.
- ↑ Vescio, MF; Longo B; Babudieri S; Starnini G; Carbonara S; Rezza G; Monarca R (April 2008). "Correlates of hepatitis C virus seropositivity in prison inmates: a meta-analysis". Journal of Epidemiology and Community Health. 62 (4): 305–13. doi:10.1136/jech.2006.051599. PMID 18339822.
- ↑ Moyer, VA; U.S. Preventive Services Task, Force (3 September 2013). "Screening for hepatitis C virus infection in adults: U.S. Preventive Services Task Force recommendation statement". Annals of Internal Medicine. 159 (5): 349–57. doi:10.7326/0003-4819-159-5-201309030-00672. PMID 23798026. Archived from the original on 28 August 2021. Retrieved 28 July 2020.
- ↑ 58.0 58.1 Marx, John (2010). Rosen's emergency medicine: concepts and clinical practice 7th edition. Philadelphia, PA: Mosby/Elsevier. p. 1154. ISBN 978-0-323-05472-0.
- ↑ Day RA, Paul P, Williams B, et al. (2009). Brunner & Suddarth's textbook of Canadian medical-surgical nursing (Canadian 2nd ed.). Philadelphia, PA: Lippincott Williams & Wilkins. p. 1237. ISBN 978-0-7817-9989-8. Archived from the original on 2016-04-25.
- ↑ "Highest Rates of Hepatitis C Virus Transmission Found in Egypt". Al Bawaaba. 2010-08-09. Archived from the original on 2012-05-15. Retrieved 2010-08-27.
- ↑ Tohme RA, Holmberg SD (June 2010). "Is sexual contact a major mode of hepatitis C virus transmission?". Hepatology. 52 (4): 1497–505. doi:10.1002/hep.23808. PMID 20635398.
- ↑ "Hepatitis C Group Education Class". United States Department of Veteran Affairs. Archived from the original on 2011-11-09.
- ↑ 63.0 63.1 63.2 63.3 63.4 63.5 Jafari, S; Copes R; Baharlou S; Etminan M; Buxton J (November 2010). "Tattooing and the risk of transmission of hepatitis C: a systematic review and meta-analysis" (PDF). International Journal of Infectious Diseases. 14 (11): e928–40. doi:10.1016/j.ijid.2010.03.019. PMID 20678951. Archived (PDF) from the original on 2012-04-26.
- ↑ "Hepatitis C" (PDF). Centers for Disease Control and Prevention (CDC). Archived (PDF) from the original on 5 January 2012. Retrieved 2 January 2012.
- ↑ Lock G; Dirscherl M; Obermeier F; et al. (September 2006). "Hepatitis C – contamination of toothbrushes: myth or reality?". J. Viral Hepat. 13 (9): 571–73. doi:10.1111/j.1365-2893.2006.00735.x. PMID 16907842.
- ↑ 66.0 66.1 66.2 "Hepatitis C FAQs for Health Professionals". Centers for Disease Control and Prevention (CDC). Archived from the original on 4 January 2012. Retrieved 2 January 2012.
- ↑ Wong T, Lee SS (February 2006). "Hepatitis C: a review for primary care physicians". CMAJ. 174 (5): 649–59. doi:10.1503/cmaj.1030034. PMC 1389829. PMID 16505462.
- ↑ Rosene-Montella, Karen (2020). "226. Common medical problems in pregnancy". In Goldman, Lee; Schafer, Andrew I. (eds.). Goldman-Cecil Medicine. Vol. 2 (26th ed.). Philadelphia: Elsevier. pp. 1587–1588. ISBN 978-0-323-55087-1. Archived from the original on 2023-10-20. Retrieved 2023-10-08.
- ↑ Mast EE (2004). Mother-to-infant hepatitis C virus transmission and breastfeeding. Advances in Experimental Medicine and Biology. Vol. 554. pp. 211–16. doi:10.1007/978-1-4757-4242-8_18. ISBN 978-1-4419-3461-1. PMID 15384578.
