Levocetirizine
 | |
 | |
| Names | |
|---|---|
| Trade names | Xyzal, Levazyr, others |
| Other names | Levocetirizine dihydrochloride |
| |
| Clinical data | |
| Drug class | Antihistamine |
| Main uses | Allergic rhinitis (hay fever), hives[1] |
| Side effects | Sleepiness, dry mouth, cough, vomiting, diarrhea[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | 5 mg[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607056 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | High |
| Protein binding | 90% |
| Metabolism | Liver 14% CYP3A4 |
| Elimination half-life | 6 to 10 hours |
| Excretion | Kidney and fecal |
| Chemical and physical data | |
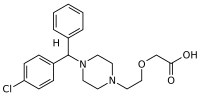
| Formula | C21H25ClN2O3 |
| Molar mass | 388.89 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Levocetirizine, sold under the brand name Xyzal among others, is an antihistamine used for the treatment of allergic rhinitis (hay fever) and long term hives of unclear cause.[1] It is less sedating than older antihistamines.[3] It is taken by mouth.[1]
Common side effects include sleepiness, dry mouth, cough, vomiting, and diarrhea.[1] Use in pregnancy appears safe but has not been well studied and use when breastfeeding is of unclear safety.[4] It is classified as a second-generation antihistamine and works by blocking histamine H1-receptors.[5][1]
Levocetirizine was approved for medical use in the United States in 2007.[1] It is available as a generic medication.[3] A month supply in the United Kingdom costs the NHS about 4.50 £ as of 2019.[3] In the United States the wholesale cost of this amount is about US$3.[6] In 2017, it was the 175th most commonly prescribed medication in the United States, with more than three million prescriptions.[7][8]
Medical uses
Levocetirizine is used for allergic rhinitis.[9] This includes allergy symptoms such as watery eyes, runny nose, sneezing, hives, and itching.[10]
Dosage
The defined daily dose is 5 mg by mouth.[2]
Side effects
Levocetirizine is a non-sedating antihistamine as it does not enter the brain in significant amounts, and is therefore unlikely to cause drowsiness. Cardiac safety with repolarization may be better than some other antihistamines, as levocetirizine does not significantly prolong the QT interval in healthy individuals.[11][12][13] Some people may still experience some slight sleepiness, headache, mouth dryness, lightheadedness, vision problems (mainly blurred vision), palpitations and fatigue.[14]
Pharmacology
Levocetirizine is an antihistamine. It acts as an inverse agonist that decreases activity at histamine H1 receptors. This in turn prevents the release of other allergy chemicals and increase the blood supply to the area, and provides relief from the typical symptoms of hay fever.
Chemistry
Chemically, levocetirizine is the active levorotary enantiomer of cetirizine, also called the l-enantiomer of cetirizine. It is a member of the diphenylmethylpiperazine group of antihistamines.
History
Levocetirizine was first launched in 2001 by Belgian pharmaceutical company UCB.
Society and culture
Cost
A month supply in the United Kingdom costs the NHS about 4.50 £ as of 2019.[3] In the United States the wholesale cost of this amount is about US$3.[6] In 2017, it was the 175th most commonly prescribed medication in the United States, with more than three million prescriptions.[7][8]
-
Levocetirizine costs (US)
-
Levocetirizine prescriptions (US)
Availability
On 31 January 2017, the Food and Drug Administration approved an over-the-counter version.[15] Although the drug was authorized by the FDA in 2007, it was already available in most European countries. Like many new drugs it entered the market at a higher price than currently available third and second generation antihistamines. In India, one form of the drug is available as Crohist MK tablets and syrup, a formulation of levocetirizine hydrochloride and montelukast. In India, Crohist MK is a Schedule 'H' drug and may only be prescribed by a registered medical practitioner.
Brand names

Levocetirizine is sold under the following brand names:
- Xyzal /ˈzaɪˌzɑːl/ in Australia, Austria, Bulgaria, Croatia, Czech Republic, Finland, France, India, Ireland (also Rinozal), Italy, Japan, Lithuania, Netherlands, Poland, Portugal, Romania, Taiwan, Thailand, Turkey, The Philippines, Serbia, Singapore, Slovakia, Slovenia, South Africa and UK. On May 25, 2007, the United States Food and Drug Administration approved Xyzal, where it is co-marketed by Sanofi-Aventis.
- Levobert in India
- Xusal in Germany, Mexico
- Xozal in Greece
- Xazal in Spain
- Allevo in Egypt
- In Hungary it is marketed as Zilola (made by Richter Gedeon), Histisynt (Actavis) and Xyzal (UCB).
