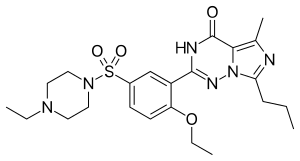
Vardenafil
 | |
| Names | |
|---|---|
| Trade names | Levitra, Staxyn, Vivanza, others |
| |
| Clinical data | |
| Drug class | PDE5 inhibitor[1] |
| Main uses | Erectile dysfunction[1] |
| Side effects | Headache, allergic reactions, low blood pressure, QT prolongation, priapism[1][2] |
| Interactions | Nitrovasodilators[2] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Onset of action | ~45 min[1] |
| Typical dose | 10 mg[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603035 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 15% |
| Protein binding | 95% |
| Metabolism | Liver (CYP3A4) |
| Elimination half-life | 4–5 hours |
| Excretion | Biliary |
| Chemical and physical data | |
| Formula | C23H32N6O4S |
| Molar mass | 488.61 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Vardenafil, sold under the brand name Levitra among others, is a medication used to treat erectile dysfunction.[1] It is taken by mouth.[1] Onset is generally in 30 to 60 minutes.[1] There is no evidence of benefit of one agent, within this family, over another.[2]
Common side effects include headache.[1] Other side effects may include allergic reactions, low blood pressure, QT prolongation, and priapism.[2] A lower dose is recommended in those with moderate liver problems.[2] Use with nitrovasodilators is not recommended.[2] It is a PDE5 inhibitor and works by decreasing the breakdown of cyclic guanosine monophosphate (cGMP).[1]
Vardenafil was approved for medical use in Europe and the United States in 2003.[1][2] It is available as a generic medication.[3] In the United Kingdom it costs about 5 pounds per 10 mg dose.[3] In the United States this amount costs about 17 USD as of 2021.[4]
Medical use
Vardenafil's indications and contraindications are the same as with other PDE5 inhibitors; it is closely related in function to sildenafil citrate (Viagra) and tadalafil (Cialis). The difference between the vardenafil molecule and sildenafil citrate is a nitrogen atom's position and the change of sildenafil's piperazine ring methyl group to an ethyl group. Tadalafil is structurally different from both sildenafil and vardenafil. Vardenafil's relatively short effective time is comparable to but somewhat longer than sildenafil's.
Beyond its indications for erectile dysfunction, vardenafil may be effective in the treatment of premature ejaculation, where it may significantly increase the time from penetration to ejaculation.[5]
Dosage
The typical dose is 10 mg, though 5 to 20 mg may be used.[1]
Side effects
The common, adverse drug reactions (side effects) are the same as with other PDE5 inhibitors. The frequent vardenafil-specific side-effect is nausea; the infrequent side effects are abdominal pain, back pain, photosensitivity, abnormal vision, eye pain, facial edema, hypotension, palpitation, tachycardia, arthralgia, myalgia, rash, itch, and priapism.
One possibly serious, but rare, side effect with vardenafil is heart attack. Also, in rare cases, vardenafil use may cause priapism, a very painful emergency condition that can cause impotence if left untreated.[6]
On 18 October 2007, the U.S. Food and Drug Administration (FDA) announced that a warning about possible deafness (sudden hearing loss) would be added to the drug labels of vardenafil, and other PDE5 inhibitors.[7]
Interactions
Vardenafil, as with all PDE5 inhibitors, should not be used by people taking nitrate medications, because combining them with vardenafil might provoke potentially life-threatening hypotension (low blood pressure).[8][9][10]
Further, vardenafil causes lengthening of the QT interval. Therefore, it should not be taken by people taking other medications that affect the QT interval (such as amiodarone).[11][12]
History
Vardenafil was co-marketed by Bayer Pharmaceuticals, GlaxoSmithKline, and Schering-Plough under the trade name Levitra. As of 2005, the co-promotion rights of GSK on Levitra have been returned to Bayer in many markets outside the U.S. In Italy, Bayer sells vardenafil as Levitra and GSK sells it as Vivanza. Thus, because of European Union trade rules, parallel imports might result in Vivanza sold next to Levitra in the EU.
An orally disintegrating form, marketed as Staxyn and Levitra Soft, has been gaining approvals in countries such as the United States[13] and Canada.[14]
Society and culture
Dose forms

It is available in 2.5 mg, 5 mg, 10 mg, and 20 mg doses in round orange tablets. The normal starting dose is 10 mg[15] (roughly equivalent to 50 mg of sildenafil). Vardenafil should be taken 1 to 2 hours prior to sexual activity, with a maximum dose frequency of once per day. In some territories, such as the UK, only certain doses may be available.
