Remdesivir
 | |
 | |
| Names | |
|---|---|
| Pronunciation | /rɛmˈdɛsɪvɪər/ rem-DESS-i-veer |
| Trade names | Veklury |
| Other names | GS-5734 |
| |
| Clinical data | |
| Main uses | COVID-19[1] |
| Side effects | Liver problems, nausea, allergic reactions[2][1] |
| Pregnancy category |
|
| Routes of use | Intravenous |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a620033 |
| Legal | |
| License data |
|
| Legal status | |
| Chemical and physical data | |
| Formula | C27H35N6O8P |
| Molar mass | 602.585 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Remdesivir, sold under the brand name Veklury, is a antiviral medication used for COVID‑19.[1][4] In the USA and EU, as of 2022, it is recommended for mild to moderate disease at risk of worsening, including those who require oxygen therapy.[11][4] In November of 2020 the World Health Organization conditionally recommendation against its use for COVID-19, regardless of disease severity.[12] It is given by injection into a vein for 3 to 10 days.[1][11]
Common side effects include increased liver enzymes and nausea.[1] Other side effects may include allergic reactions.[2] It is unclear if use during pregnancy is safe; though breastfeeding appears okay.[13] Remdesivir is converted by the body into GS-441524 triphosphate, a ribonucleotide analogue inhibitor of viral RNA polymerase.[14]
Remdesivir was approved for medical use in Europe in July 2020 and the United States in October 2020.[4][15] It was developed by Gilead Sciences and was originally studied for hepatitis C, Ebola, and Marburg infections.[16][17] In the United States it costs about 2,600 USD for five days as of 2021.[18] This amount in the UK costs about £2,040.[19]
Medical uses
COVID19
In November 2020, the World Health Organization (WHO) updated its guideline on COVID-19 to include a conditional recommendation against the use of remdesivir, triggered by results from the Solidarity trial.[12][20]
The European Medicines Agency announced that they will evaluate new data to see if a revision to the authorization of remdesivir is needed.[21] In the European Union, remdesivir was used to treat COVID‑19 in those aged twelve and older with body weight at least 40 kilograms (88 lb) with pneumonia requiring supplemental oxygen.[7][4]
In the United States, remdesivir was indicated for use in those aged twelve years and older with body weight at least 40 kilograms (88 lb) for the treatment of COVID‑19 requiring hospitalization.[10] Three days of intravenous treatment may also be used in those at high risk of severe disease, per the NIH as of December of 2021.[11]
In November 2020, the FDA issued an emergency use authorization (EUA) for the combination of baricitinib with remdesivir, for the treatment of suspected or laboratory confirmed COVID-19 in hospitalized people two years of age or older requiring supplemental oxygen, invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).[22]
Experts in the British Medical Journal stated, remdesivir "probably has no important effect on the need for mechanical ventilation and may have little or no effect on the length of hospital stay". Because of the high price, remdesivir may divert funds and efforts away from other treatments against COVID‑19.[23][24]
Dosage

It is given at a dose of 200 mg the first day, followed by 100 mg per day.[4] Generally 3 to 10 days are used.[4]
Side effects
The most common side effects in people treated with remdesivir were respiratory failure and blood biomarkers of organ impairment, including low albumin, low potassium, low count of red blood cells, low count of thrombocytes, and elevated bilirubin (jaundice).[25] Other reported adverse effects include gastrointestinal distress, elevated transaminase levels in the blood (liver enzymes), infusion site reactions, and electrocardiogram abnormalities.[26] Remdesivir may cause infusion‐related reactions, including low blood pressure, nausea, vomiting, sweating or shivering.[27]
Other possible side effects of include:
- Infusion‐related reactions. Infusion‐related reactions have been seen during a remdesivir infusion or around the time remdesivir was given.[27] Signs and symptoms of infusion‐related reactions may include: low blood pressure, nausea, vomiting, sweating, and shivering.[27]
- Increases in levels of liver enzymes, seen in abnormal liver blood tests.[27] Increases in levels of liver enzymes have been seen in people who have received remdesivir, which may be a sign of inflammation or damage to cells in the liver.[27]
Interactions
Remdesivir is at least partially metabolized by the cytochrome P450 enzymes CYP2C8, CYP2D6, and CYP3A4.[28][29] Blood plasma concentrations of remdesivir are expected to decrease if it is administered together with cytochrome P450 inducers such as rifampicin, carbamazepine, phenobarbital, phenytoin, primidone, and St John's wort.[30]
Using chloroquine or hydroxychloroquine with remdesivir may reduce the antiviral activity of remdesivir.[9][7][31] Coadministration of remdesivir and chloroquine phosphate or hydroxychloroquine sulfate is not recommended based on in vitro data demonstrating an antagonistic effect of chloroquine on the intracellular metabolic activation and antiviral activity of remdesivir.[29]
Pharmacology
Activation

Remdesivir is a ProTide (Prodrug of nucleoTide). It is able to diffuse into cells where it is converted to GS-441524 mono-phosphate via the actions of esterases (CES1 and CTSA) and a phosphoamidase (HINT1); this in turn is further phosphorylated to its active metabolite triphosphate by nucleoside-phosphate kinases.[33][34] This pathway of bioactivation is meant to occur intracellularly, but a substantial amount of remdesivir is prematurely hydrolyzed in plasma, with GS-441524 being the major metabolite in plasma, and the only metabolite remaining two hours after dosing.[14]
Mechanism of action
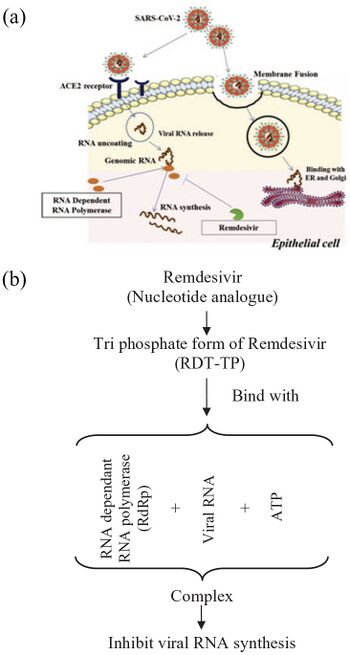
As an adenosine nucleoside triphosphate analog (GS-443902),[36] the active metabolite of remdesivir interferes with the action of viral RNA-dependent RNA polymerase and evades proofreading by viral exoribonuclease (ExoN), causing a decrease in viral RNA production.[16][37] In some viruses such as the respiratory syncytial virus it causes the RNA-dependent RNA polymerases to pause, but its predominant effect (as in Ebola) is to induce an irreversible chain termination. Unlike with many other chain terminators, this is not mediated by preventing addition of the immediately subsequent nucleotide, but is instead delayed, occurring after five additional bases have been added to the growing RNA chain.[38] For the RNA-Dependent RNA Polymerase of MERS-CoV, SARS-CoV-1, and SARS-CoV-2 arrest of RNA synthesis occurs after incorporation of three additional nucleotides.[39][34] Hence, remdesivir is classified as a direct-acting antiviral agent that works as a delayed chain terminator.[32][34]
Pharmacokinetics
In non-human primates, the plasma half-life of the prodrug is 20 minutes, with the main metabolite being the nucleoside, GS-441524. Two hours post injection, the main metabolite GS-441524 is present at micromolar concentrations, whilst intact Remdesivir is no longer detectable. Because of this rapid extracellular conversion to the nucleoside GS-441524, some researchers have questioned whether the active nucleotide triphosphate is truly derived from Remdesivir pro-drug removal or whether it occurs by GS-441524 phosphorylation, and whether direct administration of GS-441524 would constitute a cheaper and easier to administer COVID‑19 drug compared to Remdesivir.[40][14] The activated nucleotide triphosphate form has sustained intracellular levels in PBMC and presumably in other cells as well.[32]
Resistance
Mutations in the mouse hepatitis virus RNA replicase that cause partial resistance to remdesivir were identified in 2018. These mutations make the viruses less effective in nature, and the researchers believe they will likely not persist where the drug is not being used.[41]
Society and culture
Names
Remdesivir is the international nonproprietary name (INN)[42] while the development code name was GS-5734.[43]
Price
On 29 June 2020, Gilead announced that it had set the price of remdesivir at US$390 per vial for the governments of developed countries, including the United States, and US$520 for US private health insurance companies.[44] The expected course of treatment is six vials over five days for a total cost of US$2,340.[44] Being a repurposed drug, the minimum production cost for remdesivir is estimated at US$0.93 per day of treatment.[45]
Authorizations
