Ustekinumab
 Ustekinumab injection | |
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | IL-12 and IL-23 |
| Names | |
| Trade names | Stelara |
| Other names | CNTO 1275 |
| Clinical data | |
| Drug class | Monoclonal antibody |
| Main uses | Crohn's disease, ulcerative colitis, plaque psoriasis, psoriatic arthritis[1] |
| Side effects | Joint pain, back pain, diarrhea, infections[2] |
| Pregnancy category |
|
| Routes of use | Subcutaneous, intravenous |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a611013 |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Metabolism | unknown[5] |
| Elimination half-life | 15–32 days (average 3 weeks)[5] |
| Chemical and physical data | |
| Formula | C6482H10004N1712O2016S46 |
| Molar mass | 145648.06 g·mol−1 |
Ustekinumab, sold under the brand name Stelara, is a medication used to treat Crohn's disease, ulcerative colitis, plaque psoriasis, and psoriatic arthritis.[1] It is generally only used if other treatments have not been effective.[2] It is given by intravenous infusion or subcutaneous injection.[2]
Common side effects include joint pain, back pain, diarrhea, and infections.[2] Other side effects may include anaphylaxis, cancer, and reversible posterior leukoencephalopathy syndrome.[1] It is a monoclonal antibody that targets IL-12 and IL-23.[1] It is classified as a disease-modifying antirheumatic drug (DMARD).[1]
Ustekinumab was approved for medical use in Europe and the United States in 2009.[4][1] In the United Kingdom the 45 mg and 90 mg doses cost the NHS £2,150 each as of 2021.[2] In the United States it is about 12,500 USD per 45 mg.[7]
Medical uses
Ustekinumab is used to treat psoriasis.[6] This includes psoriatic arthritis when it affects the skin.[8][6] It is indicated for the treatment of adult and adolescent patients (12 years and older) with moderate to severe plaque psoriasis (Ps) who are candidates for phototherapy or systemic therapy, and adults with active psoriatic arthritis (PsA) alone or in combination with methotrexate.[6][9] It is also used to treat moderately to severely active Crohn's disease and moderately to severely active ulcerative colitis.[6]
In the European Union, ustekinumab is approved to treat moderate to severe plaque psoriasis in adults and children above the age of six years whose condition has not improved with, or who cannot use, other systemic (whole-body) psoriasis treatments, such as ciclosporin, methotrexate or PUVA (psoralen ultraviolet A);[4] active psoriatic arthritis in adults;[4] moderately to severely active Crohn's disease in adults;[4] and moderately to severely active ulcerative colitis in adults.[4]
It was found not effective for multiple sclerosis.[10]
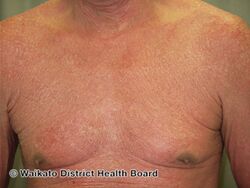
-
Erythrodermic psoriasis before ustekinumab
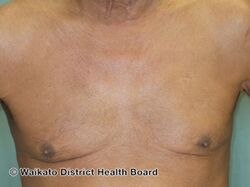
-
Erythrodermic psoriasis 2 weeks after first dose of ustekinumab
Dosage
In psoriasis a dose of 45 mg is given in those less than 100 kg, followed by further doses after a month and than every 3 months.[2] In those over 100 kg 90 mg is used per dose.[2]
In CD and UC an initial larger dose of 260 to 520 mg is given by infusion followed by 90 mg after 2 month and than 90 mg every 3 months.[2]
Side effects
According to information provided by Centocor, maker of one medication based on ustekinumab, their version of ustekinumab is associated with several types of serious adverse effects. These include an increased risk of infection, such as by tuberculosis and an increased risk of certain types of cancer. As with some other immunosuppressant drugs like ciclosporin, the brain swelling of posterior reversible encephalopathy syndrome is a risk. The pharmaceutical company also reports serious allergic reaction as a possible side effect. More common side effects are upper respiratory infection, headache, and tiredness.[11]
Clinical trials have shown that subcutaneous ustekinumab was generally well tolerated. Most treatment-emergent adverse events were of mild severity.[12]