- ↑ Page, Emma E.; Nelson, Mark; Kelleher, Peter (November 2011). "HIV and hepatitis C coinfection: pathogenesis and microbial translocation". Current opinion in HIV and AIDS. 6 (6): 472–477. doi:10.1097/COH.0b013e32834bbc71. ISSN 1746-6318. Archived from the original on 23 November 2020. Retrieved 2 November 2021.
- ↑ Shivkumar, S; Peeling, R; Jafari, Y; Joseph, L; Pant Pai, N (2012-10-16). "Accuracy of Rapid and Point-of-Care Screening Tests for Hepatitis C: A Systematic Review and Meta-analysis". Annals of Internal Medicine. 157 (8): 558–66. doi:10.7326/0003-4819-157-8-201210160-00006. PMID 23070489. Archived from the original on 2021-08-28. Retrieved 2020-07-28.
- ↑ Owens, Douglas K.; Davidson, Karina W.; Krist, Alex H.; Barry, Michael J.; Cabana, Michael; Caughey, Aaron B.; Donahue, Katrina; Doubeni, Chyke A.; Epling, John W.; Kubik, Martha; Ogedegbe, Gbenga; Pbert, Lori; Silverstein, Michael; Simon, Melissa A.; Tseng, Chien-Wen; Wong, John B. (2 March 2020). "Screening for Hepatitis C Virus Infection in Adolescents and Adults". JAMA. 323 (10): 970. doi:10.1001/jama.2020.1123. PMID 32119076.
- ↑ Moyer, VA (on behalf of the U.S. Preventive Services Task Force) (2013-06-25). "Screening for Hepatitis C Virus Infection in Adults: U.S. Preventive Services Task Force Recommendation Statement". Annals of Internal Medicine. 159 (5): 349–57. doi:10.7326/0003-4819-159-5-201309030-00672. PMID 23798026. Archived from the original on 2021-08-28. Retrieved 2020-07-28.
- ↑ Moyer, VA (U.S. Preventive Services Task Force) (2013-09-03). "Screening for hepatitis C virus infection in adults: U.S. Preventive Services Task Force recommendation statement". Annals of Internal Medicine. 159 (5): 349–57. doi:10.7326/0003-4819-159-5-201309030-00672. PMID 23798026. Archived from the original on 2021-08-28. Retrieved 2020-07-28.
- ↑ Senadhi, V (July 2011). "A paradigm shift in the outpatient approach to liver function tests". Southern Medical Journal. 104 (7): 521–25. doi:10.1097/SMJ.0b013e31821e8ff5. PMID 21886053.
- ↑ Smith BD, Morgan RL, Beckett GA, et al. (August 2012). "Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945–1965" (PDF). MMWR Recomm Rep. 61 (RR-4): 1–32. PMID 22895429. Archived (PDF) from the original on 2020-07-14. Retrieved 2020-07-28.
- ↑ "Testing Recommendations for Hepatitis C Virus Infection – HCV – Division of Viral Hepatitis". U.S. Centers for Disease Control and Prevention (CDC). 12 June 2019. Archived from the original on 25 December 2019. Retrieved 11 January 2020.
- ↑ "People Born 1945–1965 (Baby Boomers) – Populations and Settings – Division of Viral Hepatitis". U.S. Centers for Disease Control and Prevention (CDC). 26 July 2019. Archived from the original on 22 October 2019. Retrieved 11 January 2020.
- ↑ "Final Update Summary: Hepatitis C: Screening". US Preventive Services Task Force. Archived from the original on 25 December 2019. Retrieved 11 January 2020.
- ↑ Shah, Hemant; Bilodeau, Marc; Burak, Kelly W.; Cooper, Curtis; Klein, Marina; Ramji, Alnoor; Smyth, Dan; Feld, Jordan J. (4 June 2018). "The management of chronic hepatitis C: 2018 guideline update from the Canadian Association for the Study of the Liver". CMAJ. 190 (22): E677–87. doi:10.1503/cmaj.170453. ISSN 0820-3946. PMC 5988519. PMID 29866893.