Remdesivir is approved or authorized for emergency use to treat COVID‑19 in around 50 countries.[46] Remdesivir has been authorized for emergency use in India,[47] Singapore,[48] and approved for use in Japan, the European Union, the United States, and Australia for people with severe symptoms.[4][49][50][51][52][53][54][55][10] In February 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) started an evaluation to decide if the indication for remdesivir should be modified to include those not requiring supplemental oxygen.[56]
Remdesivir is the first treatment for COVID‑19 to be approved by the U.S. Food and Drug Administration (FDA).[10] The approval by the FDA does not include the entire population that had been authorized to use remdesivir under an Emergency Use Authorization (EUA) originally issued on 1 May 2020.[10] In order to ensure continued access to the pediatric population previously covered under the EUA, the FDA revised the EUA for remdesivir to authorize the drug's use for treatment of suspected or laboratory-confirmed COVID‑19 in hospitalized pediatric patients weighing 3.5 kilograms (7.7 lb) to less than 40 kilograms (88 lb) or hospitalized pediatric patients less than twelve years of age weighing at least 3.5 kilograms (7.7 lb).[10] Clinical trials assessing the safety and efficacy of remdesivir in this pediatric patient population are ongoing.[10]
Australia
In July 2020, remdesivir was provisionally approved for use in Australia for use in adults and adolescents with severe COVID‑19 symptoms who have been hospitalized.[53][57] Australia claims to have a sufficient supply of remdesivir in its national stockpile.[58]
Canada
As of 11 April 2020, access in Canada was available only through clinical trials.[59] Health Canada approved requests to treat twelve people with remdesivir under the department's special-access program (SAP).[60] Additional doses of remdesivir are not available through the SAP except for pregnant women or children with confirmed COVID‑19 and severe illness.[59]
On 19 June 2020, Health Canada received an application from Gilead for the use of remdesivir for treating COVID‑19.[59] On 27 July 2020, Health Canada conditionally approved the application.[61][6]
On 22 September 2020, Minister of Public Services and Procurement Anita Anand announced that Canada had entered into a deal to obtain up to 150,000 vials of remdesivir from Gilead starting in October.[62] As of 8 October, remdesivir was still not widely available in Alberta, because Alberta Health Services was undertaking a "formulary review" to be completed by mid-November.[63]
Czech Republic
On 17 March 2020, the drug was provisionally approved for use for COVID‑19 patients in a serious condition as a result of the outbreak in the Czech Republic.[64]
European Union
On 17 February 2016, orphan designation (EU/3/16/1615) was granted by the European Commission to Gilead Sciences International Ltd, United Kingdom, for remdesivir for the treatment of Ebola virus disease.[65]
In April 2020, the European Medicines Agency (EMA) provided recommendations on compassionate use of remdesivir for COVID‑19 in the EU.[66]
On 11 May 2020, the Committee for Medicinal Products for Human Use (CHMP) of the EMA recommended expanding the compassionate use of remdesivir to those not on mechanical ventilation.[67] In addition to those undergoing invasive mechanical ventilation, the compassionate use recommendations cover the treatment of hospitalized individuals requiring supplemental oxygen, non-invasive ventilation, high-flow oxygen devices or ECMO (extracorporeal membrane oxygenation).[67] The updated recommendations were based on preliminary results from the NIAID-ACTT study,[68] which suggested a beneficial effect of remdesivir in the treatment of hospitalized individuals with severe COVID‑19.[67][69] In addition, a treatment duration of five days was introduced alongside the longer ten-day course, based on preliminary results from another study (GS-US-540-5773) suggesting that for those not requiring mechanical ventilation or ECMO, the treatment course may be shortened from ten to five days without any loss of efficacy.[67] Individuals who receive a five-day treatment course but do not show clinical improvement will be eligible to continue receiving remdesivir for an additional five days.[67]
On 3 July 2020, the European Union granted a conditional marketing authorization for remdesivir with an indication for the treatment of coronavirus disease 2019 (COVID‑19) in adults and adolescents (aged twelve years and older with body weight at least 40 kilograms [88 lb]) with pneumonia requiring supplemental oxygen.[4][70] At the end of July, the European Union secured a €63 million (US$74 million) contract with Gilead, to make the drug available there in early August 2020.[71]
On 8 October 2020, Gilead Sciences and the European Commission announced they had signed a joint procurement framework contract in which Gilead agreed to provide up to 500,000 remdesivir treatment courses over the next six months to 37 European countries. Among the contracting countries were all 27 EU member states plus the United Kingdom, "Albania, Bosnia & Herzegovina, Iceland, Kosovo, Montenegro, North Macedonia, Norway, and Serbia".[72] At the time, the price per treatment course was not disclosed; on 13 October, Reuters reported the price was 2,070 euros, thereby implying the total value of the contract (if all 500,000 courses are ordered) is approximately €1.035 billion.[73] Under the new contract, each participating country will directly place orders with Gilead and pay Gilead directly for its own orders.[73]
Iran
Remdesivir has been also produced in Iran by Barakat; Iran is planning to increase the productions of Remdesivir ampoules from 20,000 to 150,000 ampoules per month. It has also the permission of the "Food and Drug Administration" of MOHME[74]
Japan
On 7 May 2020, Japan's Ministry of Health, Labour and Welfare approved the drug for use in Japan, in a fast-tracked process, based on the US emergency authorization.[49][52]
Mexico
On 23 October 2020, Deputy Secretary of Prevention and Health Promotion Hugo López-Gatell Ramírez stated at a news conference that Mexico would not necessarily follow the United States in approving the drug for use in Mexico. López-Gatell explained that Cofepris had already twice denied the approval of remdesivir because, in that agency's view, the evidence does not suggest "sufficient efficacy".[75] On 12 March 2020, Cofepris authorized the drug for emergency cases, advising to give continuous surveillance of the integral health of the patient.[76]
United States
On 20 March 2020, United States President Donald Trump announced that remdesivir was available for "compassionate use" for people with COVID‑19; FDA Commissioner Stephen Hahn confirmed the statement at the same press conference.[77] It was later revealed that Gilead had been providing remdesivir in response to compassionate use requests since 25 January.[17][78] On 23 March 2020, Gilead voluntarily suspended access for compassionate use (excepting cases of critically ill children and pregnant women), for reasons related to supply, citing the need to continue to provide the agent for testing in clinical trials.[79][80] It was the first medication approved for COVID19 in the USA.[15]
On 1 May 2020, the US Food and Drug Administration granted Gilead emergency use authorization (EUA) for remdesivir to be distributed and used by licensed health care providers to treat adults and children hospitalized with severe COVID‐19.[51][27] Severe COVID‐19 is defined as patients with an oxygen saturation (SpO2) <= 94% on room air or requiring supplemental oxygen or requiring mechanical ventilation or requiring extracorporeal membrane oxygenation (ECMO), a heart–lung bypass machine.[29][27][81][82] Distribution of remdesivir under the EUA will be controlled by the US government for use consistent with the terms and conditions of the EUA.[27] Gilead will supply remdesivir to authorized distributors, or directly to a US government agency, who will distribute to hospitals and other healthcare facilities as directed by the US government, in collaboration with state and local government authorities, as needed.[27] Gilead stated they were donating 1.5 million vials for emergency use[81] and estimated, as of April 2020, they had enough remdesivir for 140,000 treatment courses and expect to have 500,000 courses by October 2020, and one million courses by the end of 2020.[83][84]
