Tretinoin
 | |
| Names | |
|---|---|
| Pronunciation | See pronunciation note |
| Trade names | Vesanoid, Avita, Renova, others |
| |
| Clinical data | |
| Pregnancy category | |
| Routes of use | Topical, by mouth |
| Defined daily dose | Not established[3] |
| External links | |
| AHFS/Drugs.com | Systemic: Monograph Topical: Monograph |
| MedlinePlus | a608032 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Protein binding | > 95% |
| Elimination half-life | 0.5-2 hours |
| Chemical and physical data | |
| Formula | C20H28O2 |
| Molar mass | 300.442 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 180 °C (356 °F) |
| |
| |
Tretinoin, also known as all-trans retinoic acid (ATRA), is medication used for the treatment of acne and acute promyelocytic leukemia.[4][5][6] For acne, it is applied to the skin as a cream or ointment.[6] For leukemia, it is taken by mouth for up to three months.[4]
Common side effects when used by mouth include shortness of breath, headache, numbness, depression, skin dryness, itchiness, hair loss, vomiting, muscle pains, and vision changes.[4] Other severe side effects include high white blood cell counts and blood clots.[4] When used as a cream, side effects include skin redness, peeling, and sun sensitivity.[6] Use during pregnancy is contraindicated due to the risk of birth defects.[4][1] It is in the retinoid family of medications.[5]
Tretinoin was patented in 1957, and approved for medical use in 1962.[7] It is on the World Health Organization's List of Essential Medicines.[8] Tretinoin is available as a generic medication.[9] In the United Kingdom the cream together with erythromycin costs the NHS about £7.05 per 25 mL while the pills are £1.61 per 10 mg.[6] In 2017, it was the 293rd most commonly prescribed medication in the United States, with more than one million prescriptions.[10][11]
Medical uses
Skin use
Topical tretinoin is used for the treatment of both noninflammatory and inflammatory acne. Tretinoin is most commonly used to treat acne.[12] Multiple studies support the efficacy of topical retinoids in the treatment of acne vulgaris.[13][14] It is sometimes used in conjunction with other acne treatments, as it enhances the penetration of other topical acne medications.[15] In addition to treating active acne, retinoids accelerate the resolution of acne-induced postinflammatory hyperpigmentation.[16] It is also useful as maintenance therapy for people who have responded to initial treatment of acne and as maintenance therapy can reduce the prolonged use of antibiotics for acne.[medical citation needed]
People with sensitive skin using the topical form should avoid using any cream or lotion that has a strong drying effect, contains alcohol, astringents, spices, lime, sulfur, resorcinol, or aspirin, as these may interact with tretinoin or exacerbate its side effects.[12] In topical form, this drug is pregnancy category C and should not be used by pregnant women.[12][2]
Leukemia
Tretinoin is used to induce remission in people with acute promyelocytic leukemia who have a mutation (the t(15;17) translocation 160 and/or the presence of the PML/RARα gene) and who don't respond to anthracyclines or can't take that class of drug. It is not used for maintenance therapy.[17][18][19]
By mouth, this drug is pregnancy category D and should not be used by pregnant women as it may harm the fetus.[17][1]
Evidence does not support a benefit over chemotherapy for survival.[20]
Photoaging
Topical tretinoin have been used for mild to moderate photoaging with some cosmetic success.[21]
Dosage
The defined daily dose is not established[3]
Side effects
Skin use
Topical tretinoin is only for use on skin and it should not be applied to eyes or mucosal tissues. Common side effects include skin irritation, redness, swelling, and blistering.[12] If irritation is a problem, a decrease in the frequency of application to every other or every third night can be considered, and the frequency of application can be increased as tolerance improves. The fine skin flaking that is often seen can be gently exfoliated with a washcloth. A noncomedogenic facial moisturizer can also be applied if needed. Delaying application of the retinoid for at least 20 minutes after washing and drying the face may also be helpful. Topical retinoids are not true photosensitizing drugs, but people using topical retinoids have described symptoms of increased sun sensitivity. This is thought to be due to thinning of the stratum corneum leading to a decreased barrier against ultraviolet light exposure, as well as an enhanced sensitivity due to the presence of cutaneous irritation.[22] The use of sun-protective clothing and/or sunscreen is recommended, particularly when prolonged sun exposure is anticipated.
