Simeprevir
 | |
| Names | |
|---|---|
| Pronunciation | /sɪˈmɛprəvɪər/ si-MEP-rə-veer |
| Trade names | Olysio, Sovriad, Galexos, others |
| Other names | TMC435; TMC435350 |
| |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | By mouth (capsules) |
| Defined daily dose | 0.15 g [2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614013 |
| Legal | |
| License data | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 62% (under fed conditions) |
| Protein binding | >99.9% |
| Metabolism | Liver (CYP3A, CYP2C8, CYP2C19) |
| Elimination half-life | 10–13 hours (HCV-uninfected subjects), 41 hours (HCV-infected subjects) |
| Excretion | Feces (91%), urine (<1%) |
| Chemical and physical data | |
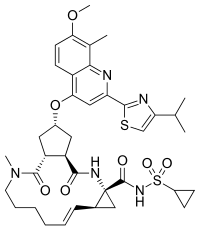
| Formula | C38H47N5O7S2 |
| Molar mass | 749.94 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Simeprevir, sold under the trade names Olysio among others, is a medication used in combination with other medications for the treatment of hepatitis C.[1] It is specifically used for hepatitis C genotype 1 and 4.[1] Medications it is used with include sofosbuvir or ribavirin and peginterferon-alfa.[1] Cure rates are in 80s to 90s percent.[3][4][5] It may be used in those who also have HIV/AIDS.[1] It is taken by mouth once daily for typically 12 weeks.[1]
Common side effects include feeling tired, headache, rash, itchiness, and sensitivity to sunlight.[1] In those with previous hepatitis B infection, active disease may recur.[1] It is not recommended in those with significant liver problems.[1] During pregnancy when used with ribavirin it may cause harm to the baby while when used with sofosbuvir its safety is unclear.[1][6] Simeprevir is a HCV protease inhibitor.[1]
Simeprevir was developed by Medivir AB and Janssen Pharmaceutica.[7] It was approved for medical use in the United States in 2013.[8] It was removed from the World Health Organization's List of Essential Medicines in 2019.[9][10] It is not available as a generic medication as of 2015[update].[6] In the United Kingdom a course of treatment with ribavirin and peginterferon-alfa cost about £29,700 in 2015.[11] In the United States a course of treatment with sofosbuvir was more than US$171,000 in 2015 with the simeprevir component costing US$66,360.[12]
Medical use
Simeprevir is indicated treating chronic hepatic C (CHC) infection as a part of a triple antiviral treatment regimen consisting of two other drugs: peginterferon-alfa (PEG-IFN) and ribavirin (RBV).[13] It is primarily effective in treating Hepatitis C virus (HCV) genotype 1 infected subjects with compensated liver disease, including cirrhosis.[13] There are currently no studies that show Simeprevir's effectiveness as a single therapy for HCV.[13] Simeprevir is generally used for HCV genotype 1 infected subjects, but off-label medical use has been indicated for type 4 genotype as well.[14]
Dosing
The defined daily dose is 0.15 gram (by mouth)[2]Simeprevir is dosed along with peg-IFN and RBV as triple therapy.[13] Appropriate dosing of Simeprevir is dependent upon the patient's liver function, kidney function, viral load, and HCV genotype.[13] This medication is not recommended for people with moderate or severe liver impairment and people with end-stage kidney disease since Simprevir was not studied for use in these patient populations.[13] Simeprevir might be discontinued depending on their viral load.[13] For instance, if the patient's viral load is detectable (>25 units/mL) during the 4th week of their treatment regimen, it is considered an inadequate treatment and simeprevir must be discontinued.[13]
Contraindications
Any contraindications that apply to peg-IFN and RBV apply to simeprevir since they must be used in combination during treatment of CHC. For example, people with sickle cell anemia are contraindicated to RBV therapy and are therefore contraindicated to simeprevir and peg-IFN combination therapy.[15] Pregnant women and men whose female partners are pregnant are contraindicated for simeprevir since peg-IFN and RBV are known to cause birth defects.[13][15][16]
Pregnancy
Simeprevir is avoided in pregnant women or women planning to be pregnant because it going to be taken with RBV and Peg-IFN, which have both shown to cause fetal problems in animal studies.[13][15][16] RBV has been shown to cause birth defects and fetal deaths in animal studies.[15] Peg-IFN has been shown to cause abortions in animal studies.[16] People must have a negative pregnancy test prior to starting therapy, use at least two effective birth control methods during treatment, and undergo monthly pregnancy tests.[13] If pregnant women are exposed to any medication regimen containing ribavirin, they are encouraged to report this through the ribavirin pregnancy registry.[15]