The initial distribution of the drug in the US was tripped up by seemingly capricious decision-making and finger-pointing, resulting in over a week of confusion and frustration among health care providers and patients alike.[85][86][87] On 9 May 2020, the United States Department of Health and Human Services (HHS) explained in a statement that it would be distributing remdesivir vials to state health departments, then would allow each department to redistribute vials to hospitals in their respective states based upon each department's insight into "community-level needs."[88] HHS also clarified that only 607,000 vials of Gilead's promised donation of 1.5 million vials would be going to American patients.[88] However, HHS did not explain why several states with some of the highest caseloads had been omitted from the first two distribution rounds, including California, Florida, and Pennsylvania.[88] In May 2020, Gilead indicated they would increase the number of doses donated to the US from 607,000 to around 940,000.[89][87] Some of the initial distribution was sent to the wrong hospitals, to hospitals with no intensive care units, and to facilities without the needed refrigeration to store it.[87]
On 29 June, HHS announced an unusual agreement with Gilead in which HHS agreed to Gilead's wholesale acquisition price, HHS would continue to work together with state governments and drug wholesaler AmerisourceBergen to allocate shipments of remdesivir vials to American hospitals through the end of September 2020, and in exchange, during that three-month timeframe (July, August, and September), American patients would be allocated over 90% of Gilead's projected remdesivir output of more than 500,000 treatment courses.[90][91] Absent from these announcements was any discussion of allocation of remdesivir production to the approximately 70 countries omitted from Gilead's generic drug licensing agreements—including much of Europe[92] and countries as populous as Brazil, China, and Mexico—or the 127 countries listed on those agreements (during the time it will take for Gilead's generic licensees to ramp up their own production).[93] As the implications of this began to sink in, several countries publicly confirmed the next day that they already had adequate supplies of remdesivir to cover current needs, including Australia,[94] Germany,[95] and the United Kingdom.[96]
On 28 August 2020, the FDA broadened the Emergency Use Authorization (EUA) for remdesivir to include all hospitalized patients with suspected or laboratory-confirmed COVID‑19, irrespective of the severity of their disease.[97][98] The Fact Sheet was updated to reflect the new guidance.[29]
On 1 October 2020, Gilead and HHS announced that HHS was relinquishing control over remdesivir allocation because production of the drug had finally caught up with US domestic demand.[99][100][101] AmerisourceBergen will remain the sole distributor of Veklury in the US through the end of 2020.[101]
On 22 October 2020, the FDA approved remdesivir and also revised the EUA to permit the use of remdesivir for treatment of suspected or laboratory confirmed COVID‑19 in hospitalized children weighing 3.5 kilograms (7.7 lb) to less than 40 kilograms (88 lb) or hospitalized children less than twelve years of age weighing at least 3.5 kilograms (7.7 lb).[10][29][102][103]
In November 2020, the FDA issued an EUA for the combination of baricitinib with remdesivir, for the treatment of suspected or laboratory-confirmed COVID-19 in hospitalized people two years of age or older requiring supplemental oxygen, invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).[22] The data supporting the EUA for baricitinib combined with remdesivir are based on a randomized, double-blind, placebo-controlled clinical trial (ACTT-2), which was conducted by the National Institute of Allergy and Infectious Diseases (NIAID).[22] The EUA was issued to Eli Lilly and Company.[22]
In the face of emerging sales of the drug's active ingredient to compounders, the FDA recommended in February 2021 that remdesivir "should not be compounded."[104]
Manufacturing

Remdesivir requires "70 raw materials, reagents, and catalysts" to make, and approximately "25 chemical steps."[105] Some of the ingredients are extremely dangerous to humans, especially trimethylsilyl cyanide.[105] The original end-to-end manufacturing process required 9 to 12 months to go from raw materials at contract manufacturers to finished product, but after restarting production in January, Gilead Sciences was able to find ways to reduce the production time to six months.[105]
In January 2020, Gilead began working on restarting remdesivir production in glass-lined steel chemical reactors at its manufacturing plant in Edmonton, Alberta.[105] On 2 February 2020, the company flew its entire stock of remdesivir, 100 kilograms in powder form (leftover from Ebola research), to its filling plant in La Verne, California to start filling vials.[105] The Edmonton plant finished its first new batch of remdesivir in April 2020.[105] Around the same time, fresh raw materials began to arrive from contract manufacturers reactivated by Gilead in January.[105]
Another challenge is getting remdesivir into patients despite the drug's "poor predicted solubility and poor stability."[106] In June 2020, Ligand Pharmaceuticals revealed that Gilead has been managing those issues by mixing Ligand's proprietary excipient Captisol (based on University of Kansas research into cyclodextrin) with remdesivir at a 30:1 ratio.[106] Since that implies an enormous amount of Captisol is needed to stabilize and deliver remdesivir (on top of amounts needed for several other drugs for which the excipient is already in regular use), Ligand announced that it is trying to boost Captisol annual manufacturing capacity to as much as 500 metric tons.[106]
On 12 May 2020, Gilead announced that it had granted non-exclusive voluntary licenses to five generic drug companies in India and Pakistan to manufacture remdesivir for distribution to 127 countries.[107][108][109] The agreements were structured so that the licensees can set their own prices and will not have to pay royalties to Gilead until the WHO declares an end to the COVID‑19 emergency or another medicine or vaccine is approved for COVID‑19, whichever comes first.[107] On 23 June 2020, India granted emergency marketing approval of generic remdesivir manufactured by two Gilead licensees, Cipla and Hetero Drugs.[110]
Remdesivir can be synthesized in multiple steps from ribose derivatives. The figure to the right is one of the synthesis routes of remdesivir invented by Chun and coauthors from Gilead Sciences.[111][112] In this method, intermediate a is firstly prepared from L-alanine and phenyl phosphorodichloridate in presence of triethylamine and dichloromethane; triple benzyl-protected ribose is oxidized by dimethyl sulfoxide with acetic anhydride and give the lactone intermediate b; pyrrolo[2,1-f] [1,2,4]triazin-4-amine is brominated, and the amine group is protected by excess trimethylsilyl chloride. n-Butyllithium undergoes a halogen-lithium exchange reaction with the bromide at −78 °C (−108 °F) to yield the intermediate c. The intermediate b is then added to a solution containing intermediate c dropwise. After quenching the reaction in a weakly acidic aqueous solution, a mixture of 1:1 anomers was obtained. It was then reacted with an excess of trimethylsilyl cyanide in dichloromethane at −78 °C (−108 °F) for 10 minutes. Trimethylsilyl triflate was added and reacts for one additional hour, and the mixture was quenched in an aqueous sodium hydrogen carbonate. A nitrile intermediate was obtained. The protective group, benzyl, was then removed with boron trichloride in dichloromethane at −20 °C (−4 °F). The excess of boron trichloride was quenched in a mixture of potassium carbonate and methanol. A benzyl-free intermediate was obtained. The isomers were then separated via reversed-phase HPLC. The optically pure compound and intermediate a are reacted with trimethyl phosphate and methylimidazole to obtain a diastereomer mixture of remdesivir. In the end, optically pure remdesivir can be obtained through chiral resolution methods.[citation needed]
Research
Remdesivir was originally created and developed by Gilead Sciences in 2009, to treat hepatitis C and respiratory syncytial virus (RSV).[17][113] It did not work against hepatitis C or RSV,[17][113] but was then repurposed and studied as a potential treatment for Ebola virus disease and Marburg virus infections.[114][113] According to the Czech News Agency, this new line of research was carried out under the direction of scientist Tomáš Cihlář.[115] A collaboration of researchers from the Centers for Disease Control and Prevention (CDC) and Gilead Sciences subsequently discovered that remdesivir had antiviral activity in vitro against multiple filoviruses, pneumoviruses, paramyxoviruses, and coronaviruses.[116]
Preclinical and clinical research and development was done in collaboration between Gilead Sciences and various US government agencies and academic institutions.[32][117][118][119]
During the mid-2010s, the Mintz Levin law firm prosecuted various patent applications for remdesivir on behalf of Gilead Sciences before the United States Patent and Trademark Office (USPTO). The USPTO granted two patents on remdesivir to Gilead Sciences on 9 April 2019: one for filoviruses,[120] and one which covered both arenaviruses and coronaviruses.[121]
