Adenoviridae
| Adenoviruses | |
|---|---|

| |
| Transmission electron micrograph of two adenovirus particles | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Varidnaviria |
| Kingdom: | Bamfordvirae |
| Phylum: | Preplasmiviricota |
| Class: | Tectiliviricetes |
| Order: | Rowavirales |
| Family: | Adenoviridae |
| Genera | |
| |
Adenoviruses (members of the family Adenoviridae) are medium-sized (90–100 nm), nonenveloped (without an outer lipid bilayer) viruses with an icosahedral nucleocapsid containing a double-stranded DNA genome.[1] Their name derives from their initial isolation from human adenoids in 1953.[2]
They have a broad range of vertebrate hosts; in humans, more than 50 distinct adenoviral serotypes have been found to cause a wide range of illnesses, from mild respiratory infections in young children (known as the common cold) to life-threatening multi-organ disease in people with a weakened immune system.[1]
Virology

Classification
This family contains the following genera:[4]
- Atadenovirus
- Aviadenovirus
- Ichtadenovirus
- Mastadenovirus (including all human adenoviruses)
- Siadenovirus
- Testadenovirus
Diversity
In humans, currently there are 88 human adenoviruses (HAdVs) in seven species (Human adenovirus A to G):[5]
- A: 12, 18, 31[6][7][8]
- B: 3, 7, 11, 14, 16, 21, 34, 35, 50, 55[9][10][11][12][13][14][15][16][17]
- C: 1, 2, 5, 6, 57[18]
- D: 8, 9, 10, 13, 15, 17, 19, 20, 22, 23, 24, 25, 26, 27, 28, 29, 30, 32, 33, 36, 37, 38, 39, 42, 43, 44, 45, 46, 47, 48, 49, 51, 53, 54, 56,58, 59, 60, 62, 63, 64, 65, 67, 69, 70, 71, 72, 73, 74, 75[19][20][21][22][23][24][1]
- E: 4[25]
- F: 40, 41[26]
- G: 52[27]
Different types/serotypes are associated with different conditions:
- Respiratory disease (mainly species HAdV-B and C)[28]
- Conjunctivitis (HAdV-B and D)[29]
- Gastroenteritis (HAdV-F types 40, 41, HAdV-G type 52)[30]
- Obesity or adipogenesis (HAdV-A type 31, HAdV-C type 5, HAdV-D types 9, 36, 37)[31][32]
All these types are called Human mastadenovirus A–G by the ICTV, because all are members of the genus Mastadenovirus.[33]

Structure
Adenoviruses are medium-sized (90–100 nm).[1] The virions are composed of one linear piece of double-stranded DNA inside an icosahedral capsid. 240 hexon proteins make up the bulk of the capsid, while twelve penton bases cap the icosahedron's corners. The penton bases are associated with protruding fibers that aid in attachment to the host cell via the receptor on its surface.[34]
In 2010, the structure of the human adenovirus was solved at the atomic level, making it the largest high-resolution model ever. The virus is composed of around 1 million amino acid residues and weighs around 150 MDa.[35][36]
Genome
The adenovirus genome is linear, non-segmented double-stranded (ds) DNA that is between 26 and 48 Kbp.[1] This allows the virus to theoretically carry 22 to 40 genes. Although this is significantly larger than other viruses in its Baltimore group, it is still a very simple virus and is heavily reliant on the host cell for survival and replication. An interesting feature of this viral genome is that it has a terminal 55 kDa protein associated with each of the 5' ends of the linear dsDNA. These are used as primers in viral replication and ensure that the ends of the virus' linear genome are adequately replicated.[37][38][39][40]

Replication

Adenoviruses possess a linear dsDNA genome and are able to replicate in the nucleus of vertebrate cells using the host's replication machinery.[1] Entry of adenoviruses into the host cell involves two sets of interactions between the virus and the host cell.[1] Most of the action occurs at the vertices. Entry into the host cell is initiated by the knob domain of the fiber protein binding to the cell receptor.[1] The two currently established receptors are: CD46 for the group B human adenovirus serotypes and the coxsackievirus/adenovirus receptor (CAR) for all other serotypes.[1] There are some reports suggesting MHC molecules and sialic acid residues functioning in this capacity as well. This is followed by a secondary interaction, where a motif in the penton base protein interacts with an integrin molecule. It is the co-receptor interaction that stimulates entry of the adenovirus. This co-receptor molecule is αV integrin. Binding to αv integrin results in endocytosis of the virus particle via clathrin-coated pits. Attachment to αV integrin stimulates cell signaling and thus induces actin polymerization, which facilitates clathrin-mediated endocytosis, and results in virion's entry into the host cell within an endosome.[41]
Once the virus has successfully gained entry into the host cell, the endosome acidifies, which alters virus topology by causing capsid components to disband. The capsid is destabilized and protein VI, which is one of the capsid constituents is released from it.[42] These changes, as well as the toxic nature of the pentons, destroy the endosome, resulting in the movement of the virion into the cytoplasm.[1] With the help of cellular microtubules, the virus is transported to the nuclear pore complex, whereby the adenovirus particle disassembles. Viral DNA is subsequently released, which can enter the nucleus via the nuclear pore.[43] After this the DNA associates with histone molecules already present in the nucleus, which allows it to interact with the host cell transcription machinery. Then, viral gene expression can occur, without integrating the viral genome into host cell chromosomes,[44] and new virus particles can be generated.[44]The adenovirus life cycle is separated by the DNA replication process into two phases: an early and a late phase.[1] In both phases, a primary transcript that is alternatively spliced to generate monocistronic mRNAs compatible with the host's ribosome is generated, allowing for the products to be translated.[45]
The early genes are responsible for expressing mainly non-structural, regulatory proteins.[1] The goal of these proteins is threefold: to alter the expression of host proteins that are necessary for DNA synthesis; to activate other virus genes; and to avoid premature death of the infected cell by the host-immune defenses (blockage of MHC class I translocation and expression).[45][46]
Some adenoviruses under specialized conditions can transform cells using their early gene products. E1A has been found to immortalize primary cells in vitro allowing E1B to assist and stably transform the cells. Nevertheless, they are reliant upon each other to successfully transform the host cell and form tumors. E1A is mostly intrinsically disordered protein and contains CR3 domain which is critical for transcriptional activation.DNA replication separates the early and late phases. Once the early genes have liberated adequate virus proteins, replication machinery, and replication substrates, replication of the adenovirus genome can occur. A terminal protein that is covalently bound to the 5' end of the adenovirus genome acts as a primer for replication, the viral DNA polymerase then uses a strand displacement mechanism ( as opposed to the conventional Okazaki fragments used in mammalian DNA replication[47]).[48][49][50]
