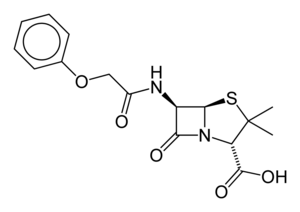
Phenoxymethylpenicillin
 | |
 | |
| Names | |
|---|---|
| Trade names | Veetids, Apocillin,[1] others |
| Other names | penicillin phenoxymethyl, penicillin V, penicillin VK |
| |
| Clinical data | |
| Drug class | Penicillin (beta lactam)[2] |
| Main uses | Strep throat, otitis media, cellulitis[3] |
| Side effects | Diarrhea, nausea, allergic reactions including anaphylaxis[3] |
| WHO AWaRe | |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | 2 grams[4] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a685015 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 60% |
| Protein binding | 80% |
| Metabolism | Liver |
| Elimination half-life | 30–60 min |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C16H18N2O5S |
| Molar mass | 350.39 g·mol−1 |
| 3D model (JSmol) | |
| |
Phenoxymethylpenicillin, also known as penicillin V (PcV) and penicillin VK, is an antibiotic useful for the treatment of a number of bacterial infections.[3] Specifically it is used for the treatment of strep throat, otitis media, and cellulitis.[3] It is also used to prevent rheumatic fever and to prevent infections following removal of the spleen.[3] It is given by mouth.[3]
Side effects include diarrhea, nausea, and allergic reactions including anaphylaxis.[3] It is not recommended in those with a history of penicillin allergy.[3] It is relatively safe for use during pregnancy.[5] It is in the penicillin and beta lactam family of medications.[2] It usually results in bacterial death.[2]
Phenoxymethylpenicillin was first made in 1948.[6] It is on the World Health Organization's List of Essential Medicines.[7] It is available as a generic medication.[2] The wholesale cost in the developing world is about US$0.05–0.96 per day.[8] In the United States a course of treatment costs less than $25.[5] In 2017, it was the 242nd most commonly prescribed medication in the United States, with more than two million prescriptions.[9][10]
Medical uses
It is in the 'access' group of the WHO AWaRe Classification.[11]
Specific uses for phenoxymethylpenicillin include:[12][13]
- Infections caused by Streptococcus pyogenes
- Tonsillitis
- Pharyngitis
- Skin infections
- Anthrax (mild uncomplicated infections)
- Lyme disease (early stage in pregnant women or young children)
- Rheumatic fever (primary and secondary prophylaxis)
- Streptococcal skin infections
- Spleen disorders (pneumococcal infection prophylaxis)
- Initial treatment for dental abscesses
- Moderate-to-severe gingivitis (with metronidazole)
- Avulsion injuries of teeth (as an alternative to tetracycline)
- Blood infection prophylaxis in children with sickle cell disease.
Penicillin V is sometimes used in the treatment of dental infections.
It is less active than benzylpenicillin (penicillin G) against Gram-negative bacteria.[14][15] Phenoxymethylpenicillin has a range of antimicrobial activity against Gram-positive bacteria that is similar to that of benzylpenicillin and a similar mode of action, but it is substantially less active than benzylpenicillin against Gram-negative bacteria.[14][15]
Phenoxymethylpenicillin is more acid-stable than benzylpenicillin, which allows it to be given orally.
Phenoxymethylpenicillin is usually used only for the treatment of mild to moderate infections, and not for severe or deep-seated infections since absorption can be unpredictable. Except for the treatment or prevention of infection with Streptococcus pyogenes (which is uniformly sensitive to penicillin), therapy should be guided by bacteriological studies (including sensitivity tests) and by clinical response.[16] People treated initially with parenteral benzylpenicillin may continue treatment with phenoxymethylpenicillin by mouth once a satisfactory response has been obtained.[12]
It is not active against beta-lactamase-producing bacteria, which include many strains of Staphylococci.[16]
Dosage
The defined daily dose is 2 grams (by mouth).[4] In those who are 40 kg or more this is generally given as 1 gm twice per day while those who weight 20 to 40 kg are treated with 500 mg twice per day.[17] Children under 6 years old may be treated with 250 mg twice per day while those under 1 year old may be treated with 125 mg twice per day.[17]
Side effects
Phenoxymethylpenicillin is usually well tolerated but may occasionally cause transient nausea, vomiting, epigastric distress, diarrhea, constipation, acidic smell to urine and black hairy tongue. A previous hypersensitivity reaction to any penicillin is a contraindication.[12][16]
Mechanism of action
It exerts a bactericidal action against penicillin-sensitive microorganisms during the stage of active multiplication. It acts by inhibiting the biosynthesis of cell-wall peptidoglycan.[citation needed]
Society and culture
Names
Penicillin VK is a potassium salt of penicillin V.[citation needed]
Cost
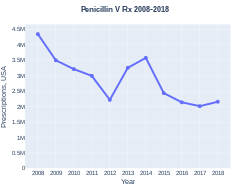
The wholesale cost in the developing world is about US$0.05–0.96 per day.[8] In the United States a course of treatment costs less than $25.[5] In 2017, it was the 242nd most commonly prescribed medication in the United States, with more than two million prescriptions.[9][10]
-
Penicillin V costs (US)
-
Penicillin V prescriptions (US)
Compendial status
References
- ↑ "Apocillin". Felleskatalogen (in Norwegian). LMI (Legemiddelindustrien). Archived from the original on 2018-06-23. Retrieved 2018-06-23.