Side effects
Severe itching (22%), sensitivity to sunlight (5%), and rash (25%) are some of the common adverse effects of simeprevir.[17] Other side effects may include nausea, muscle pain, difficulty breathing and increased bilirubin.[18] It may reactivate hepatitis B in those who have been previously infected.[19] The European Medicines Agency (EMA) has recommended screening all people for hepatitis B before starting simeprevir for hepatitis C in order to minimize the risk of hepatitis B reactivation.[20]
Combination treatment
In March 2015, Gilead Sciences e-mailed warnings to health care providers about nine people that began taking its hepatitis C drugs ledipasvir/sofosbuvir or sofosbuvir along with amiodarone, daclatasvir, or simeprevir developed abnormally slow heartbeats and one died of cardiac arrest. Three required a pacemaker to be inserted. Gilead said the combinations aren't recommended and product labels will be updated.[21]
Mechanism of action
Simeprevir is a hepatitis C virus protease inhibitor.[22]
Simeprevir is a NS3/4A protease inhibitor, thus preventing viral maturation through inhibition of protein synthesis. Simeprevir is administered as one capsule once daily with pegylated interferon and ribavirin for the treatment of genotype 1 or genotype 4 chronic hepatitis C in adult people with compensated liver disease (including cirrhosis), with or without HIV-1 co-infection, who are treatment naive or who have failed previous interferon therapy.[23][24] Genotype 1 is the most prevalent form of hepatitis C virus (HCV) worldwide.
Pharmacokinetics
Simeprevir is orally bioavailable. Its absorption increases when taken with food, and is therefore advised to be taken with food.[13] The liver's CYP3A4 enzymes mainly break down simeprevir, but CYP2C8 and CYP2C19 enzymes can also play a role.[13] Its half-life in the plasma is 41 hours in people with HCV.[13] Its peak effect happens 4 to 6 hours after taking the medication.[13] It is primarily excreted into the feces (91%).[13]
Pharmacogenomics
According to simeprevir's prescriber information, its efficacy in combination with peginterferon alfa and ribavirin is "substantially reduced in people with HCV genotype 1a with an NS3 Q80K polymorphism at baseline compared to people infected with HCV genotype 1a without Q80K polymorphism."[13] People with Q80K polymorphism are not advised to take simeprevir.[13]
Drug interactions
Simeprevir is a CYP3A4 substrate so its plasma concentration will significantly increase if taken with medications that are strong CYP3A4 inhibitors (i.e. erythromycin, ritonavir) and will significantly decrease if taken with strong CYP3A4 inducers (i.e. efavirenz, rifampin, Saint John's wort).[13] Simeprevir also inhibits intestinal (but not liver) CYP3A. For instance, midazolam, an anticonvulsant, gets metabolized by intestinal CYP3As and taking it with simeprevir can lead to increased midazolam levels that can be toxic.[13] Simeprevir also inhibits OATP1B1/3 and P-glycoprotein (P-gp) transporters, which are normally transporters that pump out drug out of the plasma.[13][25] Thus, taking simeprevir with medications that are substrates for these transporters can lead to increased plasma concentrations of these medications. For example, calcium channel blockers (i.e. diltiazem, amlodipine) are P-gp substrates and can lead to increased concentrations of these drugs when taken with simeprevir.[13] Taking ciclosporin, a substrate for OATP1B1/3, with simeprevir resulted in significant increase in ciclosporin concentration and are therefore not recommended to be taken together.[13]
Approval
In the United States, it is approved by the Food and Drug Administration (FDA) for use in combination with peginterferon-alfa and ribavirin for hepatitis C.[26] Simeprevir has been approved in Japan for the treatment of chronic hepatitis C infection, genotype 1.[27]
Clinical study
Simeprevir has been tested in combination regimens with pegylated interferon alfa-2a and ribavirin,[28] and in interferon-free regimens with other direct-acting antiviral agents including daclatasvir[29] and sofosbuvir.[30]
Results from three phase 3 randomized, double-blind, placebo controlled clinical trials (C208, C216, and HPC3007) in people with chronic HCV GT1 were favourable and resulted in FDA supporting the approval of simeprevir for Hepatitis C genotype 1.[17] Members of the FDA commented following a presentation by Johnson & Johnson (24 October 2013) that post-marketing studies in racial and ethnic minorities, people co-infected with HIV, and other underrepresented populations are needed.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Simeprevir". The American Society of Health-System Pharmacists. Archived from the original on 1 December 2016. Retrieved 30 November 2016.