The late phase of the adenovirus lifecycle is focused on producing sufficient quantities of structural protein to pack all the genetic material produced by DNA replication.[1] Once the viral components have successfully been replicated, the virus is assembled into its protein shells and released from the cell as a result of virally induced cell lysis.[1]
Multiplicity reactivation
Adenovirus is capable of multiplicity reactivation (MR)[51] (Yamamoto and Shimojo, 1971). MR is the process by which two, or more, virus genomes containing lethal damage interact within the infected cell to form a viable virus genome. Such MR was demonstrated for adenovirus 12 after virions were irradiated with UV light and allowed to undergo multiple infection of host cells.[51] In a review, numerous examples of MR in different viruses were described, and it was suggested that MR is a common form of sexual interaction that provides the survival advantage of recombinational repair of genome damages.[52]
Epidemiology
Transmission
Adenoviruses are unusually stable to chemical or physical agents and adverse pH conditions, allowing for prolonged survival outside of the body and water. Adenoviruses are spread primarily via respiratory droplets, however they can also be spread by fecal routes and via aerosols (airborne transmission).[53] Research into the molecular mechanisms underlying adenoviral transmission provide empirical evidence in support of the hypothesis that coxsackievirus/adenovirus receptors (CARs) are needed to transport adenoviruses into certain naive/progenitor cell types.[54]
Humans
Humans infected with adenoviruses display a wide range of responses, from no symptoms at all to the severe infections typical of Adenovirus serotype 14.[55]
Animals

Bat adenovirus TJM (Bt-AdV-TJM) is a novel species of the Mastadenovirus genus isolated from Myotis and Scotophilus kuhlii in China.[56] It is most closely related to the tree shrew and canine AdVs.[57]Two types of canine adenoviruses are well known, type 1 and 2. Type 1 (CAdV-1) causes infectious canine hepatitis, a potentially fatal disease involving vasculitis and hepatitis. Type 1 infection can also cause respiratory and eye infections. CAdV-1 also affects foxes (Vulpes vulpes and Vulpes lagopus) and may cause hepatitis and encephalitis. Canine adenovirus 2 (CAdV-2) is one of the potential causes of kennel cough. Core vaccines for dogs include attenuated live CAdV-2, which produces immunity to CAdV-1 and CAdV-2. CAdV-1 was initially used in a vaccine for dogs, but corneal edema was a common complication.[58]Squirrel adenovirus (SqAdV) is reported to cause enteritis in red squirrels in Europe, while gray squirrels seem to be resistant. [59]
Adenoviruses are also known to cause respiratory infections in horses, cattle, pigs, sheep, and goats. Equine adenovirus 1 can also cause fatal disease in immunocompromised Arabian foals, involving pneumonia and destruction of pancreatic and salivary gland tissue.[58]
Otarine adenovirus 1 has been isolated from sea lions (Zalophus californianus).[60]The fowl adenoviruses are associated with many disease conditions in domestic fowl like inclusion body hepatitis, hydropericardium syndrome,[61] Egg drop syndrome, Quail bronchitis, Gizzard erosions and many respiratory conditions. They have also been isolated from wild black kites (Milvus migrans).[62]Titi monkey adenovirus was isolated from a colony of monkeys.[63]
Infections
Signs and symptoms
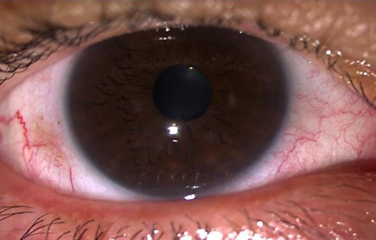
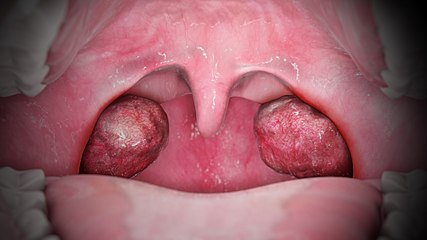
Most infections with adenovirus result in infections of the upper respiratory tract. Adenovirus infections often present as conjunctivitis, tonsillitis (which may look exactly like strep throat and cannot be distinguished from strep except by throat culture), an ear infection, or croup.[64] Adenoviruses types 40 and 41 can also cause gastroenteritis.[65] Both conjunctivitis and tonsillitis are particularly common with adenovirus infections.[66][67]
-
Adenoviral keratoconjunctivitis
-
Tonsilitis
-
Adenoviral keratoconjunctivitis
Some children can develop adenovirus bronchiolitis or pneumonia, both of which can be severe. In babies, adenoviruses can also cause coughing fits that look almost exactly like whooping cough. Adenoviruses can also cause viral meningitis or encephalitis. Rarely, adenovirus can cause hemorrhagic cystitis (inflammation of the urinary bladder—a form of urinary tract infection—with blood in the urine).[68][66][69][70]
Most people recover from adenovirus infections by themselves, but people with immunodeficiency sometimes die of adenovirus infections, and—rarely—even previously healthy people can die of these infections.[71] This may be because sometimes adenoviral infection can lead to cardiac disorders. For example, in one study, some cardiac samples of patients with dilated cardiomyopathy were positive for presence of adenovirus type 8.[72]
Transmission
Adenoviruses are often transmitted by expectoration (e.g. aerosols), but they can also be transmitted by contact with an infected person, or by virus particles left on objects. Some people with adenovirus gastroenteritis may shed the virus in their stools for months after getting over the symptoms. The virus can be passed through water in swimming pools.As with many other illnesses, good handwashing practice is one way to inhibit the person-to-person transmission of adenoviruses.Heat and bleach can neutralize the virus.[73][74][75][76]
Diagnosis
Diagnosis is from symptoms and history. Tests are only necessary in very serious cases. Tests include blood tests, eyes, nose or throat swabs, stool sample tests, and chest x-rays.[77] In the laboratory, adenovirus can be identified with antigen detection, polymerase chain reaction (PCR), virus isolation and serology. Even if adenovirus is found to be present, it may not be the cause of any symptoms. Some immunocompromised individuals can shed the virus for weeks and show no symptoms.[78]
Prevention
Currently there is a vaccine for adenovirus type 4 and 7 for US military personnel only. US military personnel are the recipients of this vaccine because they may be at a higher risk of infection.The vaccine contains a live virus, which may be shed in stool and lead to transmission. The vaccine is not approved for use outside of the military, as it has not been tested in studied in the general population or on people with weakened immune systems.[79][80]