COVID-19
Remdesivir was approved for medical use in the United States in October 2020.[54][55][10][103] The U.S. Food and Drug Administration (FDA) approved remdesivir based on the agency's analysis of data from three randomized, controlled clinical trials that included participants hospitalized with mild-to-severe COVID‑19.[10][103] The FDA granted approval and reissued the revised EUA to Gilead Sciences Inc.[10] The FDA approved remdesivir based primarily on evidence from three clinical trials (NCT04280705, NCT04292899, and NCT04292730) of 2043 hospitalized participants with COVID-19.[103] The trials were conducted at 226 sites in 17 countries including the United States.[103]
One randomized, double-blind, placebo-controlled clinical trial (ACTT-1), conducted by the National Institute of Allergy and Infectious Diseases, evaluated how long it took for participants to recover from COVID‑19 within 29 days of being treated.[10] The trial looked at 1,062 hospitalized participants with mild, moderate and severe COVID‑19 who received remdesivir (n=541) or placebo (n=521), plus standard of care.[10] Recovery was defined as either being discharged from the hospital or being hospitalized but not requiring supplemental oxygen and no longer requiring ongoing medical care.[10] The median time to recovery from COVID‑19 was 10 days for the remdesivir group compared to 15 days for the placebo group, a statistically significant difference.[10] Overall, the odds of clinical improvement at Day 15 were also statistically significantly higher in the remdesivir group when compared to the placebo group.[10]
A second randomized, open-label multi-center clinical trial of hospitalized adult participants with moderate COVID‑19 compared treatment with remdesivir for five days (n=191) and treatment with remdesivir for 10 days (n=193) with the standard of care (n=200).[10] Researchers evaluated the clinical status of participants on Day 11.[10] Overall, the odds of a subject's COVID‑19 symptoms improving were statistically significantly higher in the five-day remdesivir group at Day 11 when compared to those receiving only standard of care.[10] The odds of improvement with the 10-day treatment group when compared to those receiving only standard of care were numerically favorable, but not statistically significantly different.[10]
A third separate, randomized, open-label multi-center clinical trial of hospitalized adult participants with severe COVID‑19 compared treatment with remdesivir for five days (n= 200) and treatment with remdesivir for 10 days (n= 197).[10] Researchers evaluated the clinical status of participants on Day 14.[10] Overall, the odds of a subject's COVID‑19 symptoms improving were similar for those in the five-day remdesivir group as those in the 10-day remdesivir group, and there were no statistically significant differences in recovery rates or mortality rates between the two groups.[10]
Remdesivir/baricitinib
In May 2020, the National Institute of Allergy and Infectious Diseases (NIAID) started the Adaptive COVID-19 Treatment Trial 2 (ACTT-2) to evaluate the safety and efficacy of a treatment regimen consisting of remdesivir plus baricitinib for treating hospitalized adults who have a laboratory-confirmed SARS-CoV-2 infection with evidence of lung involvement, including a need for supplemental oxygen, abnormal chest X-rays, or illness requiring mechanical ventilation.[122][123][124]
In November 2020, the U.S. Food and Drug Administration (FDA) issued an emergency use authorization (EUA) for the drug baricitinib, in combination with remdesivir, for the treatment of suspected or laboratory-confirmed COVID-19 in hospitalized people two years of age or older requiring supplemental oxygen, invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).[22] The data supporting the EUA for baricitinib combined with remdesivir are based on a randomized, double-blind, placebo-controlled clinical trial (ACTT-2), which was conducted by the National Institute of Allergy and Infectious Diseases (NIAID).[22] The EUA was issued to Eli Lilly and Company.[22]
Remdesivir/interferon beta-1a
In August 2020, the NIAID started the Adaptive COVID-19 Treatment Trial 3 (ACTT 3) to evaluate the safety and efficacy of a treatment regimen consisting of remdesivir plus interferon beta-1a for hospitalized adults who have a laboratory-confirmed SARS-CoV-2 infection with evidence of lung involvement, including a need for supplemental oxygen, abnormal chest X-rays, or illness requiring mechanical ventilation.[123][125]
Ebola
In October 2015, the United States Army Medical Research Institute of Infectious Diseases (USAMRIID) announced preclinical results that remdesivir had blocked the Ebola virus in Rhesus monkeys. Travis Warren, who has been a USAMRIID principal investigator since 2007, said that the "work is a result of the continuing collaboration between USAMRIID and Gilead Sciences".[126] The "initial screening" of the "Gilead Sciences compound library to find molecules with promising antiviral activity" was performed by scientists at the Centers for Disease Control and Prevention (CDC).[126] As a result of this work, it was recommended that remdesivir "should be further developed as a potential treatment."[114][unreliable medical source?][126]
Remdesivir was rapidly pushed through clinical trials due to the West African Ebola virus epidemic of 2013–2016, eventually being used in people with the disease. Preliminary results were promising; it was used in the emergency setting during the Kivu Ebola epidemic that started in 2018, along with further clinical trials, until August 2019, when Congolese health officials announced that it was significantly less effective than monoclonal antibody treatments such as ansuvimab and atoltivimab/maftivimab/odesivimab. The trials, however, established its safety profile.[127]
Veterinary uses
In 2019, GS-441524 was shown to have promise for treating feline infectious peritonitis caused by a coronavirus.[128] It has not been evaluated or approved by the US Food and Drug Administration (FDA) for the treatment of feline coronavirus or feline infectious peritonitis but has been available since 2019, through websites and social media as an unregulated black market substance.[129] Because GS-441524 is the main circulating metabolite of remdesivir and because GS-441524 has similar potency against SARS-Cov-2 in vitro, some researchers have argued for the direct administration of GS-441524 as a COVID‑19 treatment.[130]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Remdesivir Monograph for Professionals". Drugs.com. Archived from the original on 14 April 2021. Retrieved 13 July 2021.
- ↑ 2.0 2.1 "Fact Sheet for Patients And Parent/Caregivers Emergency Use Authorization (EUA) Of Remdesivir For Coronavirus Disease 2019 (COVID-19)" (PDF). U.S. Food and Drug Administration (FDA). Archived from the original on 16 May 2020. Retrieved 8 May 2020.
- ↑ 3.0 3.1 "Veklury Australian prescription medicine decision summary". Therapeutic Goods Administration (TGA). 13 July 2020. Archived from the original on 13 August 2020. Retrieved 16 August 2020.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 "Veklury EPAR". European Medicines Agency (EMA). 23 June 2020. Archived from the original on 18 March 2021. Retrieved 6 July 2020.
- ↑ "Summary for ARTG Entry:338419 Veklury remdesivir 100 mg powder for injection vial". Therapeutic Goods Administration (TGA). Retrieved 16 August 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ 6.0 6.1 "Veklury Product information". Health Canada. 25 April 2012. Archived from the original on 26 October 2020. Retrieved 22 October 2020.
- ↑ 7.0 7.1 7.2 "Veklury 100 mg concentrate for solution for infusion – Summary of Product Characteristics (SmPC)". (emc). 6 July 2020. Archived from the original on 24 November 2020. Retrieved 4 October 2020.
- ↑ "Veklury 100 mg powder for concentrate for solution for infusion – Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 8 October 2020. Retrieved 4 October 2020.
- ↑ 9.0 9.1 "Remdesivir injection Remdesivir injection, powder, lyophilized, for solution". DailyMed. 1 September 2020. Archived from the original on 26 September 2020. Retrieved 25 September 2020.
- ↑ 10.00 10.01 10.02 10.03 10.04 10.05 10.06 10.07 10.08 10.09 10.10 10.11 10.12 10.13 10.14 10.15 10.16 10.17 10.18 10.19 10.20 10.21 10.22 "FDA Approves First Treatment for COVID-19". U.S. Food and Drug Administration (FDA) (Press release). 22 October 2020. Archived from the original on 22 October 2020. Retrieved 22 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 11.0 11.1 11.2 "Statement on Therapies for High-Risk, Nonhospitalized Patients". COVID-19 Treatment Guidelines. Archived from the original on 7 January 2022. Retrieved 7 January 2022.