- ↑ 2.0 2.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 28 November 2020. Retrieved 21 September 2020.
- ↑ "Initial treatment of HCV infection". www.hcvguidelines.org. October 2016. Archived from the original on 7 December 2016. Retrieved 1 December 2016.
- ↑ Majumdar, A; Kitson, MT; Roberts, SK (June 2016). "Systematic review: current concepts and challenges for the direct-acting antiviral era in hepatitis C cirrhosis". Alimentary Pharmacology & Therapeutics. 43 (12): 1276–92. doi:10.1111/apt.13633. PMID 27087015.
- ↑ Brochot, E; Helle, F; François, C; Castelain, S; Capron, D; Nguyen-Khac, E; Duverlie, G (April 2015). "Which therapeutic option for hepatitis C virus genotype 1?". Scandinavian Journal of Gastroenterology. 50 (4): 470–8. doi:10.3109/00365521.2014.978364. PMID 25396710.
- ↑ 6.0 6.1 Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 80. ISBN 9781284057560.
- ↑ Grubbs, Robert H.; O'Leary, Daniel J. (2015). Handbook of Metathesis, Volume 2: Applications in Organic Synthesis. John Wiley & Sons. p. 699. ISBN 9783527694020. Archived from the original on 29 August 2021. Retrieved 11 September 2017.
- ↑ Dugum M, O'Shea R (2014). "Hepatitis C virus: Here comes all-oral treatment". Cleveland Clinic Journal of Medicine. 81 (3): 159–72. doi:10.3949/ccjm.81a.13155. PMID 24591471.
- ↑ World Health Organization (2019). Executive summary: the selection and use of essential medicines 2019: report of the 22nd WHO Expert Committee on the selection and use of essential medicines. Geneva: World Health Organization. hdl:10665/325773. WHO/MVP/EMP/IAU/2019.05. License: CC BY-NC-SA 3.0 IGO.
- ↑ World Health Organization (2019). The selection and use of essential medicines: report of the WHO Expert Committee on Selection and Use of Essential Medicines, 2019 (including the 21st WHO Model List of Essential Medicines and the 7th WHO Model List of Essential Medicines for Children). Geneva: World Health Organization. hdl:10665/330668. ISBN 9789241210300. ISSN 0512-3054. WHO technical report series;1021.
- ↑ "Janssen will cover cost of unsuccessful hepatitis C treatment". PMLive. 16 January 2015. Archived from the original on 2 December 2016. Retrieved 1 December 2016.
- ↑ Smith, Michael (28 February 2015). "HCV Drugs Costly but Cure Might Not Be". MedPageToday. Archived from the original on 1 December 2016. Retrieved 1 December 2016.
- ↑ 13.00 13.01 13.02 13.03 13.04 13.05 13.06 13.07 13.08 13.09 13.10 13.11 13.12 13.13 13.14 13.15 13.16 13.17 13.18 13.19 13.20 13.21 13.22 "OLYSIO (simeprevir) capsules, for oral use FULL PRESCRIBING INFORMATION". September 2014. Retrieved 24 October 2014.[permanent dead link]
- ↑ "Recommendations for Testing, Managing, and Treating Hepatitis C". 2014. Archived from the original on 16 October 2014. Retrieved 24 October 2014.
- ↑ 15.0 15.1 15.2 15.3 15.4 "Highlights of Prescribing Information Copegus" (PDF). August 2011. Archived (PDF) from the original on 3 November 2014. Retrieved 24 October 2014.
- ↑ 16.0 16.1 16.2 "Highlights of Prescribing Information PEGINTRON" (PDF). July 2014. Archived (PDF) from the original on 3 November 2014. Retrieved 24 October 2014.
- ↑ 17.0 17.1 "FDA ANTIVIRAL DRUGS ADVISORY COMMITTEE MEETING" (PDF). October 2013. Archived (PDF) from the original on 26 December 2014. Retrieved 24 October 2014.