In the past, US military recruits were vaccinated against two serotypes of adenovirus, with a corresponding decrease in illnesses caused by those serotypes. That vaccine is no longer manufactured. The U.S. Army Medical Research and Materiel Command announced on 31 October 2011 that a new adenovirus vaccine, which replaces the older version that has been out of production for over a decade, was shipped to basic training sites on 18 October 2011. More information is available here.[81]
Prevention of adenovirus, as well as other respiratory illnesses, involves frequent hand washing for more than 20 seconds, avoiding touching the eyes, face, and nose with unwashed hands, and avoiding close contact with people with symptomatic adenovirus infection. Those with symptomatic adenovirus infection are additionally advised to cough or sneeze into the arm or elbow instead of the hand, to avoid sharing cups and eating utensils, and to refrain from kissing others. Chlorination of swimming pools can prevent outbreaks of conjunctivitis caused by adenovirus.[79]
Treatment
There are no proven antiviral drugs to treat adenoviral infections, so treatment is largely directed at the symptoms, such as acetaminophen for fever. The antiviral drug cidofovir has helped certain of those patients who had severe cases of illness; the number helped and to what degree, and the particular complications or symptoms it helped with, and when and where this happened, were not given in the source. A healthcare worker may give antibiotic eyedrops for conjunctivitis, while awaiting results of bacterial cultures, and to help prevent secondary bacterial infections. Currently, there is no adenovirus vaccine available to the general public, but a vaccine is available for the United States military for Types 4 and 7.[82][80][83]
Use in gene therapy and vaccination
Gene therapy

Adenoviruses have long been a popular viral vector for gene therapy due to their ability to affect both replicating and non-replicating cells, accommodate large transgenes, and code for proteins without integrating genetic material into the host cell genome.[44] More specifically, they are used as a vehicle to administer targeted therapy,[84] in the form of recombinant DNA or protein. This therapy has been found especially useful in treating monogenic disease (e.g. cystic fibrosis, X-linked SCID, alpha1-antitrypsin deficiency) and cancer.[44] In China, oncolytic adenovirus is an approved cancer treatment.[85] Specific modifications on fiber proteins are used to target Adenovirus to certain cell types;[86] a major effort is made to limit hepatotoxicity and prevent multiple organ failure. Adenovirus dodecahedron can qualify as a potent delivery platform for foreign antigens to human myeloid dendritic cells (MDC), and that it is efficiently presented by MDC to M1-specific CD8+ T lymphocytes.[87]
A safety issue with adenoviruses is that they can cause an immue response with a related inflammatory respose as occurred in the death Jesse Gelsinger in 1999. To address this risk, the genome of the viral genes have been modified to remove some viral genes such. One such modification is the gutless vector that removes almost all the viral genome.[88]: 58
Adenovirus has been used for delivery of CRISPR/Cas9 gene editing systems, but high immune reactivity to viral infection has posed challenges in use for patients.[89]
Vaccines

Modified (recombinant) adenovirus vectors, including replication incompetent types, can deliver DNA coding for specific antigens.[90]
Adenovirus have been used to produce viral vector COVID-19 vaccines. "In four candidate COVID-19 vaccines... Ad5... serves as the 'vector' to transport the surface protein gene of SARS-CoV-2".[91] The goal is to genetically express the spike glycoprotein of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). A replication-deficient chimpanzee adenovirus vaccine vector (ChAdOx1) is used by the Oxford–AstraZeneca COVID-19 vaccine that has been approved for use.[92][93] The Janssen COVID-19 vaccine uses modified recombinant adenovirus type-26 (Ad26).[94] Recombinant adenovirus type-5 (Ad5) are being used by Ad5-nCoV,[95] ImmunityBio and UQ-CSL V451. The Gam-COVID-Vac (aka Sputnik-V) product is innovative because an Ad26 based vaccine is used on the first day and an Ad5 vaccine is used on day 21.[94] Another one is ChAd-SARS-CoV-2-S; the vaccine reportedly prevented mice that were genetically modified to have human ACE2 (hACE2) receptors, presumably receptors that allow virus-entry into the cells, from being infected with SARS-CoV-2.[96][97]
Possible issues with using Adenovirus as vaccine vectors include: the human body develops immunity to the vector itself, making subsequent booster shots difficult or impossible.[98] In some cases, people have pre-existing immunity to Adenoviruses, making vector delivery ineffective.[99]
HIV infection concerns
The use of Ad5 vaccines for COVID-19 worried researchers who had experience with two failed trials of an Ad5 vaccine, Phambili and STEP, due to the increased risk for uncircumcised male patients of contracting HIV-1 via unprotected anal sex.[100] At the time, it was concluded that heightened risk of HIV reception may be observed for any Ad5-based vector vaccine.[101] In October 2020, these researchers wrote in The Lancet: "On the basis of these findings, we are concerned that use of an Ad5 vector for immunisation against SARS-CoV-2 could similarly increase the risk of HIV-1 acquisition among men who receive the vaccine."[102][103] Vaccines using other technologies would not be affected, but Sputnik V, Convidecia and ImmunityBio's hAd5 would.[104] Two studies found that Ad5-specific CD4 T cells are more susceptible to HIV infection than CD4 T cells specific to certain other vectors, such as Cytomegalovirus[105] and Canarypox.[106]
By comparison, a Science article reported that China had approved CanSino's Ebola vaccine based on an Ad5 vector. It was tested in Sierra Leone, which had high HIV prevalence, making it more likely for such problems to be detected. CanSino's CEO said "we haven't seen anything with the Ebola vaccine" and speculated that HIV susceptibility might be limited to Ad5 vaccines which produced HIV proteins. In research reported in The Lancet in May, the company's researchers acknowledged the possibility, called it "controversial" and said they would watch for it in the company's COVID-19 vaccine candidate's trials.[91][95]
See also
Notes
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 "9.11H: Double-Stranded DNA Viruses- Adenoviruses". Biology LibreTexts. 25 June 2017. Archived from the original on 8 January 2021. Retrieved 6 January 2021.
- ↑ Rowe WP, Huebner RJ, Gilmore LK, Parrott RH, Ward TG (December 1953). "Isolation of a cytopathogenic agent from human adenoids undergoing spontaneous degeneration in tissue culture". Proceedings of the Society for Experimental Biology and Medicine. 84 (3): 570–3. doi:10.3181/00379727-84-20714. PMID 13134217. S2CID 3097955.