- ↑ 12.0 12.1 World Health Organization (2020). Therapeutics and COVID-19: living guideline, 20 November 2020. World Health Organization (WHO) (Report). hdl:10665/336729. WHO/2019-nCov/remdesivir/2020.1.
- ↑ "Remdesivir (Veklury) Use During Pregnancy". Drugs.com. Archived from the original on 16 April 2021. Retrieved 13 July 2021.
- ↑ 14.0 14.1 14.2 Yan VC, Muller FL (14 May 2020). "Gilead should ditch remdesivir and focus on its simpler and safer ancestor". Stat. Boston Globe Media Partners. Archived from the original on 12 June 2021. Retrieved 11 July 2021.
- ↑ 15.0 15.1 "New Drug Therapy Approvals 2020". U.S. Food and Drug Administration (FDA). 31 December 2020. Archived from the original on 18 January 2021. Retrieved 17 January 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 16.0 16.1 Scavone C, Brusco S, Bertini M, Sportiello L, Rafaniello C, Zoccoli A, et al. (April 2020). "Current pharmacological treatments for COVID-19: What's next?". British Journal of Pharmacology. 177 (21): 4813–4824. doi:10.1111/bph.15072. eISSN 1476-5381. PMC 7264618. PMID 32329520.
- ↑ 17.0 17.1 17.2 17.3 Stephens B (18 April 2020). "The Story of Remdesivir". The New York Times. p. A23. Archived from the original on 22 May 2020. Retrieved 11 May 2020.
- ↑ Chamary, J. V. "The Strange Story Of Remdesivir, A Covid Drug That Doesn't Work". Forbes. Archived from the original on 29 August 2021. Retrieved 13 July 2021.
- ↑ "6.1. Coronavirus". British National Formulary (BNF) (82 ed.). BMJ Group and the Pharmaceutical Press. September 2021 – March 2022. pp. 665–667. ISBN 978-0-85711-413-6.
{{cite book}}: CS1 maint: date format (link) - ↑ Lamontagne F, Agoritsas T, Macdonald H, Leo YS, Diaz J, Agarwal A, et al. (September 2020). "A living WHO guideline on drugs for covid-19". BMJ. 370: m3379. doi:10.1136/bmj.m3379. PMID 32887691.
- Lay summary in: "Remdesivir: don't use drug Trump took for Covid-19, WHO says".
{{cite journal}}: Cite journal requires|journal=(help)
- Lay summary in: "Remdesivir: don't use drug Trump took for Covid-19, WHO says".
- ↑ "Update on remdesivir". European Medicines Agency (EMA). 20 November 2020. Archived from the original on 20 November 2020. Retrieved 20 November 2020.
- ↑ 22.0 22.1 22.2 22.3 22.4 22.5 22.6 "Coronavirus (COVID-19) Update: FDA Authorizes Drug Combination for Treatment of COVID-19". U.S. Food and Drug Administration (Press release). 19 November 2020. Archived from the original on 19 November 2020. Retrieved 19 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ Wilson J (30 July 2020). "Remdesivir Gets Lukewarm Endorsement From Experts in Covid Fight". Bloomberg. Archived from the original on 4 August 2020. Retrieved 31 July 2020.
- ↑ Mahase E (July 2020). "Covid-19: Remdesivir probably reduces recovery time, but evidence is uncertain, panel finds". BMJ. 370: m3049. doi:10.1136/bmj.m3049. PMID 32732277.
- ↑ Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. (May 2020). "Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial". Lancet. 395 (10236): 1569–1578. doi:10.1016/S0140-6736(20)31022-9. PMC 7190303. PMID 32423584.
- ↑ Mehta N, Mazer-Amirshahi M, Alkindi N, Pourmand A (July 2020). "Pharmacotherapy in COVID-19; A narrative review for emergency providers". The American Journal of Emergency Medicine. 38 (7): 1488–1493. doi:10.1016/j.ajem.2020.04.035. eISSN 0735-6757. PMC 7158837. PMID 32336586.
- ↑ 27.0 27.1 27.2 27.3 27.4 27.5 27.6 27.7 27.8 "Frequently Asked Questions on the Emergency Use Authorization for Remdesivir for Certain Hospitalized COVID‐19 Patients" (PDF). U.S. Food and Drug Administration (FDA). 1 May 2020. Archived from the original on 6 May 2020. Retrieved 1 May 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Summary on Compassionate Use: Remdesivir Gilead" (PDF). European Medicines Agency (EMA). 3 April 2020. Archived (PDF) from the original on 11 April 2020. Retrieved 1 May 2020.
- ↑ 29.0 29.1 29.2 29.3 29.4 "FDA EUA Remdesivir Fact Sheet for Health Care Providers" (PDF). U.S. Food and Drug Administration (FDA). 1 May 2020. Archived from the original on 12 May 2020. Retrieved 1 May 2020.
- Lay summary in: https://www.fda.gov/media/137565/download.
{{cite web}}: Missing or empty|title=(help) This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- Lay summary in: https://www.fda.gov/media/137565/download.
- ↑ "COVID-19 interactions". University of Liverpool. Archived from the original on 28 April 2020. Retrieved 28 April 2020.
- ↑ "Coronavirus (COVID-19) Update: FDA Warns of Newly Discovered Potential Drug Interaction That May Reduce Effectiveness of a COVID-19 Treatment Authorized for Emergency Use" (Press release). U.S. Food and Drug Administration (FDA). 15 June 2020. Archived from the original on 13 October 2020. Retrieved 15 June 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 32.0 32.1 32.2 32.3 Eastman RT, Roth JS, Brimacombe KR, Simeonov A, Shen M, Patnaik S, Hall MD (May 2020). "Remdesivir: A Review of Its Discovery and Development Leading to Emergency Use Authorization for Treatment of COVID-19". ACS Central Science. 6 (5): 672–683. doi:10.1021/acscentsci.0c00489. PMC 7202249. PMID 32483554.
- ↑ Sheahan TP, Sims AC, Graham RL, Menachery VD, Gralinski LE, Case JB, et al. (June 2017). "Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses". Science Translational Medicine. 9 (396): eaal3653. doi:10.1126/scitranslmed.aal3653. PMC 5567817. PMID 28659436.
- ↑ 34.0 34.1 34.2 Gordon CJ, Tchesnokov EP, Woolner E, Perry JK, Feng JY, Porter DP, Götte M (May 2020). "Remdesivir is a direct-acting antiviral that inhibits RNA-dependent RNA polymerase from severe acute respiratory syndrome coronavirus 2 with high potency". The Journal of Biological Chemistry. 295 (20): 6785–6797. doi:10.1074/jbc.RA120.013679. PMC 7242698. PMID 32284326.
- ↑ Kausar, Shamaila; Said Khan, Fahad; Ishaq Mujeeb Ur Rehman, Muhammad; Akram, Muhammad; Riaz, Muhammad; Rasool, Ghulam; Hamid Khan, Abdul; Saleem, Iqra; Shamim, Saba; Malik, Arif (January 2021). "A review: Mechanism of action of antiviral drugs". International Journal of Immunopathology and Pharmacology. 35: 205873842110026. doi:10.1177/20587384211002621. ISSN 2058-7384.
- ↑ Cho A, Saunders OL, Butler T, Zhang L, Xu J, Vela JE, et al. (April 2012). "Synthesis and antiviral activity of a series of 1'-substituted 4-aza-7,9-dideazaadenosine C-nucleosides". Bioorganic & Medicinal Chemistry Letters. 22 (8): 2705–2707. doi:10.1016/j.bmcl.2012.02.105. PMC 7126871. PMID 22446091.
- ↑ Ferner RE, Aronson JK (April 2020). "Remdesivir in covid-19". BMJ. 369: m1610. doi:10.1136/bmj.m1610. PMID 32321732. Archived from the original on 2021-03-08. Retrieved 2021-07-11.
