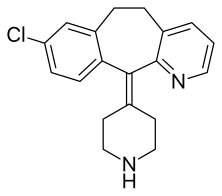
Desloratadine
 | |
 | |
| Names | |
|---|---|
| Trade names | Aerius, Allex, Clarinex, others[1][2] |
| |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | By mouth (tablets, solution) |
| Onset of action | Within 1 hour[3] |
| Duration of action | Up to 24 hours[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602002 |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | Rapidly absorbed |
| Protein binding | 83 to 87% |
| Metabolism | UGT2B10, CYP2C8 |
| Metabolites | 3-Hydroxydesloratadine |
| Elimination half-life | 27 hours |
| Excretion | 40% as conjugated metabolites into urine Similar amount into the feces |
| Chemical and physical data | |
| Formula | C19H19ClN2 |
| Molar mass | 310.83 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Desloratadine, sold under the brand name Clarinex among others, is a medication used to treat allergies such as hay fever and hives.[3] Onset occurs within an hour and lasts for up to 24 hours.[3] It is taken by mouth.[3]
Common side effects include nausea, tiredness, dry mouth, and headache.[3] A lower dose is recommended in those with kidney or liver problems.[3] While there is no clear evidence of harm during pregnancy, such use is generally not recommended.[4][5] It is a second generation antihistamine.[3] It is an active metabolite of loratadine.[3]
Desloratadine was patented in 1984 and came into medical use in 2001.[6] In the United Kingdom it is avaliable as a generic medication and costs the NHS about 1.5 pounds per month.[4] In the United States this amount costs about 17 USD.[7] It is also available with pseudoephedrine.[3]
Medical uses
Desloratadine is used to treat allergic rhinitis, nasal congestion and chronic idiopathic urticaria (hives).[8] It is the major metabolite of loratadine and the two drugs are similar in safety and effectiveness.[8] Desloratadine is available in many dosage forms and under many trade names worldwide.[9]
An emerging indication for desloratadine is in the treatment of acne, as an inexpensive adjuvant to isotretinoin and possibly as maintenance therapy or monotherapy.[10][11]
Dosage
In those over the age of 12 years the typical dose is 5 mg once per day.[4] Doses up to 20 mg per day; however, may be used.[12] In those 6 to 12 2.5 mg per day is recommended while 1.25 mg per day is recommended in those 1 to 6.[4]
Side effects
The most common side-effects are fatigue(1.2%[13]), dry mouth(3%[13]), and headache(0.6%[13]).[8]
Interactions
A number of drugs and other substances that are prone to interactions, such as ketoconazole, erythromycin and grapefruit juice, have shown no influence on desloratadine concentrations in the body. Desloratadine is judged to have a low potential for interactions.[14]
Pharmacology
Pharmacodynamics
Desloratadine is a selective H1-antihistamine which functions as an inverse agonist at the histamine H1 receptor.[15]
At very high doses, is also an antagonist at various subtypes of the muscarinic acetylcholine receptors. This effect is not relevant for the drug's action at therapeutic doses.[16]
Pharmacokinetics
Desloratadine is well absorbed from the gut and reaches highest blood plasma concentrations after about three hours. In the bloodstream, 83 to 87% of the substance are bound to plasma proteins.[14]
Desloratadine is metabolized to 3-hydroxydesloratadine in a three-step sequence in normal metabolizers. First, n-glucuronidation of desloratadine by UGT2B10; then, 3-hydroxylation of desloratadine N-glucuronide by CYP2C8; and finally, a non-enzymatic deconjugation of 3-hydroxydesloratadine N-glucuronide.[17] Both desloratadine and 3-hydroxydesloratadine are eliminated via urine and feces with a half-life of 27 hours in normal metabolizers.[14][18]

It exhibits only peripheral activity since it does not readily cross the blood-brain barrier; hence, it does not normally cause drowsiness because it does not readily enter the central nervous system.[19]
Desloratadine does not have a strong effect on a number of tested enzymes in the cytochrome P450 system. It was found to weakly inhibit CYP2B6, CYP2D6, and CYP3A4/CYP3A5, and not to inhibit CYP1A2, CYP2C8, CYP2C9, or CYP2C19. Desloratadine was found to be a potent and relatively selective inhibitor of UGT2B10, a weak to moderate inhibitor of UGT2B17, UGT1A10, and UGT2B4, and not to inhibit UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B7, UGT2B15, UGT1A7, and UGT1A8.[17]
Pharmacogenomics
2% of Caucasian people and 18% of people from African descent are desloratadine poor metabolizers. In these people, the drug reaches threefold highest plasma concentrations six to seven hours after intake, and has a half-life of about 89 hours. However, the safety profile for these subjects is not worse than for extensive (normal) metabolizers.[14][18]
Society and culture
Cost
When initially released in Canada in 2002 it sold for CA$0.82 per dose.[20]
-
Desloratadine costs (US)
-
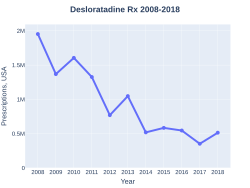
Desloratadine prescriptions (US)
References
- ↑ Murdoch, David; Goa, Karen L.; Keam, Susan J. (April 7, 2003). "Desloratadine: an update of its efficacy in the management of allergic disorders". Drugs. 63 (19): 2051–2077. doi:10.2165/00003495-200363190-00010. PMID 12962522.