- ↑ Padilla-Sanchez V (2021-07-24), Adenovirus D26 Structural Model at Atomic Resolution, doi:10.5281/zenodo.5132873, retrieved 2021-07-24
- ↑ "Virus Taxonomy: 2020 Release". International Committee on Taxonomy of Viruses (ICTV). March 2021. Archived from the original on 20 March 2020. Retrieved 22 May 2021.
- ↑ Dhingra A, Hage E, Ganzenmueller T, Böttcher S, Hofmann J, Hamprecht K, et al. (January 2019). "Molecular Evolution of Human Adenovirus (HAdV) Species C". Scientific Reports. 9 (1): 1039. Bibcode:2019NatSR...9.1039D. doi:10.1038/s41598-018-37249-4. PMC 6355881. PMID 30705303.
- ↑ "Human adenovirus 12". pubchem.ncbi.nlm.nih.gov. Archived from the original on 11 March 2023. Retrieved 11 March 2023.
- ↑ "Taxonomy browser (Human adenovirus 18)". www.ncbi.nlm.nih.gov. Archived from the original on 11 March 2023. Retrieved 11 March 2023.
- ↑ "Human adenovirus 31". pubchem.ncbi.nlm.nih.gov. Archived from the original on 11 March 2023. Retrieved 11 March 2023.
- ↑ Cao, Bin; Huang, Guo-Hong; Pu, Zeng-Hui; Qu, Jiu-Xin; Yu, Xiao-Min; Zhu, Zhen; Dong, Jian-Ping; Gao, Yan; Zhang, Yong-Xiang; Li, Xiao-Hui; Liu, Jian-Hua; Wang, Hong; Xu, Qian; Li, Hui; Xu, Wenbo; Wang, Chen (1 January 2014). "Emergence of Community-Acquired Adenovirus Type 55 as a Cause of Community-Onset Pneumonia". CHEST. 145 (1): 79–86. doi:10.1378/chest.13-1186. Archived from the original on 7 May 2022. Retrieved 12 March 2023.
- ↑ "Taxonomy browser (Human adenovirus 50)". www.ncbi.nlm.nih.gov. Archived from the original on 12 March 2023. Retrieved 12 March 2023.
- ↑ "Taxonomy browser (Human adenovirus 35)". www.ncbi.nlm.nih.gov. Archived from the original on 12 March 2023. Retrieved 12 March 2023.
- ↑ "Taxonomy browser (Human adenovirus 34)". www.ncbi.nlm.nih.gov. Archived from the original on 12 March 2023. Retrieved 12 March 2023.
- ↑ "Taxonomy browser (Human adenovirus 21)". www.ncbi.nlm.nih.gov. Archived from the original on 12 March 2023. Retrieved 12 March 2023.
- ↑ "Human adenovirus 16". pubchem.ncbi.nlm.nih.gov. Archived from the original on 12 March 2023. Retrieved 12 March 2023.
- ↑ "Taxonomy browser (Human adenovirus 11)". www.ncbi.nlm.nih.gov. Archived from the original on 12 March 2023. Retrieved 12 March 2023.
- ↑ "Acute Respiratory Disease Associated with Adenovirus Serotype 14 --- Four States, 2006--2007". www.cdc.gov. Archived from the original on 5 August 2022. Retrieved 12 March 2023.
- ↑ "Taxonomy browser (Human adenovirus 3+7)". www.ncbi.nlm.nih.gov. Archived from the original on 12 March 2023. Retrieved 12 March 2023.
- ↑ Walsh MP, Seto J, Liu EB, Dehghan S, Hudson NR, Lukashev AN, et al. (October 2011). "Computational analysis of two species C human adenoviruses provides evidence of a novel virus". Journal of Clinical Microbiology. 49 (10): 3482–90. doi:10.1128/JCM.00156-11. PMC 3187342. PMID 21849694.
- ↑ Adhikary, Arun Kumar; Banik, Urmila (December 2014). "Human adenovirus type 8: the major agent of epidemic keratoconjunctivitis (EKC)". Journal of Clinical Virology: The Official Publication of the Pan American Society for Clinical Virology. 61 (4): 477–486. doi:10.1016/j.jcv.2014.10.015. ISSN 1873-5967. Archived from the original on 7 May 2022. Retrieved 14 March 2023.
- ↑ Singh G, Zhou X, Lee JY, Yousuf MA, Ramke M, Ismail AM, et al. (November 2015). "Recombination of the epsilon determinant and corneal tropism: Human adenovirus species D types 15, 29, 56, and 69". Virology. 485: 452–9. doi:10.1016/j.virol.2015.08.018. PMC 4619159. PMID 26343864.
- ↑ Singh G, Robinson CM, Dehghan S, Schmidt T, Seto D, Jones MS, et al. (April 2012). "Overreliance on the hexon gene, leading to misclassification of human adenoviruses". Journal of Virology. 86 (8): 4693–5. doi:10.1128/jvi.06969-11. PMC 3318657. PMID 22301156.
- ↑ Robinson CM, Singh G, Henquell C, Walsh MP, Peigue-Lafeuille H, Seto D, et al. (January 2011). "Computational analysis and identification of an emergent human adenovirus pathogen implicated in a respiratory fatality". Virology. 409 (2): 141–7. doi:10.1016/j.virol.2010.10.020. PMC 3006489. PMID 21056888.
- ↑ "Family: Adenoviridae | ICTV". ictv.global. Archived from the original on 6 February 2023. Retrieved 13 March 2023.
- ↑ Berman, Jules J. (1 January 2012). "Chapter 39 - Group I Viruses: Double-Stranded DNA". Taxonomic Guide to Infectious Diseases. Academic Press. pp. 221–229. ISBN 978-0-12-415895-5. Archived from the original on 1 September 2022. Retrieved 14 March 2023.Human adenovirus D, types 8, 9, 10, 13, 15, 17, 19, 20, 22, 23, 24, 25, 26, 27, 28, 29, 30, 32, 33, 36, 37, 38, 39, 42, 43, 44, 45, 46, 47, 48, 49, 51, 53, 54, 56
- ↑ "Human adenovirus E4". pubchem.ncbi.nlm.nih.gov. Archived from the original on 11 March 2023. Retrieved 11 March 2023.