- ↑ Tchesnokov EP, Feng JY, Porter DP, Götte M (April 2019). "Mechanism of Inhibition of Ebola Virus RNA-Dependent RNA Polymerase by Remdesivir". Viruses. 11 (4): 326. doi:10.3390/v11040326. PMC 6520719. PMID 30987343.
- ↑ Gordon CJ, Tchesnokov EP, Feng JY, Porter DP, Götte M (April 2020). "The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus". The Journal of Biological Chemistry. 295 (15): 4773–4779. doi:10.1074/jbc.AC120.013056. PMC 7152756. PMID 32094225.
- ↑ Yan VC, Muller FL (July 2020). "Advantages of the Parent Nucleoside GS-441524 over Remdesivir for Covid-19 Treatment". ACS Medicinal Chemistry Letters. 11 (7): 1361–1366. doi:10.1021/acsmedchemlett.0c00316. PMC 7315846. PMID 32665809. S2CID 220056568.
- ↑ Chiotos K, Hayes M, Kimberlin DW, Jones SB, James SH, Pinninti SG, et al. (April 2020). "Multicenter initial guidance on use of antivirals for children with COVID-19/SARS-CoV-2". Journal of the Pediatric Infectious Diseases Society. 9 (6): 701–715. doi:10.1093/jpids/piaa045. PMC 7188128. PMID 32318706.
- ↑ World Health Organization (2017). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 78". WHO Drug Information. 31 (3): 549. hdl:10665/330961.
- ↑ "Pipeline". Gilead Sciences. 27 February 2020. Archived from the original on 10 April 2013. Retrieved 17 April 2020.
- ↑ 44.0 44.1 "An Open Letter from Daniel O'Day, Chairman & CEO, Gilead Sciences" (Press release). Gilead Sciences. 29 June 2020. Archived from the original on 29 June 2020. Retrieved 29 June 2020.
- ↑ Hill A, Wang J, Levi J, Heath K, Fortunak J (April 2020). "Minimum costs to manufacture new treatments for COVID-19". Journal of Virus Eradication. 6 (2): 61–69. doi:10.1016/S2055-6640(20)30018-2. PMC 7331548. PMID 32405423.
- ↑ "U.S. Food and Drug Administration Approves Gilead's Antiviral Veklury (remdesivir) for Treatment of COVID-19". Gilead Sciences, Inc. 22 October 2020. Archived from the original on 23 October 2020. Retrieved 23 October 2020.
- ↑ "India approves emergency use of remdesivir to treat Covid-19 patients". The Times of India. Gurgaon, Haryana, India: Times Internet. Reuters. 2 June 2020. Archived from the original on 4 June 2020. Retrieved 2 June 2020.
- ↑ "Singapore approves remdesivir drug for emergency COVID-19 treatment". Reuters. 10 June 2020. Archived from the original on 10 June 2020. Retrieved 10 June 2020.
- ↑ 49.0 49.1 "Gilead Announces Approval of Veklury (remdesivir) in Japan for Patients With Severe COVID-19" (Press release). Gilead Sciences. 7 May 2020. Archived from the original on 26 June 2020. Retrieved 25 June 2020 – via Business Wire.
- ↑ "Remdesivir EUA Letter of Authorization" (PDF). U.S. Food and Drug Administration (FDA). 1 May 2020. Archived from the original on 2 May 2020. Retrieved 1 May 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 51.0 51.1 "Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment" (Press release). U.S. Food and Drug Administration (FDA). 1 May 2020. Archived from the original on 11 October 2020. Retrieved 1 May 2020.
- ↑ 52.0 52.1 "Japan approves remdesivir for COVID-19 despite uncertainties". The Asahi Shimbun. 8 May 2020. Archived from the original on 25 May 2020. Retrieved 9 May 2020.
- ↑ 53.0 53.1 "Australia's first COVID treatment approved". Therapeutic Goods Administration (TGA) (Press release). 10 July 2020. Archived from the original on 11 October 2020. Retrieved 11 July 2020.
- ↑ 54.0 54.1 "Veklury: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 23 October 2020. Retrieved 22 October 2020.
- ↑ 55.0 55.1 Veklury: Summary Review (PDF). U.S. Food and Drug Administration (FDA) (Report). Archived (PDF) from the original on 23 October 2020. Retrieved 22 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "EMA starts evaluating use of Veklury in COVID-19 patients not requiring supplemental oxygen". European Medicines Agency (EMA) (Press release). 23 February 2021. Archived from the original on 23 February 2021. Retrieved 23 February 2021.
- ↑ "Remdesivir approved by the Therapeutic Goods Administration for severe coronavirus cases". Australian Broadcasting Corporation. 11 July 2020. Archived from the original on 11 July 2020. Retrieved 11 July 2020.
- ↑ Hitch G (1 July 2020). "Australia has enough coronavirus drug remdesivir thanks to early supply donation, Health Minister says". Australian Broadcasting Corporation. Sydney, Australia. Archived from the original on 11 July 2020. Retrieved 11 July 2020.
- ↑ 59.0 59.1 59.2 "Coronavirus disease (COVID-19): For health professionals". Public Health Agency of Canada. 11 April 2020. Archived from the original on 11 April 2020. Retrieved 12 April 2020.
Gilead is transitioning the provision of emergency access to remdesivir from individual compassionate use via Health Canada's Special Access Program requests to access through clinical trials.
- ↑ Blackwell T (1 May 2020). "Canadian experts don't see Remdesivir as a COVID-19 killer: 'This is not a silver bullet'". National Post. Retrieved 11 July 2020.
- ↑ Weikle B (28 July 2020). "Health Canada authorizes drug remdesivir for severe COVID-19 cases". CBC News. Archived from the original on 11 October 2020. Retrieved 10 October 2020.
- ↑ Grant K, Howlett K (22 September 2020). "Cases could spike sharply if Canadian epidemic stays on current course, Tam warns". The Globe and Mail. Archived from the original on 2 October 2020. Retrieved 10 October 2020.
- ↑ Kury de Castillo C (8 October 2020). "Calgary man dies after contracting COVID-19 at Foothills hospital; family seeks remdesivir for daughter on ventilator". Global News. Global Television Network. Archived from the original on 10 October 2020. Retrieved 13 October 2020.
- ↑ "Opatření MZ ČR – povolení LP Remdesivir" [Measures of the Ministry of Health of the Czech Republic – LP Remdesivir permit] (PDF). www.mzcr.cz (in čeština). 17 March 2020. Archived (PDF) from the original on 4 August 2020. Retrieved 24 March 2020. HTML Version Opatření MZ ČR - povolení LP REMDESIVIR at the Wayback Machine (archived 2021-03-08)(Positional parameters ignored)
- ↑ "EU/3/16/1615". European Medicines Agency (EMA). Archived from the original on 9 July 2020. Retrieved 7 July 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "EMA provides recommendations on compassionate use of remdesivir for COVID-19". European Medicines Agency (EMA). 3 April 2020. Archived from the original on 19 May 2020. Retrieved 3 May 2020.
- ↑ 67.0 67.1 67.2 67.3 67.4 "EMA recommends expanding remdesivir compassionate use to patients not on mechanical ventilation". European Medicines Agency (EMA). 11 May 2020. Archived from the original on 17 May 2020. Retrieved 11 May 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Adaptive COVID-19 Treatment Trial (ACTT)". ClinicalTrials.gov. 21 February 2020. Archived from the original on 20 May 2020. Retrieved 11 May 2020.
- ↑ "NIH Clinical Trial Shows Remdesivir Accelerates Recovery from Advanced COVID-19" (Press release). National Institute of Allergy and Infectious Diseases. 29 April 2020. Archived from the original on 30 April 2020. Retrieved 29 April 2020.
- ↑ "Veklury". Union Register of medicinal products. Archived from the original on 22 January 2021. Retrieved 14 January 2021.
- ↑ Guarascio F (29 July 2020). "EU buys remdesivir to treat 30,000 COVID patients, seeks more". Reuters. Archived from the original on 31 July 2020. Retrieved 31 July 2020.
- ↑ Saigol L (8 October 2020). "Gilead Sciences agrees to sell to Europe up to 500,000 courses of remdesivir". MarketWatch. Archived from the original on 8 October 2020. Retrieved 9 October 2020.