Leukemia use
The oral form of the drug has boxed warnings concerning the risks of retinoic acid syndrome and leukocytosis.[17]
Other significant side effects include a risk of thrombosis, benign intracranial hypertension in children, high lipids (hypercholesterolemia and/or hypertriglyceridemia), and liver damage.[17]
There are many significant side effects from this drug that include malaise (66%), shivering (63%), hemorrhage (60%), infections (58%), peripheral edema (52%), pain (37%), chest discomfort (32%), edema (29%), disseminated intravascular coagulation (26%), weight increase (23%), injection site reactions (17%), anorexia (17%), weight decrease (17%), and myalgia (14%).[17]
Respiratory side effects usually signify retinoic acid syndrome, and include upper respiratory tract disorders (63%), dyspnea (60%), respiratory insufficiency (26%), pleural effusion (20%), pneumonia (14%), rales (14%), and expiratory wheezing (14%), and many others at less than 10%.[17]
Around 23% of people taking the drug have reported earache or a feeling of fullness in their ears.[17]
Gastrointestinal disorders include bleeding (34%), abdominal pain (31%), diarrhea (23%), constipation (17%), dyspepsia (14%), and swollen belly (11%) and many others at less than 10%.[17]
In the cardiovascular system, side effects include arrhythmias (23%), flushing (23%), hypotension (14%), hypertension (11%), phlebitis (11%), and cardiac failure (6%) and for 3% of patients: cardiac arrest, myocardial infarction, enlarged heart, heart murmur, ischemia, stroke, myocarditis, pericarditis, pulmonary hypertension, secondary cardiomyopathy.[17]
In the nervous system, side effects include dizziness (20%), paresthesias (17%), anxiety (17%), insomnia (14%), depression (14%), confusion (11%), and many others at less than 10% frequency.[17]
In the urinary system, side effects include chronic kidney disease (11%) and several others at less than 10% frequency.[17]
Mechanism of action
For its use in cancer, its mechanism of action is unknown, but on a cellular level, laboratory tests show that tretinoin forces APL cells to differentiate and stops them from proliferating; in people there is evidence that it forces the primary cancerous promyelocytes to differentiate into their final form, allowing normal cells to take over the bone marrow.[17] Recent study shows that ATRA inhibits and degrades active PIN1.[23]
For its use in acne, tretinoin (along with other retinoids) are vitamin A derivatives that act by binding to two nuclear receptor families within keratinocytes: the retinoic acid receptors (RAR) and the retinoid X receptors (RXR)[16] These events contribute to the normalization of follicular keratinization and decreased cohesiveness of keratinocytes, resulting in reduced follicular occlusion and microcomedone formation.[24] In other words, it decreases the ability of epithelial cells in hair follicles to stick together, leading to fewer blackheads and whiteheads; it also seems to make the epithelial cells divide faster, causing the blackheads to be pushed out. In addition to this direct effect of tretinoin on comedogenesis, it may also improve inflammatory acne through other mechanisms. The retinoid-receptor complex competes for coactivator proteins of AP-1, a key transcription factor involved in inflammation.[16] Retinoids also down-regulate expression of toll-like receptor (TLR)-2, which has been implicated in the inflammatory response in acne.[25] Moreover, tretinoin and retinoids may enhance the penetration of other topical acne medications.[26]
Biosynthesis
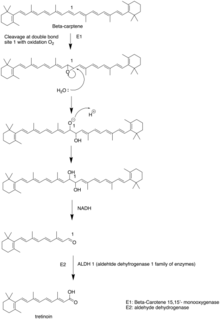
Tretinoin is synthesized from Beta-carotene. The Beta-carotene is firstly cleaved into Beta-carotene 15-15'-monooxygenase through site 1 double bond oxidized to epoxide. The epoxide is attacked by water to form diol in site 1. NADH, as a reduction agent, reduce the alcohol group to aldehydes.[27]
History
Tretinoin was co-developed for its use in acne by James Fulton and Albert Kligman when they were at University of Pennsylvania in the late 1960s.[28][29] The University of Pennsylvania held the patent for Retin-A, which it licensed to pharmaceutical companies.[29]
Etymology
The origin of the name tretinoin is uncertain,[30][31] although several sources agree (one with probability,[30] one with asserted certainty[32]) that it probably comes from trans- + retinoic [acid] + -in, which is plausible given that tretinoin is the all-trans isomer of retinoic acid. The name isotretinoin is the same root tretinoin plus the prefix iso-. Regarding pronunciation, the following variants apply equally to both tretinoin and isotretinoin. Given that retinoic is pronounced /ˌrɛtɪˈnoʊɪk/,[31][32][33][34] it is natural that /ˌtrɛtɪˈnoʊɪn/ is a commonly heard pronunciation. Dictionary transcriptions also include /ˌtrɪˈtɪnoʊɪn/ (tri-TIN-oh-in)[31][33] and /ˈtrɛtɪnɔɪn/.[32][34]
Research
Tretinoin has been explored as a treatment for hair loss, potentially as a way to increase the ability of minoxidil to penetrate the scalp, but the evidence is weak and contradictory.[35][36]
It has been used off-label to treat and reduce the appearance of stretch marks.[37] It has also been studied in skin aging.[38]
Society and culture
Cost
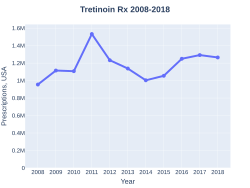
In the United Kingdom the cream together with erythromycin costs the NHS about £7.05 per 25 mL while the pills are £1.61 per 10 mg.[6] In 2017, it was the 293rd most commonly prescribed medication in the United States, with more than one million prescriptions.[10][11]
-
Tretinoin costs (US)
-
Tretinoin prescriptions (US)
See also
- Baldness treatments
- Hypervitaminosis A syndrome
- Talarozole, an experimental drug potentiating the effects of tretinoin
References
- ↑ 1.0 1.1 1.2 1.3 "Tretinoin (Vesanoid) Use During Pregnancy". Drugs.com. 25 July 2019. Archived from the original on 21 August 2019. Retrieved 16 January 2020.