- ↑ 81.0 81.1 Abdelwahab, KS; Ahmed Said, ZN (14 January 2016). "Status of hepatitis C virus vaccination: Recent update". World Journal of Gastroenterology. 22 (2): 862–73. doi:10.3748/wjg.v22.i2.862. PMC 4716084. PMID 26811632.
- ↑ Hagan, H; Pouget, ER; Des Jarlais, DC (2011-07-01). "A systematic review and meta-analysis of interventions to prevent hepatitis C virus infection in people who inject drugs". The Journal of Infectious Diseases. 204 (1): 74–83. doi:10.1093/infdis/jir196. PMC 3105033. PMID 21628661.
- ↑ Moorman, Anne C.; de Perio, Marie A.; Goldschmidt, Ronald; Chu, Carolyn; Kuhar, David; Henderson, David K.; Naggie, Susanna; Kamili, Saleem; Spradling, Philip R.; Gordon, Stuart C.; Russi, Mark B.; Teshale, Eyasu H. (24 July 2020). "Testing and Clinical Management of Health Care Personnel Potentially Exposed to Hepatitis C Virus — CDC Guidance, United States, 2020". MMWR. Recommendations and Reports. 69 (6): 1–8. doi:10.15585/mmwr.rr6906a1.
- ↑ 84.0 84.1 AASLD/IDSA HCV Guidance, Panel (September 2015). "Hepatitis C guidance: AASLD-IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus". Hepatology. 62 (3): 932–54. doi:10.1002/hep.27950. PMID 26111063.
- ↑ 85.0 85.1 "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C" (PDF). 12 April 2017. Archived from the original (PDF) on 2017-07-10. Retrieved 28 July 2017.
- ↑ Jakobsen, JC; Nielsen, EE; Feinberg, J; Katakam, KK; Fobian, K; Hauser, G; Poropat, G; Djurisic, S; Weiss, KH; Bjelakovic, M; Bjelakovic, G; Klingenberg, SL; Liu, JP; Nikolova, D; Koretz, RL; Gluud, C (18 September 2017). "Direct-acting antivirals for chronic hepatitis C." The Cochrane Database of Systematic Reviews. 9: CD012143. doi:10.1002/14651858.CD012143.pub3. PMC 6484376. PMID 28922704.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 1a Without Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 1a With Compensated Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 1b Without Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 1b With Compensated Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 2 Without Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 2 With Compensated Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 3 Without Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 3 With Compensated Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 4 Without Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 4 With Compensated Cirrhosis". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C: Treatment – Naive Genotype 5 or 6". www.hcvguidelines.org. American Association for the Study of Liver Diseases. Archived from the original on 7 July 2017. Retrieved 26 April 2017.
- ↑ "Hepatitis C Questions and Answers for Health Professionals". www.cdc.gov. 2 July 2019. Archived from the original on 4 January 2012. Retrieved 23 July 2019.
- ↑ "FDA approves Vosevi for Hepatitis C". U.S. Food and Drug Administration (FDA) (Press release). 18 July 2017. Archived from the original on 23 July 2017. Retrieved 27 July 2017.
- ↑ Liang, TJ; Ghany, MG (May 16, 2013). "Current and future therapies for hepatitis C virus infection". The New England Journal of Medicine. 368 (20): 1907–17. doi:10.1056/NEJMra1213651. PMC 3893124. PMID 23675659.
- ↑ Mücke, Marcus M; Backus, Lisa I; Mücke, Victoria T; Coppola, Nicola; Preda, Carmen M; Yeh, Ming-Lun; Tang, Lydia S Y; Belperio, Pamela S; Wilson, Eleanor M; Yu, Ming-Lung; Zeuzem, Stefan; Herrmann, Eva; Vermehren, Johannes (January 2018). "Hepatitis B virus reactivation during direct-acting antiviral therapy for hepatitis C: a systematic review and meta-analysis". The Lancet Gastroenterology & Hepatology. 3 (3): 172–80. doi:10.1016/S2468-1253(18)30002-5. PMID 29371017.