- ↑ "Olysio (simeprevir) dosing, indications, interactions, adverse effects, and more". reference.medscape.com. Archived from the original on 10 November 2016. Retrieved 10 November 2016.
- ↑ "Direct-Acting Antivirals for Hepatitis C: Drug Safety Communication - Risk of Hepatitis B Reactivating". FDA. 4 October 2016. Archived from the original on 6 October 2016. Retrieved 6 October 2016.
- ↑ "Direct-acting antivirals indicated for treatment of hepatitis C (interferon-free)". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 3 February 2020. Retrieved 4 February 2020.
- ↑ West, Stephen. "Gilead Warns After Hepatitis Patient on Heart Drug Dies" Archived 2017-03-22 at the Wayback Machine. Published 21 March 2015.
- ↑ Lin, TI; Lenz, O; Fanning, G; Verbinnen, T; Delouvroy, F; Scholliers, A; Vermeiren, K; Rosenquist, A; Edlund, M; Samuelsson, B.; Vrang, L.; De Kock, H.; Wigerinck, P.; Raboisson, P.; Simmen, K. (2009). "In vitro activity and preclinical profile of TMC435350, a potent hepatitis C virus protease inhibitor". Antimicrobial Agents and Chemotherapy. 53 (4): 1377–85. doi:10.1128/AAC.01058-08. PMC 2663092. PMID 19171797.
- ↑ European Association for the Study of the Liver (2011). "EASL Clinical Practice Guidelines: Management of hepatitis C virus infection". Journal of Hepatology. 55 (2): 245–64. doi:10.1016/j.jhep.2011.02.023. PMID 21371579.
- ↑ Zein NN (2000). "Clinical Significance of Hepatitis C Virus Genotypes". Clin. Microbiol. Rev. 13 (2): 223–235. doi:10.1128/CMR.13.2.223-235.2000. PMC 100152. PMID 10755999.
- ↑ Furihata, T; Matsumoto, S; Fu, Z; Tsubota, A; Sun, Y; Matsumoto, S; Kobayashi, K; Chiba, K (2014). "Different interaction profiles of direct-acting anti-hepatitis C virus agents with human organic anion transporting polypeptides". Antimicrobial Agents and Chemotherapy. 58 (8): 4555–64. doi:10.1128/AAC.02724-14. PMC 4135986. PMID 24867984.
- ↑ "FDA approves new treatment for hepatitis C virus". Food and Drug Administration. 22 November 2013. Archived from the original on 16 December 2013.
- ↑ "Medivir: Simeprevir has been approved in Japan for the treatment of genotype 1 chronic hepatitis C infection". The Wall Street Journal. 27 September 2013. Archived from the original on 24 November 2013.
- ↑ "Phase 3 Studies Show Simeprevir plus Interferon/Ribavirin Cures Most Patients in 24 Weeks". hivandhepatitis.com. 27 December 2012. Archived from the original on 13 March 2013.
- ↑ Medivir announces TMC435 in an expanded clinical collaboration[permanent dead link]. Medivir. 18 April 2012.
- ↑ Results from a phase IIa study evaluating Simeprevir and Sofosbuvir in prior null responder Hepatitis C patients have been presented at CROI. 6 March 2013. Archived 16 October 2012 at the Wayback Machine
Further reading
- Dean L (2016). "Simeprevir Therapy and IFNL3 Genotype". In Pratt VM, McLeod HL, Rubinstein WS, et al. (eds.). Medical Genetics Summaries. National Center for Biotechnology Information (NCBI). PMID 28520373. Bookshelf ID: NBK385156. Archived from the original on 26 October 2020. Retrieved 5 February 2020.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- All articles with dead external links
- Articles with dead external links from May 2018
- Articles with invalid date parameter in template
- Articles with permanently dead external links
- Webarchive template wayback links
- Articles with dead external links from March 2022
- Use dmy dates from September 2019
- Drugs with non-standard legal status
- Chemical articles having Jmol set/None
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Chemical articles having Jmol set
- Drugs missing an ATC code
- Drug has EMA link
- Articles containing unverified chemical infoboxes
- Articles containing potentially dated statements from 2015
- All articles containing potentially dated statements
- Cyclopentanes
- Cyclopropanes
- Isopropyl compounds
- Johnson & Johnson brands
- Macrocycles
- NS3/4A protease inhibitors
- Quinolines
- RTT
- Sulfonamides
- Thiazoles