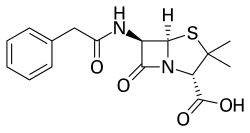
Benzylpenicillin
 | |
| Names | |
|---|---|
| Trade names | Pfizerpen, other |
| Other names | Penicillin G potassium,[1] penicillin G sodium |
| |
| Clinical data | |
| Drug class | Penicillin (β-lactam)[2] |
| Main uses | Pneumonia, strep throat, syphilis, necrotizing enterocolitis, diphtheria, gas gangrene, leptospirosis, cellulitis, tetanus[2] |
| Side effects | Diarrhea, seizures, allergic reactions including anaphylaxis[2] |
| WHO AWaRe | |
| Pregnancy category |
|
| Routes of use | IV, IM |
| Defined daily dose | 3.6 grams[4] |
| External links | |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a685013 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Protein binding | 60 % |
| Metabolism | Liver |
| Elimination half-life | 30 min |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C16H18N2O4S |
| Molar mass | 334.39 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Benzylpenicillin, also known as penicillin G, is an antibiotic used to treat a number of bacterial infections.[2] This includes pneumonia, strep throat, syphilis, necrotizing enterocolitis, diphtheria, gas gangrene, leptospirosis, cellulitis, and tetanus.[2] It is not a first-line agent for pneumococcal meningitis.[2] Benzylpenicillin is given by injection into a vein or muscle.[1] Two long-acting forms, benzathine benzylpenicillin and procaine benzylpenicillin, are available for use by injection into a muscle only.[2][1]
Side effects include diarrhea, seizures, and allergic reactions including anaphylaxis.[2] When used to treat syphilis or Lyme disease a reaction known as Jarisch–Herxheimer may occur.[2] It is not recommended in those with a history of penicillin allergy.[2] Use during pregnancy is generally safe.[1] It is in the penicillin and β-lactam class of medications.[2]
Benzylpenicillin was discovered in 1928 by Alexander Fleming, who published its details the following year.[5][6] It came into commercial use in the early 1940s.[6][7] It is on the World Health Organization's List of Essential Medicines.[8] The wholesale cost in the developing world is about US$0.24–2.72 per day.[9] In the United States a course of treatment costs $100–200.[10]
Medical uses
As an antibiotic, benzylpenicillin is noted to possess effectiveness mainly against Gram-positive organisms. Some Gram-negative organisms such as Neisseria gonorrhoeae and Leptospira weilii are also reported to be susceptible to benzylpenicillin.[11] It is in the 'access' group of the WHO AWaRe classification.[12]
Dosage
The defined daily dose is 3.6 grams (by injection).[4] For neurosyphilis the dose in adults is 1.2 to 2.4 grams (2 to 4 MIU) intravenously every 4 fours.[13] For congenital syphilis 30 mg/kg (50,000 IU/kg) twice a day for the first week and than three times a day for the next three days is used.[13] For diphtheria 600 mg (1 MIU) every six hours in adults or 30 mg/kg (50,000 IU/kg) every six hours is used.[13]
Side effects
Side effects can include hypersensitivity reactions including urticaria, fever, joint pains, rashes, angioedema, anaphylaxis, serum sickness-like reaction. Rarely CNS toxicity including convulsions (especially with high doses or in severe renal impairment), interstitial nephritis, haemolytic anaemia, leucopenia, thrombocytopenia, and coagulation disorders. Also reported diarrhoea (including antibiotic-associated colitis).
Benzylpenicillin serum concentrations can be monitored either by traditional microbiological assay or by more modern chromatographic techniques. Such measurements can be useful to avoid central nervous system toxicity in any person receiving large doses of the drug on a chronic basis, but they are especially relevant to patients with kidney failure, who may accumulate the drug due to reduced urinary excretion rates.[14][15]
Chemistry
History
Benzylpenicillin was discovered in 1928 by Alexander Fleming, who published its details the following year.[5][6] It came into commercial use in 1941[6] or 1942.[7]
Manufacture
The production of benzylpenicillin involves fermentation, recovery and purification of the penicillin.[16]
The fermentation process of the production of benzylpencillin creates the product. The presence of the product in solution inhibits the reaction and reduces the product rate and yield. Thus, in order to obtain the most product and increase the rate of reaction, it is continuously extracted.[17] This is done by mixing the mold with either glucose, sucrose, lactose, starch, or dextrin, nitrate, ammonium salt, corn steep liquor, peptone, meat or yeast extract, and small amounts of inorganic salts.[18]
The recovery of the benzylpencillin is the most important part of the production process because it affects the later purification steps if done incorrectly.[16] There are several types of techniques used to recover benzyl penicillin: aqueous two-phase extraction, liquid membrane extraction, microfiltration, and solvent extraction.[16] Extraction is more commonly used in the recovery process.[medical citation needed]
In the purification step, the benzylpencillin is separated from the extraction solution. This is normally done by using a separation column.[19]
References
- ↑ 1.0 1.1 1.2 1.3 "Penicillin G Injection - FDA prescribing information, side effects and uses". www.drugs.com. Archived from the original on 20 December 2016. Retrieved 10 December 2016.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. pp. 98, 105. hdl:10665/44053. ISBN 9789241547659.