fenoksymetylpenicillin
{{cite web}}: CS1 maint: unrecognized language (link) - ↑ 2.0 2.1 2.2 2.3 "Penicillin V". The American Society of Health-System Pharmacists. Archived from the original on 20 December 2016. Retrieved 8 December 2016.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. hdl:10665/44053. ISBN 9789241547659.
- ↑ 4.0 4.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 11 January 2020. Retrieved 21 September 2020.
- ↑ 5.0 5.1 5.2 Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 95. ISBN 9781284057560.
- ↑ Greenwood, David (2008). Antimicrobial Drugs: Chronicle of a Twentieth Century Medical Triumph. OUP Oxford. p. 121. ISBN 9780199534845. Archived from the original on 2016-12-20.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 8.0 8.1 "Penicillin, Phenoxymethyl". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 8 December 2016.
- ↑ 9.0 9.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- ↑ 10.0 10.1 "Penicillin V - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
- ↑ Zanichelli, Veronica; Sharland, Michael; Cappello, Bernadette; Moja, Lorenzo; Getahun, Haileyesus; Pessoa-Silva, Carmem; Sati, Hatim; van Weezenbeek, Catharina; Balkhy, Hanan; Simão, Mariângela; Gandra, Sumanth; Huttner, Benedikt (1 April 2023). "The WHO AWaRe (Access, Watch, Reserve) antibiotic book and prevention of antimicrobial resistance". Bulletin of the World Health Organization. 101 (4): 290–296. doi:10.2471/BLT.22.288614. ISSN 0042-9686. Archived from the original on 7 May 2023. Retrieved 17 November 2023.
- ↑ 12.0 12.1 12.2 Sweetman S., ed. (2002). Martindale: The complete drug reference (Electronic version ed.). London: Royal Pharmaceutical Society of Great Britain and the Pharmaceutical Press.
- ↑ Rossi S., ed. (2006). Australian Medicines Handbook. Adelaide: Australian Medicines Handbook Pty Ltd. ISBN 0-9757919-2-3.
- ↑ 14.0 14.1 Garrod, L. P. (1960). "Relative Antibacterial Activity of Three Penicillins". British Medical Journal. 1 (5172): 527–29. doi:10.1136/bmj.1.5172.527. PMC 1966560. PMID 13826674.
- ↑ 15.0 15.1 Garrod, L. P. (1960). "The Relative Antibacterial Activity of Four Penicillins". British Medical Journal. 2 (5214): 1695–6. doi:10.1136/bmj.2.5214.1695. PMC 2098302. PMID 13703756.
- ↑ 16.0 16.1 16.2 "Penicillin V Potassium tablet: Drug Label Sections". U.S. National Library of Medicine, Daily Med: Current Medication Information. December 2006. Archived from the original on 2009-07-27. Retrieved 2009-08-02.
- ↑ 17.0 17.1 "PHENOXYMETHYLPENICILLIN = PENICILLIN V oral - Essential drugs". medicalguidelines.msf.org. Archived from the original on 28 August 2021. Retrieved 23 August 2020.
- ↑ British Pharmacopoeia Commission Secretariat. "Index (BP 2009)" (PDF). Archived from the original (PDF) on 11 April 2009. Retrieved 26 March 2010.
External links
- "Penicillin V". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-10-21. Retrieved 2020-05-18.
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- CS1 maint: unrecognized language
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- Articles with hatnote templates targeting a nonexistent page
- All articles with unsourced statements
- Articles with unsourced statements from May 2020
- Articles with invalid date parameter in template
- Articles with unsourced statements
- Penicillins
- Enantiopure drugs
- World Health Organization essential medicines
- RTT