Bethanechol
 | |
| Names | |
|---|---|
| Trade names | Urecholine, Myotonine, others[1] |
| Other names | Bethanechol chloride, 2-[(aminocarbonyl)oxy]- N,N,N-trimethyl- 1-propanaminium Carbamyl-β-methylcholine |
| |
| Clinical data | |
| Drug class | Muscarinic agonist[1] |
| Main uses | Urinary retention, heartburn[2][3] |
| Side effects | Crampy abdominal pain, flushing, sweating, nausea, diarrhea[3] |
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682849 |
| Legal | |
| Legal status |
|
| Chemical and physical data | |
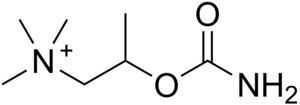
| Formula | C7H17N2O2 |
| Molar mass | 161.225 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Bethanechol, sold under the brand name Urecholine, is a medication used to treat urinary retention and heartburn.[2][3] Specifically it is used for urinary retention due to a lack of contraction of the urinary bladder.[3] It is taken by mouth, half an hour before food, 3-4 times daily.[2]
Common side effects include crampy abdominal pain, flushing, sweating, nausea, and diarrhea.[3] It should not be used in people with a very slow heart rate, epilepsy, stomach ulcer, intestinal obstruction, parkinsonism, recent heart attack, COPD, or overactive thyroid.[2] Its effects in pregnancy are not known.[2] It is a parasympathomimetic that directly stimulates muscarinic receptors without any effect on nicotinic receptors.[5]
Bethanechol was approved for medical use in the United States in 1948.[3] In the United Kingdom, one month costs the NHS up to £30 as of 2022.[2] This amount in the United States is about 25 USD.[6] It is not often used, and in the United Kingdom is deemed less suitable.[5][2]
Medical uses
Bethanechol is used to treat urinary retention.[1][2]
It is used to treat urinary retention resulting from general anesthetic, diabetic neuropathy of the bladder, or a side effect of antidepressants; or to treat gastrointestinal lack of muscular tone. The muscarinic receptors in the bladder and gastrointestinal tract stimulate contraction of the bladder and expulsion of urine, and increased gastrointestinal motility, respectively. Bethanechol should be used to treat these disorders only after mechanical obstruction is ruled out as a possible cause.
Its potential benefit in the treatment of cerebral palsy has been investigated.[7]
Atropine is given preoperatively to prevent voiding of the bowel/bladder during surgery, Bethanechol is then given postoperatively to revert this action.[8]
Dose
The dose is around 10-25 mg 3-4 times a day by mouth on an empty stomach.[2]
Contraindications
It is contraindicated in people with bradycardia, epilepsy, peptic ulcer, intestinal obstruction, parkinsonism, recent myocardial infarction, obstructive airways disease and hyperthyroidism.[2] The parasympathomimetic action of this drug will exacerbate the symptoms of these disorders.
Side effects
Side effects include stomach upset, excessive sweating, and feeling sick.[2] Its effects in pregnancy are not known.[2]
Mechanism
It is a parasympathomimetic that selectively stimulates muscarinic receptors without any effect on nicotinic receptors.[5] Unlike acetylcholine, bethanechol is not hydrolyzed by cholinesterase and will therefore have a long duration of action.[citation needed]
Society and culture
Cost
In the UK, one month treatment of urinary retention with bethanechol can cost the NHS up to £30.[2]
Names
Bethanechol is sold under the brand names Duvoid (Roberts), Myotonachol (Glenwood), Urecholine (Merck Frosst) and Urocarb (Hamilton). The name bethanechol refers to its structure as the urethane of beta-methylcholine.
References
- ↑ 1.0 1.1 1.2 Lehne, Richard A.; Rosenthal, Laura (2014). "IV. Peripheral nervous system drugs". Pharmacology for Nursing Care (8th ed.). Elsevier. pp. 124–125. ISBN 978-0-323-29354-9. Archived from the original on 20 November 2021. Retrieved 20 November 2021.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 "7. Genito-urinary system". British National Formulary (BNF) (82 ed.). London: BMJ Group and the Pharmaceutical Press. September 2021 – March 2022. p. 833. ISBN 978-0-85711-413-6.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "Bethanechol Monograph for Professionals". Drugs.com. Archived from the original on 19 January 2021. Retrieved 10 January 2022.
- ↑ Bethanechol Archived 4 November 2021 at the Wayback Machine. List of nationally authorised medicinal products. European Medicines Agency. 2021
- ↑ 5.0 5.1 5.2 Ritter, James M.; Flower, Rod; Henderson, Graeme; Loke, Yoon Kong; Rang, Humphrey P. (2020). "14. Cholinergic transmission". Rang & Dale's Pharmacology (9th ed.). Elsevier. pp. 181–184. ISBN 978-0-7020-7448-6. Archived from the original on 28 August 2021. Retrieved 20 November 2021. Cite error: Invalid
<ref>tag; name "Rang2020" defined multiple times with different content - ↑ "Bethanechol Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 10 January 2022.
- ↑ Carter WJ (September 2008). "Unexpected benefits of bethanechol in adults with cerebral palsy". The Medical Journal of Australia. 189 (5): 293. doi:10.5694/j.1326-5377.2008.tb02034.x. PMID 18759732. S2CID 29048832. Archived from the original on 18 February 2012. Retrieved 8 March 2021.
- ↑ Obied H (2011). Cholinergic Pharmacology. CSU.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Pages with reference errors
- Webarchive template wayback links
- Use dmy dates from September 2017
- Articles with invalid date parameter in template
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements
- Quaternary ammonium compounds
- Choline esters
- Carbamates
- Muscarinic agonists
- Peripherally selective drugs
- RTT