- ↑ David D. Dexter, James M. van der Veen (1978). "Conformations of penicillin G: crystal structure of procaine penicillin G monohydrate and a refinement of the structure of potassium penicillin G". J. Chem. Soc. Perkin Trans. 1: 185–190. doi:10.1039/p19780000185.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ↑ 4.0 4.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 28 July 2020. Retrieved 1 September 2020.
- ↑ 5.0 5.1 Bud, Robert (2008-06-01). "Upheaval in the moral economy of science? Patenting, teamwork and the World War II experience of penicillin". History and Technology. 24 (2): 173–190. doi:10.1080/07341510701810955. ISSN 0734-1512. Archived from the original on 28 August 2021. Retrieved 22 December 2020. Cite error: Invalid
<ref>tag; name "Bud" defined multiple times with different content - ↑ 6.0 6.1 6.2 6.3 Kiliç, Burcu (2014). "3. Innovation and the Pharmaceutical Industry". Boosting Pharmaceutical Innovation in the Post-TRIPS Era. Cheltenham: Edward Elgar Publishing. p. 65. ISBN 978-1-78254-412-8. Archived from the original on 28 August 2021. Retrieved 22 December 2020.
- ↑ 7.0 7.1 Alapi, Erika M.; Fischer, Jánus (2006). "Part III. Table of Selected Analogue Classes". In Fischer, János; Ganellin, C. Robin (eds.). Analogue-based Drug Discovery. Wiley-VCH. p. 490. ISBN 978-3-527-31257-3. Archived from the original on 20 December 2016.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Penicillin, Benzyl". International Drug Price Indicator Guide. Archived from the original on 13 October 2018. Retrieved 8 December 2016.
- ↑ Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 95. ISBN 9781284057560.
- ↑ "Penicillin G" (PDF). Toku-E. 2010-10-10. Archived from the original (PDF) on 2016-03-03. Retrieved 2012-06-11.
- ↑ Zanichelli, Veronica; Sharland, Michael; Cappello, Bernadette; Moja, Lorenzo; Getahun, Haileyesus; Pessoa-Silva, Carmem; Sati, Hatim; van Weezenbeek, Catharina; Balkhy, Hanan; Simão, Mariângela; Gandra, Sumanth; Huttner, Benedikt (1 April 2023). "The WHO AWaRe (Access, Watch, Reserve) antibiotic book and prevention of antimicrobial resistance". Bulletin of the World Health Organization. 101 (4): 290–296. doi:10.2471/BLT.22.288614. ISSN 0042-9686. Archived from the original on 7 May 2023. Retrieved 17 November 2023.
- ↑ 13.0 13.1 13.2 "BENZYLPENICILLIN = PENICILLIN G injectable - Essential drugs". medicalguidelines.msf.org. Archived from the original on 28 August 2021. Retrieved 1 September 2020.
- ↑ Fossieck B Jr, Parker RH. Neurotoxicity during intravenous infusion of penicillin. A review. J. Clin. Pharmacol. 14: 504- 512, 1974.
- ↑ Baselt R (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. pp. 1195–1196.
- ↑ 16.0 16.1 16.2 Liu, Qingfen; Li, Yingbo; Li, Wangliang; Liang, Xiangfeng; Zhang, Chao; Liu, Huizhou (February 2016). "Efficient Recovery of Penicillin G by a Hydrophobic Ionic Liquid". ACS Sustainable Chemistry & Engineering. 4 (2): 609–615. doi:10.1021/acssuschemeng.5b00975.
- ↑ Barros, José (4 January 2016). "Use Extraction to Improve Penicillin G Recovery". American Chemical Society. Archived from the original on 6 May 2019. Retrieved 2019-05-06.
- ↑ "Separation and Purification of Pharmaceuticals and Antibiotics" (PDF). Archived (PDF) from the original on 6 May 2019. Retrieved 6 May 2019.
- ↑ Saino Y, Kobayashi F, Inoue M, Mitsuhashi S (October 1982). "Purification and properties of inducible penicillin beta-lactamase isolated from Pseudomonas maltophilia". Antimicrobial Agents and Chemotherapy. 22 (4): 564–70. doi:10.1128/AAC.22.4.564. PMC 183794. PMID 6983856.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Pages with reference errors
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from February 2020
- Articles with invalid date parameter in template
- Articles with changed KEGG identifier
- Articles with changed EBI identifier
- Benzyl compounds
- Enantiopure drugs
- Glycine receptor agonists
- Penicillins
- World Health Organization essential medicines
- RTT