- ↑ Lee B, Damon CF, Platts-Mills JA (October 2020). "Pediatric acute gastroenteritis associated with adenovirus 40/41 in low-income and middle-income countries". Current Opinion in Infectious Diseases. 33 (5): 398–403. doi:10.1097/QCO.0000000000000663. PMC 8286627. PMID 32773498.
- ↑ Jones MS, Harrach B, Ganac RD, Gozum MM, Dela Cruz WP, Riedel B, et al. (June 2007). "New adenovirus species found in a patient presenting with gastroenteritis". Journal of Virology. 81 (11): 5978–84. doi:10.1128/JVI.02650-06. PMC 1900323. PMID 17360747.
- ↑ Xu, Deshun; Chen, Liping; Wu, Xiaofang; Ji, Lei (21 April 2022). "Molecular typing and epidemiology profiles of human adenovirus infection among hospitalized patients with severe acute respiratory infection in Huzhou, China". PLOS ONE. 17 (4): e0265987. doi:10.1371/journal.pone.0265987. ISSN 1932-6203. Archived from the original on 29 December 2022. Retrieved 20 March 2023.
- ↑ Labib, Bisant A.; Minhas, Bhawanjot K.; Chigbu, DeGaulle I. (17 March 2020). "Management of Adenoviral Keratoconjunctivitis: Challenges and Solutions". Clinical Ophthalmology. 14: 837–852. doi:10.2147/OPTH.S207976. Archived from the original on 17 February 2023. Retrieved 20 March 2023.
- ↑ do Nascimento, Lilian Gonçalves; Fialho, Alexandre Madi; de Andrade, Juliana da Silva Ribeiro; de Assis, Rosane Maria Santos; Fumian, Tulio Machado (2 July 2022). "Human enteric adenovirus F40/41 as a major cause of acute gastroenteritis in children in Brazil, 2018 to 2020". Scientific Reports. 12 (1): 11220. doi:10.1038/s41598-022-15413-1. ISSN 2045-2322. Archived from the original on 3 July 2022. Retrieved 20 March 2023.
- ↑ "Adenovirus | Home | CDC". www.cdc.gov. Centers for Disease control and prevention. 31 January 2020. Archived from the original on 16 April 2022. Retrieved 6 January 2021.
- ↑ Voss JD, Atkinson RL, Dhurandhar NV (November 2015). "Role of adenoviruses in obesity". Reviews in Medical Virology. 25 (6): 379–87. doi:10.1002/rmv.1852. PMID 26352001. S2CID 5370331. Archived from the original on 2022-02-19. Retrieved 2023-02-14.
- ↑ "Complete genomes: Mastadenovirus". www.ncbi.nlm.nih.gov. Archived from the original on 10 March 2023. Retrieved 10 March 2023.
- ↑ Harrach B, Benkő M (2021). "Adenoviruses (Adenoviridae)". Encyclopedia of Virology. Vol. 2. Elsevier. ISBN 9780128145166. Archived from the original on 4 March 2023. Retrieved 12 March 2021.
- ↑ Reddy VS, Natchiar SK, Stewart PL, Nemerow GR (August 2010). "Crystal structure of human adenovirus at 3.5 A resolution". Science. 329 (5995): 1071–5. Bibcode:2010Sci...329.1071R. doi:10.1126/science.1187292. PMC 2929978. PMID 20798318.
- Lay summary in: "Scientists unveil structure of adenovirus, the largest high-resolution complex ever found". Science Daily. August 28, 2010.
- ↑ Harrison SC (August 2010). "Virology. Looking inside adenovirus". Science. 329 (5995): 1026–7. Bibcode:2010Sci...329.1026H. doi:10.1126/science.1194922. PMID 20798308. S2CID 206528739.
- ↑ "Complete genomes: Virus". www.ncbi.nlm.nih.gov. Archived from the original on 15 June 2022. Retrieved 6 March 2023.
- ↑ Koonin, Eugene V.; Krupovic, Mart; Agol, Vadim I. (18 August 2021). "The Baltimore Classification of Viruses 50 Years Later: How Does It Stand in the Light of Virus Evolution?". Microbiology and molecular biology reviews: MMBR. 85 (3): e0005321. doi:10.1128/MMBR.00053-21. ISSN 1098-5557. Archived from the original on 12 February 2023. Retrieved 6 March 2023.
- ↑ Kulanayake, Shermila; Tikoo, Suresh K. (28 February 2021). "Adenovirus Core Proteins: Structure and Function". Viruses. 13 (3): 388. doi:10.3390/v13030388. ISSN 1999-4915. Archived from the original on 9 December 2022. Retrieved 22 March 2023.
- ↑ Doerfler, Walter (1996). "Adenoviruses". Medical Microbiology (4th ed.). University of Texas Medical Branch at Galveston. ISBN 978-0-9631172-1-2. Archived from the original on 2022-01-22. Retrieved 22 March 2023.
- ↑ Wu E, Nemerow GR (April 2004). "Virus yoga: the role of flexibility in virus host cell recognition". Trends in Microbiology. 12 (4): 162–9. doi:10.1016/j.tim.2004.02.005. PMID 15051066.
- ↑ Flint J, Skalka AM, Rall GF, Racaniello VR (2015). Principles of Virology. Molecular Biology. Vol. I. doi:10.1128/9781555818951. ISBN 9781555819330.
- ↑ Meier O, Greber UF (February 2004). "Adenovirus endocytosis". The Journal of Gene Medicine. 6 (Suppl 1): S152-63. doi:10.1002/jgm.553. PMID 14978758. S2CID 22241820.
- ↑ 44.0 44.1 44.2 44.3 Lee CS, Bishop ES, Zhang R, Yu X, Farina EM, Yan S, et al. (June 2017). "Adenovirus-Mediated Gene Delivery: Potential Applications for Gene and Cell-Based Therapies in the New Era of Personalized Medicine". Genes & Diseases. 4 (2): 43–63. doi:10.1016/j.gendis.2017.04.001. PMC 5609467. PMID 28944281.
- ↑ 45.0 45.1 Hoeben, Rob C.; Uil, Taco G. (1 March 2013). "Adenovirus DNA replication". Cold Spring Harbor Perspectives in Biology. 5 (3): a013003. doi:10.1101/cshperspect.a013003. ISSN 1943-0264. Archived from the original on 22 February 2023. Retrieved 7 March 2023.