Pregnancy
It is unknown if the medication is safe during pregnancy or breastfeeding.[5]
Mechanism of action

Ustekinumab is designed to interfere with the triggering of the body's inflammatory response through the suppression of certain cytokines. Specifically, it blocks interleukin IL-12 and IL-23 which help activate certain T-cells. It binds to the p-40 subunit of both IL-12 and IL-23 so that they subsequently cannot bind to their receptors.[14]
History
As of January 2007[update], there were five NIH-listed research studies involving CNTO 1275 on a multinational basis, including three Phase II and two Phase III trials. Three studies were focused on patients with psoriasis, one on psoriatic arthritis, and one on multiple sclerosis.[citation needed]
On December 4, 2007, a Biologic License Application (BLA) with the US Food and Drug Administration (FDA) was filed by Centocor and Janssen-Cilag International (collaborator) submitted a Marketing Authorization Application (MAA) to the European Medicines Agency (EMA). On November 21, 2008, the Committee for Medicinal Products for Human Use (CHMP) of the EMA adopted a positive opinion for ustekinumab for the treatment of moderate to severe plaque psoriasis in adults who failed to respond to other systemic therapies.[4][15]
Society and culture
Approval
Since 2009, ustekinumab is approved in Canada, the European Union, and the United States to treat moderate to severe plaque psoriasis.[16] On September 24, 2013, the US Food and Drug Administration (FDA) approved the use of ustekinumab for the treatment of psoriatic arthritis.[citation needed]
On December 12, 2008, Health Canada approved the use of ustekinumab for the treatment of chronic moderate to severe plaque psoriasis in adults who are candidates for phototherapy or systemic therapy.[17]
The FDA approved ustekinumab on September 25, 2009, for the treatment of adults with moderate to severe plaque psoriasis.[18][19]
The FDA approved ustekinumab in September 2016, for the treatment of Crohn's disease.[20]
Since September 2017, ustekinumab is available on the AU Pharmaceutical Benefits Scheme for the treatment of severe Crohn's disease in adults.[21]
In 2019, the European Commission approved the use of ustekinumab for adults with moderately to severely active ulcerative colitis.[22][4]
In October 2019, the FDA approved the use of ustekinumab for adults with moderately to severely active ulcerative colitis.[23][24]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 "Ustekinumab Monograph for Professionals". Drugs.com. Archived from the original on 2 February 2017. Retrieved 13 September 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 1165. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ "Ustekinumab (Stelara) Use During Pregnancy". Drugs.com. 26 November 2019. Archived from the original on 22 May 2020. Retrieved 6 April 2020.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 "Stelara EPAR". European Medicines Agency (EMA). Archived from the original on 6 April 2020. Retrieved 6 April 2020.
- ↑ 5.0 5.1 5.2 5.3 "Stelara 45 mg solution for injection (vials) - Summary of Product Characteristics (SmPC)". (emc). 27 February 2020. Archived from the original on 6 April 2020. Retrieved 6 April 2020.
- ↑ 6.0 6.1 6.2 6.3 6.4 "Stelara- ustekinumab injection, solution Stelara- ustekinumab solution". DailyMed. 24 March 2020. Archived from the original on 2 July 2019. Retrieved 6 April 2020.
- ↑ "Stelara Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 22 January 2021. Retrieved 13 September 2021.
- ↑ "Ustekinumab". The American Society of Health-System Pharmacists. Archived from the original on 2 February 2017. Retrieved 8 January 2017.
- ↑ Toussirot E (April 2012). "The IL23/Th17 pathway as a therapeutic target in chronic inflammatory diseases". Inflamm Allergy Drug Targets. 11 (2): 159–68. doi:10.2174/187152812800392805. PMID 22280236.
