Anagrelide
 | |
| Names | |
|---|---|
| Trade names | Agrylin, Xagrid, others |
| |
| Clinical data | |
| Drug class | Antithrombotic[1] |
| Main uses | High platelets in essential thrombocytosis[2] |
| Side effects | Headache, palpitations, diarrhea, weakness, swelling, nausea, shortness of breath, itchiness, heart burn[2] |
| Pregnancy category |
|
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601020 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Metabolism | Liver, partially through CYP1A2 |
| Elimination half-life | 1.3 hours |
| Excretion | Urine (<1%) |
| Chemical and physical data | |
| Formula | C10H7Cl2N3O |
| Molar mass | 256.09 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Anagrelide, sold under the brand name Agrylin among others, is a medication used to treat high platelets in essential thrombocytosis or other myeloproliferative disorders.[2] It is used when other treatments are not effective or tolerated.[3] It is taken by mouth.[3]
Common side effects may include headache, palpitations, diarrhea, weakness, swelling, nausea, shortness of breath, itchiness, and heart burn.[2] Other side effects may include arrhythmias, QT prolongation, bleeding, and interstitial nephritis.[1] Safety in pregnancy is unclear, with evidence of harm in other animals.[2] It is an antithrombotic.[1] Once started, suddenly stopping anagrelide may cause a rebound rise in platelets and increase risk of blood clots.[3] Monitoring of the platelet count is recommended with any withdrawal of the drug.[3]
Anagrelide was approved for medical use in the United States in 1997 and Europe in 2004.[4][5] It is available as a generic medication.[3] In the United Kingdom 100 pills of 500 micrograms costs the NHS about £400 as of 2021.[3] This amount in the United States costs about 52 USD.[6]
Medical uses
Anagrelide is used to treat essential thrombocytosis, especially when the current treatment of the patient is insufficient.[7] Essential thrombocytosis patients who are suitable for anagrelide often meet one or more of the following factors:[8][9]
- age over 60 years
- platelet count over 1000×109/L
- a history of thrombosis
According to a 2005 trial, the combination of hydroxyurea with aspirin is superior to the combination of anagrelide and aspirin for the initial management of essential thrombocytosis. The hydroxyurea arm had a lower likelihood of myelofibrosis, arterial thrombosis, and bleeding, but it had a slightly higher rate of venous thrombosis.[7] Anagrelide can be useful in times when hydroxyurea proves ineffective.
Dosage
It is generally started at a dose of 500 micrograms twice per day.[3] This is increased to a typical dose of 500 micrograms to 1,500 micrograms twice per day.[3] Doses up to 10,000 micrograms per day may be used.[3]
Side effects
Common side effects are headache, diarrhea, unusual weakness/fatigue, hair loss, nausea.[3]
The same MRC trial mentioned above also analyzed the effects of anagrelide on bone marrow fibrosis, a common feature in patients with myelofibrosis. The use of anagrelide was associated with a rapid increase in the degree of reticulin deposition (the mechanism by which fibrosis occurs), when compared to those in whom hydroxyurea was used. Patients with myeloproliferative conditions are known to have a very slow and somewhat variable course of marrow fibrosis increase. This trend may be accelerated by anagrelide. This increase in fibrosis appeared to be linked to a drop in hemoglobin as it progressed. Stopping anagrelide (and switching patients to hydroxyurea) appeared to reverse the degree of marrow fibrosis. Thus, patients on anagrelide may need to be monitored on a periodic basis for marrow reticulin scores, especially if anemia develops, or becomes more pronounced if present initially.[10]
Less common side effects include: congestive heart failure, myocardial infarction, cardiomyopathy, cardiomegaly, complete heart block, atrial fibrillation, cerebrovascular accident, pericarditis, pulmonary infiltrates, pulmonary fibrosis, pulmonary hypertension, pancreatitis, gastric/duodenal ulceration, renal impairment/failure and seizure.
Due to these issues, anagrelide should not generally be considered for first line therapy for essential thrombocytosis.
Mechanism of action
Anagrelide works by inhibiting the maturation of platelets from megakaryocytes.[11] The exact mechanism of action is unclear, although it is known to be a phosphodiesterase inhibitor.[12] It is a potent (IC50 = 36nM) inhibitor of phosphodiesterase-II.[citation needed] It inhibits PDE-3 and phospholipase A2.[13]
Synthesis
Phosphodiesterase inhibitor with antiplatelet activity.
| Synthesis 1[14][15] | Synthesis 2 |
|---|---|
 |

|
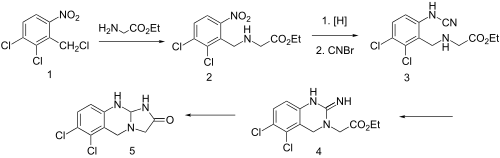
Condensation of benzyl chloride 1 with ethyl ester of glycine gives alkylated product 2. Reduction of the nitro group leads to the aniline and reaction of this with cyanogen bromide possibly gives cyanamide 3 as the initial intermediate. Addition of the aliphatic would then lead to formation of the quinazoline ring (4). Amide formation between the newly formed imide and the ester would then serve to form the imidazolone ring, whatever the details of the sequence, there is obtained anagrelide (5).
