Mozavaptan
Jump to navigation
Jump to search
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
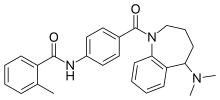
| Formula | C27H29N3O2 |
| Molar mass | 427.548 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Mozavaptan (INN) is a vasopressin receptor antagonist marketed by Otsuka. In Japan, it was approved in October 2006 for hyponatremia (low blood sodium levels) caused by syndrome of inappropriate antidiuretic hormone (SIADH) due to ADH producing tumors.
References
- Spreitzer H (November 20, 2006). "Neue Wirkstoffe - Conivaptan". Österreichische Apothekerzeitung (in German) (24/2006).
- "Conivaptan hydrochloride". Molecule of the Month. Prous Science. November 2006. Archived from the original on 2012-02-13.
Categories:
- Articles with short description
- Short description matches Wikidata
- Drugs not assigned an ATC code
- Drugs with non-standard legal status
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Drugboxes which contain changes to verified fields
- CS1 German-language sources (de)
- Diuretics
- Benzanilides
- Benzazepines
- Vasopressin receptor antagonists
- 2-Tolyl compounds
- All stub articles
- Antihypertensive agent stubs