Vardenafil is also available under the name Staxyn as a tablet which dissolves on the tongue rather than being swallowed in the form of a pill.[16]
Tainted supplements
The USFDA has found vardenafil and other synthetic PDE5 inhibitors in numerous products marketed as "herbal" supplements or "all natural" products for male enhancement.[17][18]
Notes
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 "Levitra". Archived from the original on 4 March 2021. Retrieved 13 September 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Vardenafil Monograph for Professionals". Drugs.com. Archived from the original on 6 May 2021. Retrieved 13 September 2021.
- ↑ 3.0 3.1 BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 862. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ "Vardenafil Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 13 May 2021. Retrieved 13 September 2021.
- ↑ Aversa A, Pili M, Francomano D, Bruzziches R, Spera E, La Pera G, Spera G (July 2009). "Effects of vardenafil administration on intravaginal ejaculatory latency time in men with lifelong premature ejaculation". International Journal of Impotence Research. 21 (4): 221–7. doi:10.1038/ijir.2009.21. PMID 19474796.
- ↑ Schools of Pharmacy (Glen L. Stimmel, Pharm.D., and Mary A. Gutierrez, Pharm.D.) and Medicine (Glen L. Stimmel, Pharm.D.), University of Southern California, Los Angeles, California. "Counseling Patients About Sexual Issues: Drug-Induced Priapism". Medscape. Archived from the original on 2015-05-11. Retrieved 2010-12-06.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ↑ "FDA Announces Revisions to Labels for Cialis, Levitra and Viagra". Food and Drug Administration. 2007-10-18. Archived from the original on 2016-10-22. Retrieved 2009-08-06.
- ↑ Kloner RA (December 2005). "Pharmacology and drug interaction effects of the phosphodiesterase 5 inhibitors: focus on alpha-blocker interactions". The American Journal of Cardiology. 96 (12B): 42M–46M. doi:10.1016/j.amjcard.2005.07.011. PMID 16387566.
- ↑ Carson CC (February 2006). "PDE5 inhibitors: are there differences?". The Canadian Journal of Urology. 13 Suppl 1: 34–9. PMID 16526979.
- ↑ "Levitra for Treatment of Erectile Dysfunction". www.erectiledysfunction.com. Archived from the original on 2017-10-27. Retrieved 2017-10-27.
- ↑ Lepor H, Lepor NE, Hill LA, Trohman RG (2008). "The QT Interval and Selection of Alpha-Blockers for Benign Prostatic Hyperplasia". Reviews in Urology. 10 (2): 85–91. PMC 2483321. PMID 18660858.
- ↑ Learning, Jones & Bartlett (2010-06-29). 2011 Nurse's Drug Handbook. Jones & Bartlett Publishers. ISBN 9781449653729. Archived from the original on 2021-08-27. Retrieved 2021-06-27.
- ↑ "New erectile dysfunction treatment STAXYN™ approved in the U.S. - Pharmaceutical Processing". pharmpro.com. 2010-06-21. Archived from the original on 2012-04-06.
- ↑ "STAXYN® - New Innovation in Erectile Dysfunction Helps Younger Men Rise to the Occasion". Archived from the original on 2014-11-08. Retrieved 2021-06-27.
- ↑ "Levitra Dosage". Drugs.com. Archived from the original on 2018-02-23. Retrieved 2018-02-23.
- ↑ "Vardenafil". Medicine Net. Archived from the original on 2017-10-07. Retrieved 2014-02-18.
- ↑ "Tainted Products Marketed as Dietary Supplements". United States Food and Drug Administration. Archived from the original on 2021-04-13. Retrieved 2021-06-27.
- ↑ Tucker J, Fischer T, Upjohn L, Mazzera D, Kumar M (October 2018). "Unapproved Pharmaceutical Ingredients Included in Dietary Supplements Associated With US Food and Drug Administration Warnings". JAMA Network Open. 1 (6): e183337. doi:10.1001/jamanetworkopen.2018.3337. PMC 6324457. PMID 30646238.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 maint: date format
- CS1 maint: multiple names: authors list
- CS1: long volume value
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Erectile dysfunction drugs
- Lactams
- PDE5 inhibitors
- Schering-Plough brands
- GlaxoSmithKline brands
- Merck & Co. brands
- Bayer brands
- Phenol ethers
- Piperazines
- Sulfonamides
- RTT