Research
Anagrelide controlled release (GALE-401) is in phase III clinical trials by Galena Biopharma for the treatment of essential thrombocytosis.[16]
References
- ↑ 1.0 1.1 1.2 "Anagrelide". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Retrieved 14 January 2022.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Anagrelide Monograph for Professionals". Drugs.com. Archived from the original on 25 February 2021. Retrieved 14 January 2022.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 "9. Blood and nutrition: platelet disorders". BNF 85: March-September 2023. BMJ Group and the Pharmaceutical Press. 2023. p. 1140. ISBN 978-0857114587.
- ↑ "DailyMed - ANAGRELIDE capsule". dailymed.nlm.nih.gov. Archived from the original on 24 March 2021. Retrieved 14 January 2022.
- ↑ "Xagrid". Archived from the original on 6 May 2021. Retrieved 14 January 2022.
- ↑ "Anagrelide Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 14 January 2022.
- ↑ 7.0 7.1 Harrison CN, Campbell PJ, Buck G, et al. (July 2005). "Hydroxyurea compared with anagrelide in high-risk essential thrombocythemia". N. Engl. J. Med. 353 (1): 33–45. doi:10.1056/NEJMoa043800. PMID 16000354.
- ↑ Reilly, John T. (1 February 2009). "Anagrelide for the treatment of essential thrombocythemia: a survey among European hematologists/oncologists". Hematology. 14 (1): 1–10. doi:10.1179/102453309X385115. PMID 19154658. S2CID 26257327.
- ↑ Brière, Jean B (1 January 2007). "Essential thrombocythemia". Orphanet Journal of Rare Diseases. 2 (1): 3. doi:10.1186/1750-1172-2-3. PMC 1781427. PMID 17210076.
- ↑ Campbell PJ, Bareford D, Erber WN, et al. (June 2009). "Reticulin accumulation in essential thrombocythemia: prognostic significance and relationship to therapy". J. Clin. Oncol. 27 (18): 2991–9. doi:10.1200/JCO.2008.20.3174. PMC 3398138. PMID 19364963.
- ↑ Petrides PE (2006). "Anagrelide: what was new in 2004 and 2005?". Semin. Thromb. Hemost. 32 (4 Pt 2): 399–408. doi:10.1055/s-2006-942760. PMID 16810615.
- ↑ Jones GH, Venuti MC, Alvarez R, Bruno JJ, Berks AH, Prince A (February 1987). "Inhibitors of cyclic AMP phosphodiesterase. 1. Analogues of cilostamide and anagrelide". J. Med. Chem. 30 (2): 295–303. doi:10.1021/jm00385a011. PMID 3027338.
- ↑ Harrison CN, Bareford D, Butt N, et al. (May 2010). "Guideline for investigation and management of adults and children presenting with a thrombocytosis". Br. J. Haematol. 149 (3): 352–75. doi:10.1111/j.1365-2141.2010.08122.x. PMID 20331456. S2CID 2301197.
- ↑ W. N. Beverung, A. Partyka, U.S. Patent 3,932,407; 31617 US Re. 31617; T. A. Jenks et al., U.S. Patent 4,146,718 (1976, 1984, 1979 all to Bristol-Myers).
- ↑ Yamaguchi, Hitoshi; Ishikawa, Fumiyoshi (1981). "Synthesis and reactions of 2-chloro-3,4-dihydrothienopyrimidines and -quinazolines". Journal of Heterocyclic Chemistry. 18: 67–70. doi:10.1002/jhet.5570180114.
- ↑ Inc., Galena Biopharma (2016-12-28). "Galena Biopharma Confirms Regulatory Pathway for GALE-401 (Anagrelide Controlled Release)". globenewswire.com. Archived from the original on 2020-10-27. Retrieved 2021-09-28.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- All articles with unsourced statements
- Articles with unsourced statements from December 2008
- Articles with invalid date parameter in template
- Drugs with unknown mechanisms of action
- Orphan drugs
- Antineoplastic drugs
- Phosphodiesterase inhibitors
- Imidazoquinazolines
- Lactams
- Chloroarenes
- Takeda Pharmaceutical Company brands
- RTT