- ↑ 2.0 2.1 2.2 "Tretinoin topical Use During Pregnancy". Drugs.com. 1 July 2019. Archived from the original on 29 November 2020. Retrieved 16 January 2020.
- ↑ 3.0 3.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 25 February 2021. Retrieved 17 September 2020.
- ↑ 4.0 4.1 4.2 4.3 4.4 "Tretinoin". The American Society of Health-System Pharmacists. Archived from the original on 30 November 2016. Retrieved 8 December 2016.
- ↑ 5.0 5.1 Tivnan, Amanda (2016). Resistance to Targeted Therapies Against Adult Brain Cancers. Springer. p. 123. ISBN 9783319465050. Archived from the original on 2017-11-05.
- ↑ 6.0 6.1 6.2 6.3 6.4 British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. pp. 627, 821–822. ISBN 9780857111562.
- ↑ Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 476. ISBN 9783527607495. Archived from the original on 2017-11-05.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Tretinoin topical". The American Society of Health-System Pharmacists. Archived from the original on 16 May 2016. Retrieved 8 December 2016.
- ↑ 10.0 10.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- ↑ 11.0 11.1 "Tretinoin - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
- ↑ 12.0 12.1 12.2 12.3 "Tretinoin Cream- tretinoin cream". DailyMed. 1 December 2018. Archived from the original on 11 April 2021. Retrieved 16 January 2020.
- ↑ Leyden JJ, Shalita A, Thiboutot D, Washenik K, Webster G Topical Retinoids in Inflammatory Acne: A Retrospective, Investigator-Blinded, Vehicle-Controlled, Photographic Assessment https://pubmed.ncbi.nlm.nih.gov/15811485/ Archived 2020-07-11 at the Wayback Machine
- ↑ Webster G, Cargill DI, Quiring J, Vogelson CT, Slade HB A combined analysis of 2 randomized clinical studies of tretinoin gel 0.05% for the treatment of acne. https://pubmed.ncbi.nlm.nih.gov/19363908/ Archived 2020-07-13 at the Wayback Machine
- ↑ Management of Acne: A Report From a Global Alliance to Improve Outcomes in Acne https://pubmed.ncbi.nlm.nih.gov/12833004/ Archived 2020-07-11 at the Wayback Machine
- ↑ 16.0 16.1 16.2 Kang S, Voorhees JJ. Topical retinoids. In: Fitzpatrick's Dermatology in General Medicine, 7th ed, Wolff K, Goldsmith LA, Katz SI, et al (Eds), McGraw Hill, New York 2008. p.2106.
- ↑ 17.00 17.01 17.02 17.03 17.04 17.05 17.06 17.07 17.08 17.09 17.10 17.11 "Tretinoin capsule". DailyMed. 12 December 2018. Archived from the original on 29 August 2021. Retrieved 16 January 2020.
- ↑ Huang ME, Ye YC, Chen SR, Chai JR, Lu JX, Zhoa L, et al. (August 1988). "Use of all-trans retinoic acid in the treatment of acute promyelocytic leukemia" (PDF). Blood. 72 (2): 567–72. doi:10.1182/blood.V72.2.567.567. PMID 3165295.
- ↑ Castaigne S, Chomienne C, Daniel MT, Ballerini P, Berger R, Fenaux P, Degos L (November 1990). "All-trans retinoic acid as a differentiation therapy for acute promyelocytic leukemia. I. Clinical results" (PDF). Blood. 76 (9): 1704–9. doi:10.1182/blood.V76.9.1704.1704. PMID 2224119.
- ↑ Küley-Bagheri, Yasemin; Kreuzer, Karl-Anton; Monsef, Ina; Lübbert, Michael; Skoetz, Nicole (2018-08-06). Cochrane Haematological Malignancies Group (ed.). "Effects of all-trans retinoic acid (ATRA) in addition to chemotherapy for adults with acute myeloid leukaemia (AML) (non-acute promyelocytic leukaemia (non-APL))". Cochrane Database of Systematic Reviews. doi:10.1002/14651858.CD011960.pub2. Archived from the original on 2021-08-27. Retrieved 2020-07-10.