- ↑ 73.0 73.1 Guarascio F (13 October 2020). "EU makes one billion-euro bet on Gilead's COVID drug before trial results". Reuters. Archived from the original on 23 November 2020. Retrieved 11 July 2021.
- ↑ Increasing the production of Remedisivir anti-corona drug by the executive staff of Farman Imam افزایش تولید داروی ضدکرونای رمدیسیویر توسط ستاد اجرایی فرمان امام (ره) - ایسنا at the Wayback Machine (archived 2021-07-25) isna.ir
- ↑ "Mexico will not follow FDA in approving Gilead's COVID-19 drug". Reuters. October 23, 2020. Archived from the original on October 30, 2020. Retrieved October 25, 2020.
- ↑ "COFEPRIS emite autorización para uso de emergencia de Remdesivir". Cofepris. March 12, 2020. Archived from the original on March 12, 2021. Retrieved March 12, 2020.
- ↑ Naftulin J (20 March 2020). "The FDA is allowing two drugs to be used for 'compassionate use' to treat the coronavirus. Here's what that means". Business Insider. New York City: Springer. Archived from the original on 1 May 2020. Retrieved 11 July 2021.
- ↑ Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, et al. (June 2020). "Compassionate Use of Remdesivir for Patients with Severe Covid-19". N Engl J Med. 382 (24): 2327–2336. doi:10.1056/NEJMoa2007016. PMC 7169476. PMID 32275812.
- ↑ "Coronavirus COVID-19 (SARS-CoV-2)". Johns Hopkins ABX Guide. Archived from the original on 11 April 2020. Retrieved 12 April 2020.
Remdesivir: Likely the most promising drug.
- ↑ Cerullo M (23 March 2020). "Gilead suspends emergency access to experimental coronavirus drug remdesivir". CBS News. Archived from the original on 21 April 2020. Retrieved 23 March 2020.
- ↑ 81.0 81.1 "U.S. Emergency Approval Broadens Use of Gilead's COVID-19 Drug Remdesivir". The New York Times. Reuters. 1 May 2020. Archived from the original on 2 May 2020. Retrieved 1 May 2020.
- ↑ Holland S, Beasley D (4 May 2020). "U.S. emergency approval broadens use of Gilead's COVID-19 drug remdesivir". Reuters. Archived from the original on 11 May 2020. Retrieved 8 May 2020.
- ↑ Jarvis LM (20 April 2020). "Scaling up remdesivir amid the coronavirus crisis". Chemical and Engineering News. Archived from the original on 5 May 2020. Retrieved 11 July 2021.
- ↑ Rowland C (10 April 2020). "Gilead's experimental drug remdesivir shows 'hopeful' signs in small group of coronavirus patients". The Washington Post. Archived from the original on 7 May 2020. Retrieved 8 May 2020.
- ↑ Kolata G (8 May 2020). "Haphazard Rollout of Coronavirus Drug Frustrates Doctors". The New York Times. Archived from the original on 8 May 2020. Retrieved 8 May 2020.
- ↑ Swan J (8 May 2020). "Scoop: Trump officials' dysfunction harms delivery of coronavirus drug". Axios. Archived from the original on 12 May 2020. Retrieved 11 May 2020.
- ↑ 87.0 87.1 87.2 Abutaleb Y, Dawsey J, Sun LH, McGinley L (28 May 2020). "Administration initially dispensed scarce covid-19 drug to some hospitals that didn't need it". The Washington Post. Archived from the original on 28 May 2020. Retrieved 29 May 2020.
- ↑ 88.0 88.1 88.2 Facher L (9 May 2020). "Trump administration announces plan to distribute Covid-19 drug amid concerns over allocation". Stat. Boston, Massachusetts: Boston Globe Media. Archived from the original on 10 May 2020. Retrieved 11 May 2020.
- ↑ Branswell H (19 May 2020). "Gilead ups its donation of the Covid-19 drug remdesivir". Stat. Boston, Massachusetts: Boston Globe Media Partners. Archived from the original on 20 December 2020. Retrieved 19 May 2020.}
- ↑ Kolata G (29 June 2020). "Remdesivir, the First Coronavirus Drug, Gets a Price Tag". The New York Times. Archived from the original on 29 June 2020. Retrieved 29 June 2020.
- ↑ "Trump Administration Secures New Supplies of Remdesivir for the United States" (Press release). U.S. Department of Health and Human Services (HHS). 29 June 2020. Archived from the original on 29 June 2020. Retrieved 29 June 2020.
- ↑ Boseley S (30 June 2020). "US buys up world stock of key Covid-19 drug". The Guardian. Archived from the original on 23 June 2021. Retrieved 11 July 2021.
- ↑ Baragona S (29 June 2020). "US Procures Almost Entire Supply of COVID-19 Drug". Voice of America. Washington, D.C.: U.S. Agency for Global Media. Archived from the original on 30 June 2020. Retrieved 30 June 2020.
- ↑ Davey M (1 July 2020). "Gilead donates Covid-19 drug remdesivir to Australia's medical stockpile after US buys up supply". The Guardian. Archived from the original on 1 July 2020. Retrieved 1 July 2020.
- ↑ Rinke A (1 July 2020). "Germany has for now enough remdesivir for COVID-19 therapy: govt". Reuters. Archived from the original on 1 July 2020. Retrieved 1 July 2020.
- ↑ Stout A, Mason J (1 July 2020). "UK emergency remdesivir supplies adequate to treat COVID-19, official says". Reuters. Archived from the original on 9 March 2021. Retrieved 11 July 2021.
- ↑ "COVID-19 Update: FDA Broadens Emergency Use Authorization for Veklury (remdesivir) to Include All Hospitalized Patients for Treatment of COVID-19". U.S. Food and Drug Administration (FDA) (Press release). 28 August 2020. Archived from the original on 29 August 2020. Retrieved 28 August 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Gilead's Investigational Antiviral Veklury (Remdesivir) Receives U.S. Food and Drug Administration Emergency Use Authorization for the Treatment of Patients With Moderate COVID-19" (Press release). Gilead Sciences. 28 August 2020. Archived from the original on 3 September 2020. Retrieved 28 August 2020 – via Business Wire.
- ↑ Walker J (October 1, 2020). "All Remdesivir Supplies to Be Distributed in U.S. by Maker Gilead Sciences". The Wall Street Journal. Archived from the original on August 29, 2021. Retrieved July 11, 2021.
- ↑ "Gilead Sciences Update on Supply and Distribution of Veklury (remdesivir) in the United States". Gilead Sciences, Inc. (Press release). 1 October 2020. Archived from the original on 29 August 2021. Retrieved 4 October 2020.
- ↑ 101.0 101.1 "Veklury (remdesivir) Now Available Directly from Distributor". U.S. Department of Health & Human Services (HHS) (Press release). 1 October 2020. Archived from the original on 27 October 2020. Retrieved 28 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "FDA's approval of Veklury (remdesivir) for the treatment of COVID-19". U.S. Food and Drug Administration (FDA). 22 October 2020. Archived from the original on 22 October 2020. Retrieved 23 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 103.0 103.1 103.2 103.3 103.4 "Drug Trials Snapshots: Veklury". U.S. Food and Drug Administration (FDA). 27 October 2020. Archived from the original on 14 July 2021. Retrieved 11 July 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "FDA Says COVID-19 Treatment Remdesivir Shouldn't Be Compounded". FDANews. 11 February 2021. Archived from the original on 11 February 2021. Retrieved 11 February 2021.
- ↑ 105.0 105.1 105.2 105.3 105.4 105.5 105.6 Langreth R (14 May 2020). "All Eyes on Gilead". Bloomberg Businessweek. Archived from the original on 21 May 2020. Retrieved 14 May 2020.
- ↑ 106.0 106.1 106.2 Pipkin J, Antle V, Garcia-Fandiño R (June 2020). "FORMULATION FORUM – Application of Captisol Technology to Enable the Formulation of Remdesivir in Treating COVID-19". Drug Development & Delivery. Archived from the original on 20 July 2020. Retrieved 20 July 2020.