- ↑ Sanders, Mick (2011). Mosby's Paramedic Textbook. Jones & Bartlett Publishers. p. 839. ISBN 978-0-323-07275-5. Archived from the original on 2016-05-11.
- ↑ Ciria R, Pleguezuelo M, Khorsandi SE, et al. (May 2013). "Strategies to reduce hepatitis C virus recurrence after liver transplantation". World J Hepatol. 5 (5): 237–50. doi:10.4254/wjh.v5.i5.237. PMC 3664282. PMID 23717735.
- ↑ Coilly A, Roche B, Samuel D (February 2013). "Current management and perspectives for HCV recurrence after liver transplantation". Liver Int. 33 Suppl 1: 56–62. doi:10.1111/liv.12062. PMID 23286847.
- ↑ Gurusamy, KS; Tsochatzis, E; Toon, CD; Xirouchakis, E; Burroughs, AK; Davidson, BR (4 December 2013). "Antiviral interventions for liver transplant patients with recurrent graft infection due to hepatitis C virus". The Cochrane Database of Systematic Reviews (12): CD006803. doi:10.1002/14651858.CD006803.pub4. PMID 24307460.
- ↑ 106.0 106.1 Hepatitis C and CAM: What the Science Says Archived 2011-03-20 at the Wayback Machine. National Center for Complementary and Alternative Medicine (NCCAM). March 2011. (Retrieved 7 March 2011)
- ↑ Liu, J; Manheimer E; Tsutani K; Gluud C (March 2003). "Medicinal herbs for hepatitis C virus infection: a Cochrane hepatobiliary systematic review of randomized trials". The American Journal of Gastroenterology. 98 (3): 538–44. PMID 12650784.
- ↑ Rambaldi, A; Jacobs, BP; Gluud, C (17 October 2007). "Milk thistle for alcoholic and/or hepatitis B or C virus liver diseases". The Cochrane Database of Systematic Reviews (4): CD003620. doi:10.1002/14651858.CD003620.pub3. PMID 17943794.
- ↑ Helms, Richard A.; Quan, David J., eds. (2006). Textbook of Therapeutics: Drug and Disease Management (8. ed.). Philadelphia, PA [u.a.]: Lippincott Williams & Wilkins. p. 1340. ISBN 978-0-7817-5734-8. Archived from the original on 5 December 2015. Retrieved 7 November 2014.
- ↑ Morgan RL, Baack B, Smith BD, Yartel A, Pitasi M, Falck-Ytter Y (March 2013). "Eradication of Hepatitis C Virus Infection and the Development of Hepatocellular Carcinoma: A Meta-analysis of Observational Studies". Annals of Internal Medicine. 158 (5 Pt 1): 329–37. doi:10.7326/0003-4819-158-5-201303050-00005. PMID 23460056.
- ↑ Fung J; Lai CL; Hung I; et al. (September 2008). "Chronic hepatitis C virus genotype 6 infection: response to pegylated interferon and ribavirin". The Journal of Infectious Diseases. 198 (6): 808–12. doi:10.1086/591252. PMID 18657036.
- ↑ GBD 2015 Disease and Injury Incidence and Prevalence, Collaborators. (8 October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- ↑ Lozano, R (2012-12-15). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". Lancet. 380 (9859): 2095–128. doi:10.1016/S0140-6736(12)61728-0. hdl:10536/DRO/DU:30050819. PMID 23245604. Archived from the original on 2020-05-19. Retrieved 2020-07-28.
- ↑ Mohd Hanafiah, K; Groeger, J; Flaxman, AD; Wiersma, ST (April 2013). "Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence". Hepatology. 57 (4): 1333–42. doi:10.1002/hep.26141. PMID 23172780.
- ↑ Yu ML; Chuang WL (March 2009). "Treatment of chronic hepatitis C in Asia: when East meets West". J. Gastroenterol. Hepatol. 24 (3): 336–45. doi:10.1111/j.1440-1746.2009.05789.x. PMID 19335784.