- In Bangladesh, levocetirizine available as Alcet by Healthcare pharma, Curin by Beximco pharma and Seasonix by Incepta.
- In India, levocetirizine is marketed by GlaxoSmithKline under the brand name Vozet. Torrent Pharma launched UVNIL for the rural market. Kaptab Pharmaceuticals markets a mouth dissolving form in India.
- In Pakistan levocetirizine was first launched in a liquid formulation by Novartis Consumer Health Division under the name of T-Day Syrup.
- In Nepal levocetirizine is available in tablets with the brand name Curin manufactured by Beximco Pharma.[16]
- In the Czech Republic and Slovakia, it is also marketed as Zenaro as a prescription drug and is made by Zentiva (Sanofi).
- In Chile, it is marketed by GSK as Xuzal and as Zival and by Laboratorio Saval.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 "Levocetirizine Dihydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 1 April 2019. Retrieved 22 March 2019.
- ↑ 2.0 2.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 30 October 2020. Retrieved 7 September 2020.
- ↑ 3.0 3.1 3.2 3.3 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 280–281. ISBN 9780857113382.
- ↑ "Levocetirizine Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 22 March 2019. Retrieved 3 March 2019.
- ↑ Wallace DV, Dykewicz MS, Bernstein DI, Blessing-Moore J, Cox L, Khan DA, Lang DM, Nicklas RA, Oppenheimer J, Portnoy JM, Randolph CC, Schuller D, Spector SL, Tilles SA (August 2008). "The diagnosis and management of rhinitis: an updated practice parameter". The Journal of Allergy and Clinical Immunology. 122 (2 Suppl): S1-84. doi:10.1016/j.jaci.2008.06.003. PMID 18662584.
- ↑ 6.0 6.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 7.0 7.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 8.0 8.1 "Levocetirizine Dihydrochloride - Drug Usage Statistics". ClinCalc. Archived from the original on 13 April 2020. Retrieved 11 April 2020.
- ↑ Holgate, Stephen; Powell, Richard; Jenkins, Maureen; Ali, Omar (2005-06-13). "A treatment for allergic rhinitis:a view on the role of levocetirizine". Current Medical Research and Opinion. Informa Healthcare. 21 (7): 1099–1106. doi:10.1185/030079905x53298. ISSN 0300-7995. PMID 16004679.
The variable efficacy and durability of response of different antihistamines arise from differing modulatory effects on the H(1)-receptor. Conclusion: These findings support both the short-term and long-term use of levocetirizine in the clinical management of allergic rhinitis. The World Health Organization (WHO) ARIA Guidelines (Allergic Rhinitis and its Impact on Asthma), recommend using a combination of a non-sedating antihistamine with a decongestant, or glucocorticosteroids for treating allergic rhinitis - with the order and combination of treatment depending on severity and duration of symptoms.
- ↑ "Levocetirizine Oral". WebMD. Archived from the original on 2019-08-16. Retrieved 2017-08-21.
- ↑ Hulhoven R, Rosillon D, Letiexhe M, Meeus MA, Daoust A, Stockis A (November 2007). "Levocetirizine does not prolong the QT/QTc interval in healthy subjects: results from a thorough QT study". European Journal of Clinical Pharmacology. 63 (11): 1011–7. doi:10.1007/s00228-007-0366-5. PMID 17891537.
- ↑ "Cetirizine and loratadine: minimal risk of QT prolongation". Prescrire International. 19 (105): 26–8. February 2010. PMID 20455340.
- ↑ Poluzzi E, Raschi E, Godman B, Koci A, Moretti U, Kalaba M, Wettermark B, Sturkenboom M, De Ponti F (2015). "Pro-arrhythmic potential of oral antihistamines (H1): combining adverse event reports with drug utilization data across Europe". PLOS ONE. 10 (3): e0119551. Bibcode:2015PLoSO..1019551P. doi:10.1371/journal.pone.0119551. PMC 4364720. PMID 25785934.
- ↑ XOZAL technical specifications booklet.
- ↑ "Prescription to Over-the-Counter (OTC) Switch List". Archived from the original on April 24, 2019. Retrieved February 9, 2017.
- ↑ "Curin". Archived from the original on October 22, 2012. Retrieved August 29, 2012.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Articles with changed CASNo identifier
- Articles with changed EBI identifier
- Acetic acids
- Belgian inventions
- Benzene derivatives
- Chloroarenes
- Enantiopure drugs
- Ethers
- H1 receptor antagonists
- Peripherally selective drugs
- Piperazines
- RTT

