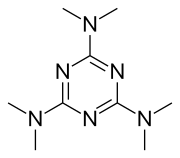
Altretamine
 | |
 | |
| Names | |
|---|---|
| Pronunciation | al tret' a meen[1] |
| Trade names | Hexalen |
| Other names | Hexamethylmelamine, 2,4,6-Tris(dimethylamino)-1,3,5-triazine |
| |
| Clinical data | |
| Drug class | Alkylating agent[1] |
| Main uses | Ovarian cancer[1] |
| Side effects | Nausea, vomiting, diarrhea, hair loss, bone marrow suppression, peripheral nerve problems, rash[1] |
| Pregnancy category |
|
| Routes of use | By mouth (capsules) |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601200 |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Protein binding | 94% |
| Metabolism | Extensive liver |
| Metabolites | Pentamethylmelamine, tetramethylmelamine |
| Elimination half-life | 4.7–10.2 hours |
| Chemical and physical data | |
| Formula | C9H18N6 |
| Molar mass | 210.285 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Altretamine, sold under the brand name Hexalen, is a medication used to treat ovarian cancer.[1] Specifically it is used for advanced disease when other treatments are not effective.[1] It is taken by mouth.[1]
Common side effects include nausea, vomiting, diarrhea, hair loss, bone marrow suppression, peripheral nerve problems, and rash.[1] Other side effects may include mood disorders and further cancer.[2] Use in pregnancy may harm the baby.[2] It is an alkylating agent.[1]
Altretamine was approved for medical use in the United States in 1990.[1] As of 2022 it is not commercially available in the United States.[3]
Medical uses
It is indicated for use as a single agent in the palliative treatment of people with persistent or recurrent ovarian cancer following first-line therapy with cisplatin and/or alkylating agent-based combination.[4]
It is not considered a first-line treatment,[5] but it can be useful as salvage therapy.[6] It also has the advantage of being less toxic than other drugs used for treating refractory ovarian cancer.[7]
Side effects
Side effects include nausea, vomiting, anemia and peripheral sensory neuropathy.[8]
Interactions
Combination with pyridoxine (vitamin B6) decreases neurotoxicity but has been found to reduce the effectiveness of an altretamine/cisplatin regime.[9] MAO inhibitor can cause severe orthostatic hypotension when combined with altretamine; and cimetidine can increase its elimination half-life and toxicity.[8]
Mechanism
The precise mechanism by which altretamine exerts its anti-cancer effect is unknown but it is classified by MeSH as an alkylating antineoplastic agent.[10]
This unique structure is believed to damage tumor cells through the production of the weakly alkylating species formaldehyde, a product of CYP450-mediated N-demethylation. Administered orally, altretamine is extensively metabolized on first pass, producing primarily mono- and didemethylated metabolites. Additional demethylation reactions occur in tumor cells, releasing formaldehyde in situ before the drug is excreted in the urine. The carbinolamine (methylol) intermediates of CYP450-mediated metabolism also can generate electrophilic iminium species that are capable of reacting covalently with DNA guanine and cytosine residues as well as protein. Iminium-mediated DNA cross-linking and DNA-protein interstrand cross-linking, mediated through both the iminium intermediate and formaldehyde, have been demonstrated, although the significance of DNA cross-linking on altretamine antitumor activity is uncertain.[11]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 "Altretamine". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 6 May 2021. Retrieved 14 January 2022.
- ↑ 2.0 2.1 "Altretamine Monograph for Professionals". Drugs.com. Archived from the original on 26 January 2021. Retrieved 14 January 2022.
- ↑ "Drugs@FDA: FDA-Approved Drugs". www.accessdata.fda.gov. Archived from the original on 27 August 2021. Retrieved 14 January 2022.
- ↑ "Hexalen (altretamine) Capsule. Human Prescription Drug Label". dailymed.nlm.nih.gov. Eisai Inc. Archived from the original on 28 August 2016. Retrieved 24 August 2016.
- ↑ Keldsen N, Havsteen H, Vergote I, Bertelsen K, Jakobsen A (2003). "Altretamine (hexamethylmelamine) in the treatment of platinum-resistant ovarian cancer: a phase II study". Gynecol. Oncol. 88 (2): 118–22. doi:10.1016/S0090-8258(02)00103-8. PMID 12586589.
- ↑ Chan JK, Loizzi V, Manetta A, Berman ML (2004). "Oral altretamine used as salvage therapy in recurrent ovarian cancer". Gynecol. Oncol. 92 (1): 368–71. doi:10.1016/j.ygyno.2003.09.017. PMID 14751188.
- ↑ Malik IA (2001). "Altretamine is an effective palliative therapy of patients with recurrent epithelial ovarian cancer". Jpn. J. Clin. Oncol. 31 (2): 69–73. doi:10.1093/jjco/hye012. PMID 11302345.
- ↑ 8.0 8.1 Drugs.com: Altretamine Monograph Archived 26 January 2021 at the Wayback Machine
- ↑ Wiernik, P. H.; Yeap, B.; Vogl, S. E.; Kaplan, B. H.; Comis, R. L.; Falkson, G.; Davis, T. E.; Fazzini, E.; Cheuvart, B.; Horton, J. (1992). "Hexamethylmelamine and low or moderate dose cisplatin with or without pyridoxine for treatment of advanced ovarian carcinoma: A study of the Eastern Cooperative Oncology Group". Cancer Investigation. 10 (1): 1–9. doi:10.3109/07357909209032783. PMID 1735009.
- ↑ Damia G, D'Incalci M (1995). "Clinical pharmacokinetics of altretamine". Clinical Pharmacokinetics. 28 (6): 439–48. doi:10.2165/00003088-199528060-00002. PMID 7656502.
- ↑ Lemke, Thomas L.; Williams, David A., eds. (2008). Foye's Principles of Medicinal Chemistry (6th ed.). Philadelphia: Lippincott Williams & Wilkins. ISBN 978-0-7817-6879-5.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Use dmy dates from April 2017
- Articles with invalid date parameter in template
- DNA replication inhibitors
- Orphan drugs
- Triazines
- RTT