- ↑ "U.S. 2014 Surveillance Data for Viral Hepatitis, Statistics & Surveillance, Division of Viral Hepatitis". CDC. Archived from the original on 2016-08-08. Retrieved 2016-08-04.
- ↑ Table 4.5. "Number and rate of deaths with hepatitis C listed as a cause of death, by demographic characteristic and year – United States, 2004–2008". Viral Hepatitis on the CDC web site. Centers for Disease Control and Prevention, Atlanta, GA. Archived from the original on 9 March 2014. Retrieved 28 July 2013.
- ↑ "Hepatitis Death Rate Creeps past AIDS". New York Times. 27 February 2012. Archived from the original on 30 June 2017. Retrieved 28 July 2013.
- ↑ "Hepatitis C Kills More Americans than Any Other Infectious Disease". Centers for Disease Control and Prevention. May 4, 2016. Archived from the original on 9 August 2016. Retrieved 3 August 2016.
- ↑ Blatt, L. M.; Tong, M. (2004). Colacino, J. M.; Heinz, B. A. (eds.). Hepatitis prevention and treatment. Basel: Birkhäuser. p. 32. ISBN 978-3-7643-5956-0. Archived from the original on 2016-06-24.
- ↑ Blachier M, Leleu H, Peck-Radosavljevic M, Valla DC, Roudot-Thoraval F (March 2013). "The burden of liver disease in Europe: a review of available epidemiological data". J. Hepatol. 58 (3): 593–608. doi:10.1016/j.jhep.2012.12.005. PMID 23419824.
- ↑ 122.0 122.1 122.2 "Commissioning supplement: Health inequalities tell a tale of data neglect". Health Service Journal. 19 March 2015. Archived from the original on 28 July 2015. Retrieved 30 April 2015.
- ↑ "More than half of patients using needle exchange pilot tested positive for Hepatitis C". Pharmaceutical Journal. 17 May 2018. Archived from the original on 16 August 2018. Retrieved 15 August 2018.
- ↑ "Legal action firm among winners for largest medicines procurement". Health Service Journal. 30 April 2019. Archived from the original on 30 April 2019. Retrieved 9 June 2019.
- ↑ Holmberg, Scott (2011-05-12). Brunette, Gary W.; Kozarsky, Phyllis E.; Magill, Alan J.; Shlim, David R.; Whatley, Amanda D. (eds.). CDC Health Information for International Travel 2012. New York: Oxford University Press. p. 231. ISBN 978-0-19-976901-8.
{{cite book}}: CS1 maint: url-status (link) - ↑ "Hepatitis C". World Health Organization (WHO). June 2011. Archived from the original on 2011-07-12. Retrieved 2011-07-13.
- ↑ 127.0 127.1 127.2 127.3 127.4 127.5 Lombardi, Andrea; Mondelli, Mario U.; ESCMID Study Group for Viral Hepatitis (ESGVH) (March 2019). "Hepatitis C: Is eradication possible?". Liver International. 39 (3): 416–26. doi:10.1111/liv.14011. ISSN 1478-3231. PMID 30472772.
- ↑ 128.0 128.1 Boyer, JL (2001). Liver cirrhosis and its development: proceedings of the Falk Symposium 115. Springer. pp. 344. ISBN 978-0-7923-8760-2.
- ↑ Choo QL, Kuo G, Weiner AJ, Overby LR, Bradley DW, Houghton M (April 1989). "Isolation of a cDNA clone derived from a blood-borne non-A, non-B viral hepatitis genome" (PDF). Science. 244 (4902): 359–62. Bibcode:1989Sci...244..359C. CiteSeerX 10.1.1.469.3592. doi:10.1126/science.2523562. PMID 2523562. Archived (PDF) from the original on 2021-02-26. Retrieved 2020-07-28.
- ↑ Kuo G, Choo QL, Alter HJ, et al. (April 1989). "An assay for circulating antibodies to a major etiologic virus of human non-A, non-B hepatitis". Science. 244 (4902): 362–64. Bibcode:1989Sci...244..362K. doi:10.1126/science.2496467. PMID 2496467.