- ↑ Segal BM, Constantinescu CS, Raychaudhuri A, Kim L, Fidelus-Gort R, Kasper LH (September 2008). "Repeated subcutaneous injections of IL12/23 p40 neutralising antibody, ustekinumab, in patients with relapsing-remitting multiple sclerosis: a phase II, double-blind, placebo-controlled, randomised, dose-ranging study". Lancet Neurol. 7 (9): 796–804. doi:10.1016/S1474-4422(08)70173-X. PMID 18703004. S2CID 20290673.
- ↑ "Important Safety Information". Stelara (ustekinumab). Janssen Biotech. Archived from the original on 2021-03-29. Retrieved 2021-06-15.
- ↑ Weber J, Keam SJ (2009). "Ustekinumab". BioDrugs. 23 (1): 53–61. doi:10.2165/00063030-200923010-00006. PMID 19344192.
- ↑ Koutruba, Nora; Emer, Jason; Lebwohl, Mark (15 April 2010). "Review of ustekinumab, an interleukin-12 and interleukin-23 inhibitor used for the treatment of plaque psoriasis". Therapeutics and Clinical Risk Management. 6: 123–141. doi:10.2147/tcrm.s5599. ISSN 1178-203X.
- ↑ Koutruba, N; Emer, J; Lebwohl, M (15 April 2010). "Review of ustekinumab, an interleukin-12 and interleukin-23 inhibitor used for the treatment of plaque psoriasis". Therapeutics and Clinical Risk Management. 6: 123–41. doi:10.2147/tcrm.s5599. PMC 2857612. PMID 20421912.
- ↑ "FDA Issues Complete Response Letter to Centocor for Ustekinumab Biologic License Application". Drugs.com (Press release). Centocor. 25 September 2009. Archived from the original on 6 April 2020. Retrieved 6 April 2020.
- ↑ "Medarex to Receive Milestone Payment for Approval of Stelara (Ustekinumab) for the Treatment of Moderate to Severe Plaque Psoriasis". Archived from the original on 2013-08-01. Retrieved 2021-06-15.
- ↑ "drugbank.ca". Archived from the original on 31 January 2020. Retrieved 4 October 2019.
- ↑ "Stelara approved for moderate to severe psoriasis. Sept 2009". Archived from the original on 2015-09-20. Retrieved 2021-06-15.
- ↑ "Drug Approval Package: Stelara (Ustekinumab) Injection NDA #125261". U.S. Food and Drug Administration (FDA). 24 December 1999. Archived from the original on 6 April 2020. Retrieved 6 April 2020.
- ↑ "Stelara (ustekinumab) Injection NDA #761044". U.S. Food and Drug Administration (FDA). 12 January 2018. Archived from the original on 6 April 2020. Retrieved 6 April 2020.
- ↑ "12 Sept 2017". Archived from the original on 2021-03-15. Retrieved 2021-06-15.
- ↑ "EC approves expanded use of Stelara for moderately to severely active ulcerative colitis". Healio.com. Archived from the original on 9 September 2019. Retrieved 26 September 2019.
- ↑ "Janssen's Stelara Gets FDA Approval for Ulcerative Colitis". FDA News. WCG. Archived from the original on January 13, 2020. Retrieved October 31, 2019.
- ↑ "Stelara: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 20 October 2020. Retrieved 6 April 2020.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 maint: date format
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Drugs that are a monoclonal antibody
- Articles containing potentially dated statements from January 2007
- Articles with invalid date parameter in template
- All articles containing potentially dated statements
- All articles with unsourced statements
- Articles with unsourced statements from August 2020
- Articles with changed CASNo identifier
- Chemicals that do not have a ChemSpider ID assigned
- Articles with changed EBI identifier
- Immunosuppressants
- Johnson & Johnson brands
- Janssen Biotech
- Monoclonal antibodies
- RTT