- ↑ 107.0 107.1 Silverman E (12 May 2020). "Gilead signs deals for generic companies to make and sell remdesivir". Stat. Archived from the original on 16 May 2020. Retrieved 12 May 2020.
- ↑ "India, Pakistan to make drug to 'fight coronavirus'". BBC News Online. 14 May 2020. Archived from the original on 28 May 2020. Retrieved 23 May 2020.
- ↑ "Voluntary Licensing Agreements for Remdesivir". Gilead Sciences, Inc. 24 October 2020. Archived from the original on 11 July 2021. Retrieved 11 July 2021.
- ↑ Rajagopal D (23 June 2020). "Cipla, Hetero receive drug controller's emergency approval for Remdesivir for severe Covid-19 patients". The Economic Times. Mumbai, India: Bennett, Coleman & Co. Ltd. Archived from the original on 6 March 2021. Retrieved 1 July 2020.
- ↑ US 9724360, Chun BK, Clarke MO, Doerffler E, Hui HC, Jordan R, Mackman RL, Parrish JP, Ray AS, Siegel D, "Methods for treating Filoviridae virus infections", published 5 May 2016, issued 8 August 2017, assigned to Gilead Sciences Inc.
- ↑ WO 2017184668, Clarke MO, Jordan R, Mackman RL, Ray AS, Siegel D, "Preparation of amino acid-containing nucleosides for treating flaviviridae virus infections", published 26 October 2017, assigned to Glead Sciences Inc
- ↑ 113.0 113.1 113.2 "Final report confirms remdesivir benefits for COVID-19". National Institutes of Health (NIH) (Press release). 19 October 2020. Archived from the original on 22 October 2020. Retrieved 23 October 2020.
- ↑ 114.0 114.1 Warren TK, Jordan R, Lo MK, Ray AS, Mackman RL, Soloveva V, et al. (March 2016). "Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys". Nature. 531 (7594): 381–385. Bibcode:2016Natur.531..381W. doi:10.1038/nature17180. PMC 5551389. PMID 26934220.
- ↑ Czech News Agency, "Did Czech scientists create the cure for coronavirus?" Did Czech scientists create the cure for coronavirus? at the Wayback Machine (archived 2020-05-13), Aktuálně.cz, 5 February 2020.
- ↑ Lo MK, Jordan R, Arvey A, Sudhamsu J, Shrivastava-Ranjan P, Hotard AL, et al. (March 2017). "GS-5734 and its parent nucleoside analog inhibit Filo-, Pneumo-, and Paramyxoviruses". Scientific Reports. 7: 43395. Bibcode:2017NatSR...743395L. doi:10.1038/srep43395. PMC 5338263. PMID 28262699.
- ↑ Silverman E (8 May 2020). "U.S. government contributed research to a Gilead remdesivir patent – but didn't get credit". Stat. Archived from the original on 28 May 2020. Retrieved 28 May 2020.
- ↑ Ardizzone K (20 March 2020). "Role of the Federal Government in the Development of Remdesivir" (PDF). Knowledge Ecology International. Archived (PDF) from the original on 27 May 2020. Retrieved 28 May 2020.
- ↑ "Investigational compound remdesivir, developed by UAB and NIH researchers, being used for treatment of novel coronavirus". UAB News. Archived from the original on 26 May 2020. Retrieved 28 May 2020.
- ↑ US 10251898, Chun BK, Clarke MO, Doerffler E, Hui HC, Jordan R, Mackman RL, Parrish JP, Ray AS, Siegel D, "Methods for treating Filoviridae virus infections", published 1 November 2018, issued 9 April 2019, assigned to Gilead Sciences, Inc.
- ↑ US 10251904, Clarke MO, Feng JY, Jordan R, Mackman RL, Ray AS, Siegel D, "Methods for treating arenaviridae and coronaviridae virus infections", published 16 March 2017, issued 9 April 2019, assigned to Gilead Sciences, Inc.
- ↑ "NIH Clinical Trial Testing Antiviral Remdesivir Plus Anti-Inflammatory Drug Baricitinib for COVID-19 Begins". National Institute of Allergy and Infectious Diseases (NIAID) (Press release). 8 May 2020. Archived from the original on 6 October 2020. Retrieved 5 August 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 123.0 123.1 "NIH Clinical Trial Testing Remdesivir Plus Interferon Beta-1a for COVID-19 Treatment Begins". National Institute of Allergy and Infectious Diseases (NIAID) (Press release). 30 July 2020. Archived from the original on 6 October 2020. Retrieved 5 August 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Adaptive COVID-19 Treatment Trial 2 (ACTT-2)". ClinicalTrials.gov. 26 May 2020. Archived from the original on 5 August 2020. Retrieved 6 August 2020.
- ↑ "Adaptive COVID-19 Treatment Trial 3 (ACTT-3)". ClinicalTrials.gov. 30 July 2020. Archived from the original on 6 October 2020. Retrieved 5 August 2020.
- ↑ 126.0 126.1 126.2 Antiviral Compound Provides Full Protection from Ebola Virus in Nonhuman Primates (PDF) (Report). San Diego, California: United States Army Medical Research Institute of Infectious Diseases (USAMRIID). 9 October 2015. Archived (PDF) from the original on 24 December 2016. Retrieved 15 March 2020.
- ↑ Cao YC, Deng QX, Dai SX (April 2020). "Remdesivir for severe acute respiratory syndrome coronavirus 2 causing COVID-19: An evaluation of the evidence". Travel Medicine and Infectious Disease. 35: 101647. doi:10.1016/j.tmaid.2020.101647. PMC 7151266. PMID 32247927.
- ↑ Pedersen NC, Perron M, Bannasch M, Montgomery E, Murakami E, Liepnieks M, et al. (April 2019). "Efficacy and safety of the nucleoside analog GS-441524 for treatment of cats with naturally occurring feline infectious peritonitis". Journal of Feline Medicine and Surgery. 21 (4): 271–281. doi:10.1177/1098612X19825701. PMC 6435921. PMID 30755068.
- ↑ Pedersen NC (18 June 2019). "Black market production and sale of GS-441524 and GC376" (PDF). Davis, California: Feline Infectious Peritonitis Therapeutics/Clinical Trials Team, UC Davis. Archived (PDF) from the original on 25 March 2020. Retrieved 14 April 2020.
- ↑ Westgate J (7 May 2020). "Vet science 'being ignored' in quest for COVID-19 drug". vettimes.co.uk. Archived from the original on 4 December 2020. Retrieved 11 July 2021.
Further reading
- Goldman JD, Lye DC, Hui DS, Marks KM, Bruno R, Montejano R, et al. (November 2020). "Remdesivir for 5 or 10 Days in Patients with Severe Covid-19". The New England Journal of Medicine. 383 (19): 1827–37. doi:10.1056/NEJMoa2015301. PMID 32459919.
- Australian Public Assessment Report for Remdesivir (Report). Therapeutic Goods Administration (TGA). Archived from the original on 2020-10-26. Retrieved 2021-07-11.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Pages with non-numeric formatnum arguments
- CS1 maint: url-status
- Wikipedia articles incorporating the PD-notice template
- CS1 maint: date format
- CS1 errors: missing periodical
- CS1 errors: missing title
- CS1 errors: bare URL
- CS1 čeština-language sources (cs)
- Webarchive template wayback links
- Webarchive template warnings
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Articles containing unverified chemical infoboxes
- All articles with unsourced statements
- Articles with unsourced statements from July 2020
- Articles with invalid date parameter in template
- All articles lacking reliable references
- Articles lacking reliable references from May 2020
- Drugs not assigned an ATC code
- Articles using infobox templates with no data rows
- Anti–RNA virus drugs
- Antiviral drugs
- COVID-19 drug development
- Experimental drugs
- Gilead Sciences
- Heterocyclic compounds (2 rings)
- Nitriles
- Nitrogen heterocycles
- Nucleotides
- Orphan drugs
- Phenol esters
- Phosphoramidates
- RTT