- ↑ "2000 Winners Albert Lasker Award for Clinical Medical Research". Archived from the original on February 25, 2008. Retrieved 2006-04-21.
{{cite web}}: CS1 maint: bot: original URL status unknown (link). Retrieved 20 February 2008. - ↑ "The Nobel Prize in Physiology or Medicine 2020". NobelPrize.org. Archived from the original on 5 October 2020. Retrieved 14 October 2020.
- ↑ EP patent 0318216, Houghton, M; Choo, Q-L & Kuo, G, "NANBV diagnostics", issued 1989-05-31, assigned to Chiron
- ↑ Wilken. "United States Court of Appeals for the Federal Circuit". United States Court of Appeals for the Federal Circuit. Archived from the original on 19 November 2009. Retrieved 11 January 2012.
- ↑ Eurosurveillance editorial team (2011-07-28). "World Hepatitis Day 2011" (PDF). Eurosurveillance. 16 (30). PMID 21813077. Archived (PDF) from the original on 2011-11-25.
- ↑ 136.0 136.1 Wong, JB (2006). "Hepatitis C: cost of illness and considerations for the economic evaluation of antiviral therapies". PharmacoEconomics. 24 (7): 661–72. doi:10.2165/00019053-200624070-00005. PMID 16802842.
- ↑ 137.0 137.1 El Khoury, AC; Klimack, WK; Wallace, C; Razavi, H (1 December 2011). "Economic burden of hepatitis C-associated diseases in the United States". Journal of Viral Hepatitis. 19 (3): 153–60. doi:10.1111/j.1365-2893.2011.01563.x. PMID 22329369.
- ↑ "Hepatitis C Prevention, Support and Research ProgramHealth Canada". Public Health Agency of Canada. Nov 2003. Archived from the original on 22 March 2011. Retrieved 10 January 2012.
- ↑ Thomas, Howard; Lemon, Stanley; Zuckerman, Arie, eds. (2008). Viral Hepatitis (3rd ed.). Oxford: John Wiley & Sons. p. 532. ISBN 978-1-4051-4388-2. Archived from the original on 2016-06-17.
- ↑ "Spanish Anesthetist Infected Patients". The Washington Post. 15 May 2007. Archived from the original on 22 August 2016. Retrieved 13 July 2016.
- ↑ "Spanish Hep C anaesthetist jailed". BBC. 15 May 2007. Archived from the original on 23 October 2007. Retrieved 13 July 2016.
- ↑ Arshad M, El-Kamary SS, Jhaveri R (April 2011). "Hepatitis C virus infection during pregnancy and the newborn period – are they opportunities for treatment?". J. Viral Hepat. 18 (4): 229–36. doi:10.1111/j.1365-2893.2010.01413.x. PMID 21392169.
- ↑ Hunt CM, Carson KL, Sharara AI (May 1997). "Hepatitis C in pregnancy". Obstet Gynecol. 89 (5 Pt 2): 883–90. doi:10.1016/S0029-7844(97)81434-2. PMID 9166361.
- ↑ 144.0 144.1 Thomas SL, Newell ML, Peckham CS, Ades AE, Hall AJ (February 1998). "A review of hepatitis C virus (HCV) vertical transmission: risks of transmission to infants born to mothers with and without HCV viraemia or human immunodeficiency virus infection". Int J Epidemiol. 27 (1): 108–17. doi:10.1093/ije/27.1.108. PMID 9563703.
- ↑ Fischler B (June 2007). "Hepatitis C virus infection". Semin Fetal Neonatal Med. 12 (3): 168–73. doi:10.1016/j.siny.2007.01.008. PMID 17320495.
- ↑ Indolfi G, Resti M (May 2009). "Perinatal transmission of hepatitis C virus infection". J. Med. Virol. 81 (5): 836–43. doi:10.1002/jmv.21437. PMID 19319981.
- ↑ González-Peralta RP (November 1997). "Hepatitis C virus infection in pediatric patients". Clin Liver Dis. 1 (3): 691–705, ix. doi:10.1016/s1089-3261(05)70329-9. PMID 15560066.