- ↑ Yadav, Rohitash; Chaudhary, Jitendra Kumar; Jain, Neeraj; Chaudhary, Pankaj Kumar; Khanra, Supriya; Dhamija, Puneet; Sharma, Ambika; Kumar, Ashish; Handu, Shailendra (6 April 2021). "Role of Structural and Non-Structural Proteins and Therapeutic Targets of SARS-CoV-2 for COVID-19". Cells. 10 (4): 821. doi:10.3390/cells10040821. ISSN 2073-4409. Archived from the original on 31 October 2022. Retrieved 18 March 2023.
- ↑ Balakrishnan, Lata; Bambara, Robert A. (1 February 2013). "Okazaki fragment metabolism". Cold Spring Harbor Perspectives in Biology. 5 (2): a010173. doi:10.1101/cshperspect.a010173. ISSN 1943-0264. Archived from the original on 6 February 2023. Retrieved 7 March 2023.
- ↑ Berk, A. J. (1986). "Functions of adenovirus E1A". Cancer Surveys. 5 (2): 367–387. ISSN 0261-2429. Archived from the original on 17 June 2022. Retrieved 17 March 2023.
- ↑ Singh G, Ismail AM, Lee JY, Ramke M, Lee JS, Dyer DW, et al. (February 2019). "Divergent Evolution of E1A CR3 in Human Adenovirus Species D". Viruses. 11 (2): 143. doi:10.3390/v11020143. PMC 6409611. PMID 30744049.
- ↑ Granoff, Allan; Webster, Robert G. (27 July 1999). Encyclopedia of Virology. Elsevier. ISBN 978-0-08-054797-8. Archived from the original on 22 March 2023. Retrieved 22 March 2023.google books does not offer page number
- ↑ 51.0 51.1 Yamamoto H, Shimojo H (August 1971). "Multiplicity reactivation of human adenovirus type 12 and simian virus 40 irradiated by ultraviolet light". Virology. 45 (2): 529–31. doi:10.1016/0042-6822(71)90355-2. PMID 4328814.
- ↑ Michod RE, Bernstein H, Nedelcu AM (May 2008). "Adaptive value of sex in microbial pathogens". Infection, Genetics and Evolution. 8 (3): 267–85. doi:10.1016/j.meegid.2008.01.002. PMID 18295550.
- ↑ Wang CC, Prather KA, Sznitman J, Jimenez JL, Lakdawala SS, Tufekci Z, Marr LC (August 2021). "Airborne transmission of respiratory viruses". Science. 373 (6558): eabd9149. doi:10.1126/science.abd9149. PMC 8721651. PMID 34446582.
- ↑ Wan YY, Leon RP, Marks R, Cham CM, Schaack J, Gajewski TF, DeGregori J (December 2000). "Transgenic expression of the coxsackie/adenovirus receptor enables adenoviral-mediated gene delivery in naive T cells". Proceedings of the National Academy of Sciences of the United States of America. 97 (25): 13784–13789. Bibcode:2000PNAS...9713784W. doi:10.1073/pnas.250356297. PMC 17653. PMID 11095726.
- ↑ Vento, Todd J.; Prakash, Vidhya; Murray, Clinton K.; Brosch, Lorie C.; Tchandja, Juste B.; Cogburn, Cynthia; Yun, Heather C. (15 May 2011). "Pneumonia in military trainees: a comparison study based on adenovirus serotype 14 infection". The Journal of Infectious Diseases. 203 (10): 1388–1395. doi:10.1093/infdis/jir040. ISSN 1537-6613. Archived from the original on 21 August 2020. Retrieved 9 March 2023.
- ↑ Chen LH, Wu ZQ, Hu YF, Yang F, Yang J, Jin Q (June 2012). "[Genetic diversity of adenoviruses in bats of China]". Bing du Xue Bao = Chinese Journal of Virology. 28 (4): 403–8. PMID 22978165.
- ↑ Li Y, Ge X, Zhang H, Zhou P, Zhu Y, Zhang Y, et al. (April 2010). "Host range, prevalence, and genetic diversity of adenoviruses in bats". Journal of Virology. 84 (8): 3889–97. doi:10.1128/JVI.02497-09. PMC 2849498. PMID 20089640.
- ↑ 58.0 58.1 Fenner FJ, Gibbs EP, Murphy FA, Rott R, Studdert MJ, White DO (1993). Veterinary Virology (2nd ed.). Academic Press, Inc. ISBN 978-0-12-253056-2.
- ↑ Abendroth, Björn; Höper, Dirk; Ulrich, Rainer G.; Larres, Gudrun; Beer, Martin (October 2017). "A red squirrel associated adenovirus identified by a combined microarray and deep sequencing approach". Archives of Virology. 162 (10): 3167–3172. doi:10.1007/s00705-017-3463-5. ISSN 1432-8798. Archived from the original on 29 December 2020. Retrieved 9 March 2023.
- ↑ Goldstein T, Colegrove KM, Hanson M, Gulland FM (May 2011). "Isolation of a novel adenovirus from California sea lions Zalophus californianus". Diseases of Aquatic Organisms. 94 (3): 243–8. doi:10.3354/dao02321. PMID 21790072.
- ↑ "Inclusion Body Hepatitis and Hepatitis Hydropericardium Syndrome in Poultry - Poultry". Veterinary Manual. Archived from the original on 2022-11-29. Retrieved 2023-02-14.
- ↑ Kumar R, Kumar V, Asthana M, Shukla SK, Chandra R (January 2010). "Isolation and identification of a fowl adenovirus from wild Black Kites (Milvus migrans)". Journal of Wildlife Diseases. 46 (1): 272–6. doi:10.7589/0090-3558-46.1.272. PMID 20090043.
- ↑ Chen EC, Yagi S, Kelly KR, Mendoza SP, Tarara RP, Canfield DR, et al. (July 2011). Nemerow GR (ed.). "Cross-species transmission of a novel adenovirus associated with a fulminant pneumonia outbreak in a new world monkey colony". PLOS Pathogens. 7 (7): e1002155. doi:10.1371/journal.ppat.1002155. PMC 3136464. PMID 21779173.
- ↑ "Croup". The Lecturio Medical Concept Library. 30 April 2020. Archived from the original on 13 July 2021. Retrieved 11 July 2021.
- ↑ Uhnoo I, Svensson L, Wadell G (September 1990). "Enteric adenoviruses". Bailliere's Clinical Gastroenterology. 4 (3): 627–42. doi:10.1016/0950-3528(90)90053-j. PMID 1962727.
- ↑ 66.0 66.1 "Symptoms of Adenovirus". Centers for Disease Control and Prevention. 24 January 2023. Archived from the original on 28 February 2023. Retrieved 8 March 2023.
