Etirinotecan pegol
Jump to navigation
Jump to search
 | |
| Clinical data | |
|---|---|
| Trade names | Onzeald |
| Other names | NKTR-102 |
| Routes of administration | Intravenous infusion |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | none |
| Metabolites | irinotecan and its metabolites |
| Elimination half-life | 38 days |
| Excretion | mostly via kidneys |
| Identifiers | |
| CAS Number | |
| DrugBank | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
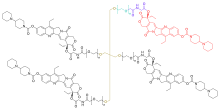
| Formula | C153H176N20O36[C8H16O4]n (n≈113) |
| Molar mass | 20,900–24,900 g/mol[1] |
Etirinotecan pegol (trade name Onzeald) is a drug developed by Nektar Therapeutics for the treatment of certain kinds of breast cancer with brain metastases. The European Medicines Agency refused to grant it a marketing authorisation in 2017.[2]
It works as a topoisomerase I inhibitor.[3] Chemically, it consists of four units of irinotecan (a topoisomerase I inhibitor in use since the late 1990s[4]) linked by carboxymethyl glycine and polyethylene glycol (PEG) chains to a central pentaerythritol ether, resulting in a much longer biological half-life (38 days) than that of irinotecan. It is formulated as a dihydrochloride and with 1.2 units of trifluoroacetate.[1]
References
- ^ a b "Onzeald: EPAR – Refusal public assessment report" (PDF). European Medicines Agency. 2018-02-02.
- ^ "Onzeald". European Medicines Agency. 2017-11-10.
- ^ Twelves C, Cortés J, O'Shaughnessy J, Awada A, Perez EA, Im SA, et al. (May 2017). "Health-related quality of life in patients with locally recurrent or metastatic breast cancer treated with etirinotecan pegol versus treatment of physician's choice: Results from the randomised phase III BEACON trial". European Journal of Cancer. 76: 205–215. doi:10.1016/j.ejca.2017.02.011. PMID 28360015.
- ^ "Drug Approval Package: Camptosar (Irinotecan Hydrochloride) NDA# 20-571/S-008". U.S. Food and Drug Administration (FDA). Retrieved 25 May 2020.
Categories:
- Articles with short description
- Short description matches Wikidata
- Infobox-drug molecular-weight unexpected-character
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without InChI source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Topoisomerase inhibitors
- All stub articles
- Antineoplastic and immunomodulating drug stubs