- ↑ Han, A; Chien, AL; Kang, S (July 2014). "Photoaging". Dermatologic clinics. 32 (3): 291–9, vii. doi:10.1016/j.det.2014.03.015. PMID 24891052.
- ↑ Zaenglein AL (September 2008). "Topical retinoids in the treatment of acne vulgaris". Semin Cutan Med Surg. 27 (3): 177–82. doi:10.1016/j.sder.2008.06.001. PMID 18786495.
- ↑ Wei S, Kozono S, Kats L, Nechama M, Li W, Guarnerio J, et al. (May 2015). "Active Pin1 is a key target of all-trans retinoic acid in acute promyelocytic leukemia and breast cancer". Nature Medicine. 21 (5): 457–66. doi:10.1038/nm.3839. PMC 4425616. PMID 25849135.
- ↑ Fernandez [Graber] EM, Zaenglein A, Thiboutot D. Acne Treatment Methodologies. In: Cosmetic Formulation of Skin Care Products, Taylor and Francis Group, New York 2006. p.273.
- ↑ Liu PT, Krutzik SR, Kim J, Modlin RL Cutting edge: all-trans retinoic acid down-regulates TLR2 expression and function. https://pubmed.ncbi.nlm.nih.gov/15728448/ Archived 2020-07-11 at the Wayback Machine
- ↑ Management of Acne: A Report From a Global Alliance to Improve Outcomes in Acne https://pubmed.ncbi.nlm.nih.gov/12833004/ Archived 2020-07-11 at the Wayback Machine
- ↑ Woggon, Wolf-D. (2009). "Oxidative cleavage of carotenoids catalyzed by enzyme models and beta-carotene 15,15´-monooxygenase". Pure and Applied Chemistry. 74 (8): 1397–1408. doi:10.1351/pac200274081397. ISSN 1365-3075.
- ↑ Vivant Pharmaceuticals, LLC Press Release. July 10, 2013, Vivant Skin Care Co-founder James E. Fulton, MD, Loses Colon Cancer Battle
- ↑ 29.0 29.1 Gellene, Denis (22 February 2010). "Dr. Albert M. Kligman, Dermatologist, Dies at 93". The New York Times. Archived from the original on 5 November 2017. Retrieved 25 February 2017.
- ↑ 30.0 30.1 Merriam-Webster, Merriam-Webster's Unabridged Dictionary, Merriam-Webster, archived from the original on 2020-05-25, retrieved 2016-07-12.
- ↑ 31.0 31.1 31.2 Oxford Dictionaries, Oxford Dictionaries Online, Oxford University Press, archived from the original on 2014-10-22.
- ↑ 32.0 32.1 32.2 Houghton Mifflin Harcourt, The American Heritage Dictionary of the English Language, Houghton Mifflin Harcourt, archived from the original on 2015-09-25, retrieved 2015-01-24.
- ↑ 33.0 33.1 Merriam-Webster, Merriam-Webster's Medical Dictionary, Merriam-Webster, archived from the original on 2020-10-10, retrieved 2015-01-21.
- ↑ 34.0 34.1 Elsevier, Dorland's Illustrated Medical Dictionary, Elsevier, archived from the original on 2014-01-11, retrieved 2015-01-21.
- ↑ Ralph M. Trüeb. The Difficult Hair Loss Patient: Guide to Successful Management of Alopecia and Related Conditions. Springer, 2015. ISBN 9783319197012 Pg. 95 Archived 2017-11-05 at the Wayback Machine
- ↑ Rogers NE, Avram MR (October 2008). "Medical treatments for male and female pattern hair loss". Journal of the American Academy of Dermatology. 59 (4): 547–66, quiz 567–8. doi:10.1016/j.jaad.2008.07.001. PMID 18793935.
- ↑ Arthur W. Perry (2007). Straight talk about cosmetic surgery. Yale University Press. pp. 63. ISBN 978-0-300-12104-9.
- ↑ Mukherjee S, Date A, Patravale V, Korting HC, Roeder A, Weindl G (December 2006). "Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety". Clinical Interventions in Aging. 1 (4): 327–48. doi:10.2147/ciia.2006.1.4.327. PMC 2699641. PMID 18046911.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Tretinoin Topical". MedlinePlus. Archived from the original on 2019-06-30. Retrieved 2020-01-17.
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- Articles with hatnote templates targeting a nonexistent page
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from July 2020
- Articles with invalid date parameter in template
- Anti-acne preparations
- Antineoplastic drugs
- Aromatase inhibitors
- Hoffmann-La Roche brands
- Carboxylic acids
- Carotenoids
- Cyclohexenes
- Retinoids
- World Health Organization essential medicines
- RTT