- ↑ Suskind DL, Rosenthal P (February 2004). "Chronic viral hepatitis". Adolesc Med Clin. 15 (1): 145–58, x–xi. doi:10.1016/j.admecli.2003.11.001. PMID 15272262.
- ↑ Iorio, A; Marchesini, E; Awad, T; Gluud, LL (20 January 2010). "Antiviral treatment for chronic hepatitis C in patients with human immunodeficiency virus". The Cochrane Database of Systematic Reviews (1): CD004888. doi:10.1002/14651858.CD004888.pub2. PMID 20091566.
- ↑ Ahn, J; Flamm, SL (August 2011). "Hepatitis C therapy: other players in the game". Clinics in Liver Disease. 15 (3): 641–56. doi:10.1016/j.cld.2011.05.008. PMID 21867942.
- ↑ Vermehren, J; Sarrazin, C (February 2011). "New HCV therapies on the horizon". Clinical Microbiology and Infection. 17 (2): 122–34. doi:10.1111/j.1469-0691.2010.03430.x. PMID 21087349.
- ↑ Feld, Jordan J.; Jacobson, Ira M.; Hézode, Christophe; Asselah, Tarik; Ruane, Peter J.; Gruener, Norbert; Abergel, Armand; Mangia, Alessandra; Lai, Ching-Lung; Chan, Henry L.Y.; Mazzotta, Francesco; Moreno, Christophe; Yoshida, Eric; Shafran, Stephen D.; Towner, William J.; Tran, Tram T.; McNally, John; Osinusi, Anu; Svarovskaia, Evguenia; Zhu, Yanni; Brainard, Diana M.; McHutchison, John G.; Agarwal, Kosh; Zeuzem, Stefan (16 November 2015). "Sofosbuvir and Velpatasvir for HCV Genotype 1, 2, 4, 5, and 6 Infection". New England Journal of Medicine. 373 (27): 2599–2607. doi:10.1056/NEJMoa1512610. hdl:10722/226358. PMID 26571066. Archived from the original on 16 February 2020. Retrieved 28 July 2020.
- ↑ Gurusamy, KS; Tsochatzis, E; Toon, CD; Davidson, BR; Burroughs, AK (2 December 2013). "Antiviral prophylaxis for the prevention of chronic hepatitis C virus in patients undergoing liver transplantation". The Cochrane Database of Systematic Reviews (12): CD006573. doi:10.1002/14651858.CD006573.pub3. PMC 6599865. PMID 24297303.
- ↑ 155.0 155.1 Sandmann, L; Ploss, A (2013-01-05). "Barriers of hepatitis C virus interspecies transmission". Virology. 435 (1): 70–80. doi:10.1016/j.virol.2012.09.044. PMC 3523278. PMID 23217617.
External links
| Classification | |
|---|---|
| External resources |
- Hepatitis C at Curlie
- "Recommendations for Testing, Managing, and Treating Hepatitis C". www.hcvguidelines.org. IDSA/AASLD. Archived from the original on 25 July 2017. Retrieved 28 July 2017.
- "Hepatitis C". MedlinePlus. U.S. National Library of Medicine. Archived from the original on 2020-07-29. Retrieved 2020-07-28.
- Pages with script errors
- CS1: long volume value
- Webarchive template wayback links
- CS1 maint: url-status
- CS1 maint: bot: original URL status unknown
- Good articles
- Articles containing potentially dated statements from 2017
- Articles with invalid date parameter in template
- All articles containing potentially dated statements
- Articles containing potentially dated statements from 2012
- Articles with hatnote templates targeting a nonexistent page
- Articles containing potentially dated statements from 2016
- Articles containing potentially dated statements from 2011
- Articles with hAudio microformats
- Spoken articles
- Articles with Curlie links
- Portal templates with all redlinked portals
- Hepatitis C
- Healthcare-associated infections
- Infectious causes of cancer
- RTT(full)
- RTTEM
- Viral diseases
- RTT
- RTTOSH