Peginterferon alfa-2a
| Names | |
|---|---|
| Trade names | Pegasys, others |
| Clinical data | |
| Drug class | Alpha interferon[1] |
| Main uses | Hepatitis C, hepatitis B[1] |
| Side effects | Headache, feeling tired, depression, trouble sleeping, hair loss, nausea, pain at the site of injection, fever[1] |
| Pregnancy category |
|
| Routes of use | Subcutaneous |
| Defined daily dose | 26 mcg[2] |
| External links | |
| AHFS/Drugs.com | Professional Drug Facts |
| MedlinePlus | a605029 |
| Legal | |
| License data | |
| Legal status |
|
| Chemical and physical data | |
| Formula | C860H1353N227O255S9 |
| Molar mass | 19241 g/mol (unpegylated) 40000 g/mol (pegylated) g·mol−1 |
Pegylated interferon alfa-2a, sold under the brand name Pegasys among others, is medication used to treat hepatitis C and hepatitis B.[1] For hepatitis C it is typically used together with ribavirin and cure rates are between 24 and 92%.[1][3] For hepatitis B it may be used alone.[4] It is given by injection under the skin.[1]
Side effects are common.[5] They may include headache, feeling tired, depression, trouble sleeping, hair loss, nausea, pain at the site of injection, and fever.[1] Severe side effects may include psychosis, autoimmune disorders, blood clots, or infections.[1] Use with ribavirin is not recommended during pregnancy.[1] Pegylated interferon alfa-2a is in the alpha interferon family of medications.[1] It is pegylated to protect the molecule from breakdown.[5]
Pegylated interferon alfa-2a was approved for medical use in the United States in 2002.[1] It was on the World Health Organization's List of Essential Medicines; however, was removed in 2023.[6] The wholesale cost in the developing world is between 500.00 and US$4,800.00 for 12 weeks.[7] In the United States this costs about US$9,250.00,[5] while in the United Kingdom 12 weeks costs the NHS about 1,490 pounds.[4]
Medical uses
This drug is approved around the world for the treatment of chronic hepatitis C (including people with HIV co-infection, cirrhosis, 'normal' levels of ALT) and has recently been approved (in the EU, U.S., China and many other countries) for the treatment of chronic hepatitis B. It is also used in the treatment of certain T-cell lymphomas, particularly mycosis fungoides.
Peginterferon alfa-2a is a long acting interferon. Interferons are proteins released in the body in response to viral infections. Interferons are important for fighting viruses in the body, for regulating reproduction of cells, and for regulating the immune system.[8]
Host genetic factors
For genotype 1 hepatitis C treated with pegylated interferon alfa-2a or pegylated interferon alfa-2b combined with ribavirin, it has been shown that genetic polymorphisms near the human IL28B gene, encoding interferon lambda 3, are associated with significant differences in response to the treatment. This finding, originally reported in Nature,[9] showed genotype 1 hepatitis C patients carrying certain genetic variant alleles near the IL28B gene are more likely to achieve sustained virological response after the treatment than others. Another report in Nature [10] demonstrated the same genetic variants are also associated with the natural clearance of the genotype 1 hepatitis C virus.
Other infections
Has also been used for Middle East respiratory syndrome and hepatitis E.[11]
Dosage
The defined daily dose is 26 mcg (by injection).[2]
Side effects
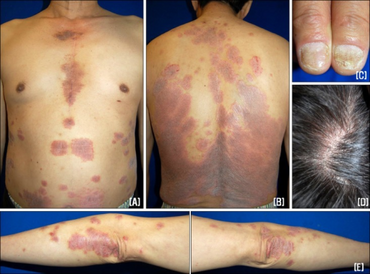
Peginterferon alfa-2a has the following side effects when administered:[12]
- Fever
- Lack of appetite
- Pale skin
- Cough
- Fatigue
- Sore throat
Manufacture
It is pegylated with a branched 40 kg/mol PEG chain. The drug is manufactured under the brand name Pegasys by Roche Pharmaceuticals
Research
A Cochrane review sought to determine whether interferon alfa-2a could be used as a treatment for individuals with neovascular age-related macular degeneration. They found no evidence of improved visual acuity with potential harm.[13]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Peginterferon Alfa-2a (Professional Patient Advice) - Drugs.com". www.drugs.com. Archived from the original on 16 January 2017. Retrieved 12 January 2017.
- ↑ 2.0 2.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 6 August 2020. Retrieved 14 September 2020.
- ↑ "Pegasys 135 mcg and 180 mcg solution for injection in pre-filled pen - Summary of Product Characteristics (SPC) - (eMC)". www.medicines.org.uk. Archived from the original on 13 January 2017. Retrieved 12 January 2017.
- ↑ 4.0 4.1 British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. p. 639. ISBN 9780857111562.
- ↑ 5.0 5.1 5.2 "Peginterferon alfa-2a (Pegasys) - Treatment - Hepatitis C Online". www.hepatitisc.uw.edu. Archived from the original on 23 December 2016. Retrieved 12 January 2017.
- ↑ "eEML - Electronic Essential Medicines List". list.essentialmeds.org. Archived from the original on 3 February 2023. Retrieved 15 September 2023.
- ↑ "Inclusion of the injectable formulation of peginterferon alfa-2a and -2b is proposed for the treatment of hepatitis C among adults" (PDF). who.int. p. 11. Archived (PDF) from the original on 13 January 2017. Retrieved 12 January 2017.
- ↑ "Interferon alfa-2a Uses, Side Effects & Warnings". Drugs.com. Archived from the original on 2020-01-11. Retrieved 2020-01-10.
- ↑ Ge D, Fellay J, Thompson AJ, et al. (2009). "Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance". Nature. 461 (7262): 399–401. doi:10.1038/nature08309. PMID 19684573.
- ↑ Thomas DL, Thio CL, Martin MP, et al. (2009). "Genetic variation in IL28B and spontaneous clearance of hepatitis C virus". Nature. 461 (7265): 798–801. doi:10.1038/nature08463. PMC 3172006. PMID 19759533.
- ↑ "Peginterferon Alfa Monograph for Professionals - Drugs.com". www.drugs.com. Archived from the original on 16 January 2017. Retrieved 12 January 2017.
- ↑ "Peginterferon alfa-2a Side Effects: Common, Severe, Long Term". Drugs.com. Archived from the original on 17 June 2019. Retrieved 31 October 2021.
- ↑ Reddy U, Krzystolik M (2006). "Antiangiogenic therapy with interferon alfa for neovascular age-related macular degeneration". Cochrane Database Syst Rev. 1: CD005138. doi:10.1002/14651858.CD005138.pub2. PMID 16437522.
External links
| Identifiers: |
|
|---|
- "Peginterferon alfa-2a". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-03-24. Retrieved 2020-03-24.
- Interferon and hepatitis c at pegasys.com Archived 2006-10-21 at the Wayback Machine
- Pegasys at the US National Library of Medicine Medical Subject Headings (MeSH)
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chem-molar-mass both hardcoded and calculated
- Infobox-drug molecular-weight unexpected-character
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles without a structure image
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Articles with changed CASNo identifier
- Chemicals that do not have a ChemSpider ID assigned
- Articles with changed EBI identifier
- Webarchive template wayback links
- Antivirals
- Immunostimulants
- World Health Organization essential medicines (removed)
- Hoffmann-La Roche brands
- Genentech brands
- RTT