- ↑ Proenca‐Modena, José Luiz; de Souza Cardoso, Ricardo; Criado, Miriã Ferreira; Milanez, Guilherme Paier; de Souza, William Marciel; Parise, Pierina Lorencini; Bertol, Jéssica Wildgrube; de Jesus, Bruna Lais Santos; Prates, Mirela Cristina Moreira; Silva, Maria Lúcia; Buzatto, Guilherme Pietrucci; Demarco, Ricardo Cassiano; Valera, Fabiana Cardoso Pereira; Tamashiro, Edwin; Anselmo‐Lima, Wilma Terezinha; Arruda, Eurico (2019). "Human adenovirus replication and persistence in hypertrophic adenoids and palatine tonsils in children". Journal of Medical Virology. 91 (7): 1250–1262. doi:10.1002/jmv.25441. ISSN 0146-6615. Archived from the original on 11 December 2022. Retrieved 16 March 2023.
- ↑ Vidal, Luine Rosele; de Almeida, Sérgio Monteiro; Cavalli, Bárbara Maria; Dieckmann, Tatiana Gutierrez; Raboni, Sonia Mara; Salvador, Gabriel L. O.; Pereira, Luciane Aparecida; Rotta, Indianara; Nogueira, Meri Bordignon (August 2019). "Human adenovirus meningoencephalitis: a 3-years' overview". Journal of Neurovirology. 25 (4): 589–596. doi:10.1007/s13365-019-00758-7. ISSN 1538-2443. Archived from the original on 14 March 2022. Retrieved 13 March 2023.
- ↑ "Hemorrhagic Cystitis: Practice Essentials, Anatomy, Pathophysiology". Medscape. 3 October 2022. Archived from the original on 12 February 2023. Retrieved 18 March 2023.
- ↑ Gabrielli, Andrea; Layon, A. Joseph; Yu, Mihae (17 November 2011). Civetta, Taylor, and Kirby's Manual of Critical Care. Lippincott Williams & Wilkins. ISBN 978-0-7817-6915-0. Archived from the original on 20 March 2023. Retrieved 20 March 2023.
- ↑ Burkholder A (2007-12-19). "A killer cold? Even the healthy may be vulnerable". CNN. Archived from the original on 2021-01-28. Retrieved 2007-12-19.
- ↑ Hosseini SM, Mirhosseini SM, Taghian M, Salehi M, Farahani MM, Bakhtiari F, et al. (October 2018). "First evidence of the presence of adenovirus type 8 in myocardium of patients with severe idiopathic dilated cardiomyopathy". Archives of Virology. 163 (10): 2895–2897. doi:10.1007/s00705-018-3942-3. PMID 30022238. S2CID 49870344.
- ↑ "How Adenovirus Spreads | CDC". www.cdc.gov. 3 March 2023. Archived from the original on 17 January 2023. Retrieved 8 March 2023.
- ↑ Evans, Alfred S.; Kaslow, Richard A. (30 June 1997). Viral Infections of Humans: Epidemiology and Control. Springer Science & Business Media. p. 129. ISBN 978-0-306-44855-3. Archived from the original on 10 March 2023. Retrieved 10 March 2023.
- ↑ Usman, Norina; Suarez, Manuel (2022). "Adenoviruses". StatPearls. StatPearls Publishing. Archived from the original on 2023-01-31. Retrieved 2023-03-19.
- ↑ Canada, Public Health Agency of (19 April 2011). "Pathogen safety data sheet: Infectious substances – Adenovirus (serotypes 40 and 41)". www.canada.ca. Archived from the original on 20 August 2022. Retrieved 19 March 2023.
- ↑ "Default - Stanford Children's Health". Archived from the original on April 3, 2020.
- ↑ "Adenovirus | Clinical Diagnosis | CDC". 2019-08-29. Archived from the original on 2021-09-05. Retrieved 2023-02-14.
- ↑ 79.0 79.1 "Adenovirus | Prevention and Treatment | CDC". 2019-09-03. Archived from the original on 2022-12-22. Retrieved 2023-02-14.
- ↑ 80.0 80.1 "Adenovirus Vaccine Information Statement | CDC". www.cdc.gov. 23 November 2022. Archived from the original on 14 August 2021. Retrieved 11 March 2023.
- ↑ "USAMRMC protects Soldiers against unseen enemy". Archived from the original on 2022-06-26. Retrieved 2023-02-14.
- ↑ Gu, Jie; Su, Qing-Qing; Zuo, Ting-Ting; Chen, Yan-Bin (February 2021). "Adenovirus diseases: a systematic review and meta-analysis of 228 case reports". Infection. 49 (1): 1–13. doi:10.1007/s15010-020-01484-7. ISSN 1439-0973. PMC 7962627. PMID 32720128.
- ↑ "Cidofovir". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 9 August 2022. Retrieved 15 March 2023.
- ↑ Thacker EE, Nakayama M, Smith BF, Bird RC, Muminova Z, Strong TV, et al. (November 2009). "A genetically engineered adenovirus vector targeted to CD40 mediates transduction of canine dendritic cells and promotes antigen-specific immune responses in vivo". Vaccine. 27 (50): 7116–24. doi:10.1016/j.vaccine.2009.09.055. PMC 2784276. PMID 19786146.
- ↑ Harrington KJ, Vile RG, Pandha HS, eds. (May 2008). Viral Therapy of Cancer. Hoboken, N.J.: Wiley. pp. 1–13. ISBN 9780470019221.
- ↑ Xin KQ, Sekimoto Y, Takahashi T, Mizuguchi H, Ichino M, Yoshida A, Okuda K (May 2007). "Chimeric adenovirus 5/35 vector containing the clade C HIV gag gene induces a cross-reactive immune response against HIV". Vaccine. 25 (19): 3809–15. doi:10.1016/j.vaccine.2007.01.117. PMID 17386962.
- ↑ Naskalska A, Szolajska E, Chaperot L, Angel J, Plumas J, Chroboczek J (December 2009). "Influenza recombinant vaccine: matrix protein M1 on the platform of the adenovirus dodecahedron". Vaccine. 27 (52): 7385–93. doi:10.1016/j.vaccine.2009.09.021. PMID 19766576.
- ↑ Nóbrega, Clévio (2020). A handbook of gene and cell therapy. Liliana Mendonça, Carlos A. Matos. Cham: Springer. ISBN 978-3-030-41333-0. OCLC 1163431307. Archived from the original on 2023-03-04. Retrieved 2023-02-14.