- ↑ "Allex EPAR". European Medicines Agency (EMA). Archived from the original on 2020-12-27. Retrieved 2020-04-05.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 "Desloratadine Monograph for Professionals". Drugs.com. Archived from the original on 2020-08-12. Retrieved 2021-07-16.
- ↑ 4.0 4.1 4.2 4.3 BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 294. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ "Desloratadine Use During Pregnancy". Drugs.com. Archived from the original on 6 May 2021. Retrieved 16 July 2021.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 549. ISBN 9783527607495. Archived from the original on 2020-12-27. Retrieved 2020-05-09.
- ↑ "Desloratadine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 10 September 2016. Retrieved 16 July 2021.
- ↑ 8.0 8.1 8.2 See S (2003). "Desloratadine for allergic rhinitis". Am Fam Physician. 68 (10): 2015–6. PMID 14655812. Archived from the original on 2005-07-24. Retrieved 2005-08-01.
- ↑ Drugs.com Desloratadine entry at drugs.com international Archived 2020-08-03 at the Wayback Machine Page accessed May 4, 2015
- ↑ Lee HE, Chang IK, Lee Y, Kim CD, Seo YJ, Lee JH, Im M (2014). "Effect of antihistamine as an adjuvant treatment of isotretinoin in acne: a randomized, controlled comparative study". J Eur Acad Dermatol Venereol. 28 (12): 1654–60. doi:10.1111/jdv.12403. PMID 25081735. S2CID 3406128.
- ↑ Layton AM (2016). "Top Ten List of Clinical Pearls in the Treatment of Acne Vulgaris". Dermatol Clin. 34 (2): 147–57. doi:10.1016/j.det.2015.11.008. PMID 27015774.
- ↑ "Help, "hive" got a rash! – Article 2: Practical tips in the management of chronic spontaneous urticaria (CSU) | This Changed My Practice". thischangedmypractice.com. Archived from the original on 30 June 2023. Retrieved 1 June 2023.
- ↑ 13.0 13.1 13.2 González-Núñez, Vanesa; Valero, Antonio; Mullol, Joaquim (2013-04-11). "Safety evaluation of desloratadine in allergic rhinitis". Expert Opinion on Drug Safety. Informa Healthcare. 12 (3): 445–453. doi:10.1517/14740338.2013.788148. ISSN 1474-0338. PMID 23574541. S2CID 40472187.
- ↑ 14.0 14.1 14.2 14.3 "Aerius: EPAR – Product Information" (PDF). European Medicines Agency. 2017-06-07. Archived (PDF) from the original on 2019-07-05. Retrieved 2017-10-13.
- ↑ Canonica GW, Blaiss M (2011). "Antihistaminic, anti-inflammatory, and antiallergic properties of the nonsedating second-generation antihistamine desloratadine: a review of the evidence". World Allergy Organ J. 4 (2): 47–53. doi:10.1097/WOX.0b013e3182093e19. PMC 3500039. PMID 23268457.
- ↑ "Aerius: EPAR – Scientific Discussion" (PDF). European Medicines Agency. 2006-04-03. Archived (PDF) from the original on 2018-03-16. Retrieved 2017-10-13.
- ↑ 17.0 17.1 Kazmi, F.; Yerino, P.; Barbara, J. E.; Parkinson, A. (2015-07-01). "Further Characterization of the Metabolism of Desloratadine and Its Cytochrome P450 and UDP-glucuronosyltransferase Inhibition Potential: Identification of Desloratadine as a Relatively Selective UGT2B10 Inhibitor". Drug Metabolism and Disposition. 43 (9): 1294–1302. doi:10.1124/dmd.115.065011. ISSN 1521-009X. PMID 26135009.
- ↑ 18.0 18.1 Drugs.com: Desloratadine Monograph.
- ↑ Mann R, Pearce G, Dunn N, Shakir S (2000). "Sedation with "non-sedating" antihistamines: four prescription-event monitoring studies in general practice". BMJ. 320 (7243): 1184–6. doi:10.1136/bmj.320.7243.1184. PMC 27362. PMID 10784544. Archived from the original on 2018-11-01. Retrieved 2006-05-31.
- ↑ "Report on New Patented Drugs - Aerius". pmprb-cepmb.gc.ca. 13 May 2014. Archived from the original on 18 October 2020. Retrieved 25 November 2019.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 maint: date format
- Webarchive template wayback links
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Benzocycloheptapyridines
- Chloroarenes
- H1 receptor antagonists
- Human drug metabolites
- Peripherally selective drugs
- Piperidines
- Schering-Plough brands
- Merck & Co. brands
- RTT