- ↑ Boucher, Paul; Cui, Xiaoxia; Curiel, David T. (10 November 2020). "Adenoviral vectors for in vivo delivery of CRISPR-Cas gene editors". Journal of Controlled Release: Official Journal of the Controlled Release Society. 327: 788–800. doi:10.1016/j.jconrel.2020.09.003. ISSN 1873-4995. Archived from the original on 8 March 2023. Retrieved 8 March 2023.
- ↑ Cross R (12 May 2020). "Adenoviral vectors are the new COVID-19 vaccine front-runners. Can they overcome their checkered past?". Chemical & Engineering News. 98 (19). Archived from the original on 23 August 2020. Retrieved 15 December 2020.
- ↑ 91.0 91.1 Cohen J (19 October 2020). "Could certain COVID-19 vaccines leave people more vulnerable to the AIDS virus?". American Association for the Advancement of Science. Science (magazine). Archived from the original on 3 October 2021. Retrieved 15 December 2020.
- ↑ Folegatti PM, Ewer KJ, Aley PK, Angus B, Becker S, Belij-Rammerstorfer S, et al. (August 2020). "Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: a preliminary report of a phase 1/2, single-blind, randomised controlled trial". Lancet. 396 (10249): 467–478. doi:10.1016/S0140-6736(20)31604-4. PMC 7445431. PMID 32702298.
- ↑ "The Oxford/AstraZeneca COVID-19 vaccine: what you need to know". www.who.int. Archived from the original on 2022-11-26. Retrieved 2021-03-07.
- ↑ 94.0 94.1 "An Open Study of the Safety, Tolerability and Immunogenicity of the Drug "Gam-COVID-Vac" Vaccine Against COVID-19". Clinical Trials. 22 June 2020. Archived from the original on 4 May 2021. Retrieved 22 December 2020.
- ↑ 95.0 95.1 Zhu FC, Li YH, Guan XH, Hou LH, Wang WJ, Li JX, et al. (June 2020). "Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial". Lancet. 395 (10240): 1845–1854. doi:10.1016/s0140-6736(20)31208-3. PMC 7255193. PMID 32450106.
- ↑ "Experimental Nasal Vaccine Protects Upper and Lower Respiratory Tracts against SARS-CoV-2". Sci News. 27 August 2020. Archived from the original on 27 August 2020. Retrieved 28 August 2020.
- ↑ Hassan AO, Kafai NM, Dmitriev IP, Fox JM, Smith BK, Harvey IB, et al. (October 2020). "A Single-Dose Intranasal ChAd Vaccine Protects Upper and Lower Respiratory Tracts against SARS-CoV-2". Cell. 183 (1): 169–184.e13. doi:10.1016/j.cell.2020.08.026. PMC 7437481. PMID 32931734.
- ↑ Logunov DY, Dolzhikova IV, Zubkova OV, Tukhvatulin AI, Shcheblyakov DV, Dzharullaeva AS, et al. (September 2020). "Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: two open, non-randomised phase 1/2 studies from Russia". Lancet. 396 (10255): 887–897. doi:10.1016/S0140-6736(20)31866-3. PMC 7471804. PMID 32896291.
- ↑ Fausther-Bovendo H, Kobinger GP (2014). "Pre-existing immunity against Ad vectors: humoral, cellular, and innate response, what's important?". Human Vaccines & Immunotherapeutics. 10 (10): 2875–84. doi:10.4161/hv.29594. PMC 5443060. PMID 25483662.
- ↑ Collins S (9 December 2020). "HIV risk from some COVID-19 vaccines might be unlikely due to rarity of vector viruses involved". HIV i-BASE. Archived from the original on 1 December 2020. Retrieved 15 December 2020.
- ↑ Fauci AS, Marovich MA, Dieffenbach CW, Hunter E, Buchbinder SP (2014-04-04). "Immune Activation with HIV Vaccines: Implications of the Adenovirus Vector Experience". Science. 344 (6179): 49–51. doi:10.1126/science.1250672. ISSN 0036-8075. PMC 4414116. PMID 24700849.
- ↑ Rosenberg J (25 October 2020). "Researchers Warn of Heightened Risk of HIV With Certain COVID-19 Vaccines". AJMC. Archived from the original on 16 December 2020. Retrieved 15 December 2020.
- ↑ Buchbinder SP, McElrath MJ, Dieffenbach C, Corey L (2020-10-31). "Use of adenovirus type-5 vectored vaccines: a cautionary tale". Lancet. 396 (10260): e68–e69. doi:10.1016/S0140-6736(20)32156-5. PMC 7571904. PMID 33091364.
- ↑ Buhl L. "Most COVID-19 Vaccines Won't Affect HIV Risk: Here's What the Science Tells Us". Archived from the original on 2021-06-13. Retrieved 2023-02-14.
- ↑ Hu H, Eller MA, Zafar S, Zhou Y, Gu M, Wei Z, et al. (2014-09-16). "Preferential infection of human Ad5-specific CD4 T cells by HIV in Ad5 naturally exposed and recombinant Ad5-HIV vaccinated individuals". Proceedings of the National Academy of Sciences of the United States of America. 111 (37): 13439–44. Bibcode:2014PNAS..11113439H. doi:10.1073/pnas.1400446111. ISSN 0027-8424. PMC 4169982. PMID 25197078.
- ↑ Auclair S, Liu F, Niu Q, Hou W, Churchyard G, Morgan C, et al. (2018-02-23). "Distinct susceptibility of HIV vaccine vector-induced CD4 T cells to HIV infection". PLOS Pathogens. 14 (2): e1006888. doi:10.1371/journal.ppat.1006888. ISSN 1553-7374. PMC 5841825. PMID 29474461.
External links
- Centers for Disease Control and Prevention—National Center for Diseases—Division of Viral and Rickettsial Diseases, Respiratory and Enteric Viruses Branch
- MicrobiologyBytes: Adenoviruses
- General information on Adenovirus Archived 2023-01-31 at the Wayback Machine
- Sequenced adenoviruses Archived 2021-01-26 at the Wayback Machine
- Viralzone: Adenoviridae Archived 2010-06-20 at the Wayback Machine
- Adenovirus Research Discussion Group Archived 2012-07-30 at the Wayback Machine
- 3D macromolecular structures of Adenoviruses archived in the EM Data Bank(EMDB) Archived 2023-03-04 at the Wayback Machine
- Molecule of the Month: Adenovirus Archived 2015-10-17 at the Wayback Machine
- ICTV Archived 2015-07-10 at the Wayback Machine