Plerixafor
 | |
 | |
| Names | |
|---|---|
| Trade names | Mozobil |
| Other names | JM 3100, AMD3100 |
| |
| Clinical data | |
| Drug class | CXCR4 chemokine receptor blocker[1] |
| Main uses | Stimulate release of stem cells from the bone marrow[1] |
| Side effects | Diarrhea, nausea, pain at the site of injection[1] |
| Pregnancy category |
|
| Routes of use | Subcutaneous injection |
| Typical dose | 0.24 mg/kg up to 40 mg[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a609018 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Protein binding | Up to 58% |
| Metabolism | None |
| Elimination half-life | 3–5 hours |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C28H54N8 |
| Molar mass | 502.796 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Plerixafor, sold under the brand name Mozobil, is a medication used to stimulate release of stem cells from the bone marrow into the blood to be collected for later transplanted back.[1] It is used together with a granulocyte-colony stimulating factor (G-CSF).[1] It is given by injection under the skin.[1]
Common side effects include diarrhea, nausea, and pain at the site of injection.[1] Other side effects may include low platelets, splenic rupture, and release of leukemia cells into the blood.[2] Use in pregnancy may harm the baby.[2] It works by blocking the activity of the CXCR4 chemokine receptor.[1]
Plerixafor was approved for medical use in the United States in 2008 and Europe in 2009.[2][1] In the United Kingdom a vial of 24 mg costs the NHS about £4,900 as of 2021.[3] In the United States this amount costs about 9,300 USD.[4]
Medical uses
Peripheral blood stem cell mobilization, which is important as a source of hematopoietic stem cells for transplantation, is generally performed using granulocyte colony-stimulating factor (G-CSF), but is ineffective in around 15 to 20% of patients. Combination of G-CSF with plerixafor increases the percentage of persons that respond to the therapy and produce enough stem cells for transplantation.[5] The drug is approved for patients with lymphoma and multiple myeloma.[6]
Dosage
It is given at a dose of 0.24 mg/kg up to a dose of 40 mg once daily for up to four days.[2]
Side effects
Nausea, diarrhea and local reactions were observed in over 10% of patients. Other problems with digestion and general symptoms like dizziness, headache, and muscular pain are also relatively common; they were found in more than 1% of patients. Allergies occur in less than 1% of cases. Most adverse effects in clinical trials were mild and transient.[6][7]
The European Medicines Agency has listed a number of safety concerns to be evaluated on a post-marketing basis, most notably the theoretical possibilities of spleen rupture and tumor cell mobilisation. The first concern has been raised because splenomegaly was observed in animal studies, and G-CSF can cause spleen rupture in rare cases. Mobilisation of tumor cells has occurred in patients with leukaemia treated with plerixafor.[8]
Pregnancy and lactation
Studies in pregnant animals have shown teratogenic effects. Plerixafor is therefore contraindicated in pregnant women except in critical cases. Fertile women are required to use contraception. It is not known whether the drug is secreted into the breast milk. Breast feeding should be discontinued during therapy.[6]
Interactions
No interaction studies have been conducted. The fact that plerixafor does not interact with the cytochrome system indicates a low potential for interactions with other drugs.[6]
Pharmacology
Mechanism of action
In the form of its zinc complex, plerixafor acts as an antagonist (or perhaps more accurately a partial agonist) of the alpha chemokine receptor CXCR4 and an allosteric agonist of CXCR7.[9] The CXCR4 alpha-chemokine receptor and one of its ligands, SDF-1, are important in hematopoietic stem cell homing to the bone marrow and in hematopoietic stem cell quiescence. The in vivo effect of plerixafor with regard to ubiquitin, the alternative endogenous ligand of CXCR4, is unknown. Plerixafor has been found to be a strong inducer of mobilization of hematopoietic stem cells from the bone marrow to the bloodstream as peripheral blood stem cells.[10] Additionally, plerixafor inhibits CD20 expression on B cells by interfering with CXCR4/SDF1 axis that regulates its expression.[citation needed]
Pharmacokinetics
Following subcutaneous injection, plerixafor is absorbed quickly and peak concentrations are reached after 30 to 60 minutes. Up to 58% are bound to plasma proteins, the rest mostly resides in extravascular compartments. The drug is not metabolized in significant amounts; no interaction with the cytochrome P450 enzymes or P-glycoproteins has been found. Plasma half life is 3 to 5 hours. Plerixafor is excreted via the kidneys, with 70% of the drug being excreted within 24 hours.[6]
Chemistry
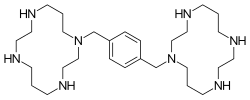
Plerixafor is a macrocyclic compound and a bicyclam derivative, the cyclam rings being linked at the amine nitrogen atoms by a 1,4-xylyl spacer.[5] It is a strong base; all eight nitrogen atoms accept protons readily. The two macrocyclic rings form chelate complexes with bivalent metal ions, especially zinc, copper and nickel, as well as cobalt and rhodium. The biologically active form of plerixafor is its zinc complex.[11]
Synthesis
Three of the four nitrogen atoms of the macrocycle cyclam... (1,4,8,11-tetraazacyclotetradecane) are protected with tosyl groups. The product is treated with 1,4-bis(brommethyl)benzene and potassium carbonate in acetonitrile. After cleaving of the tosyl groups with hydrobromic acid, plerixafor octahydrobromide is obtained.[12]
History
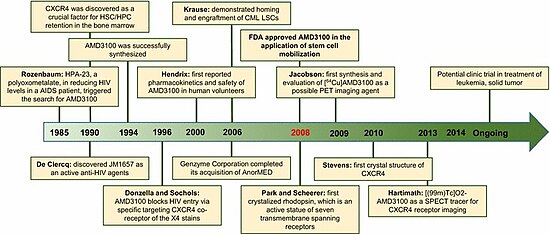
The molecule was first synthesised in 1987 to carry out basic studies on the redox chemistry of dimetallic coordination compounds.[14]
Then, it was serendipitously discovered by another chemist that such a molecule could have a potential use in the treatment of HIV because of its role in the blocking of CXCR4, a chemokine receptor which acts as a co-receptor for certain strains of HIV (along with the virus's main cellular receptor, CD4).[15]
Development of this indication was terminated because of lacking oral availability and cardiac disturbances. Further studies led to the new indication for cancer patients.[15]
Society and culture
Plerixafor has orphan drug status in the United States and European Union for the mobilization of hematopoietic stem cells. It was approved by the U.S. Food and Drug Administration (FDA) for this indication on 15 December 2008.[16] In the European Union, the drug was approved after a positive Committee for Medicinal Products for Human Use assessment report on 29 May 2009.[8] The drug was approved for use in Canada by Health Canada on 8 December 2011.[17]
Research
Anti-cancer properties
Plerixafor was seen to reduce metastasis in mice in several studies.[18] It has also been shown to reduce recurrence of glioblastoma in a mouse model after radiotherapy. In this model, the cancer cells that survived radiation critically depended on bone marrow derived cells for vasculogenesis, and the recruitment of the latter was mediated by SDF-1 CXCR4 interactions, which are blocked by plerixafor.[19]
Use in stem cell research
Researchers at Imperial College have demonstrated that plerixafor in combination with vascular endothelial growth factor (VEGF) can mobilise mesenchymal stem cells and endothelial progenitor cells into the peripheral blood of mice.[20]
In double‐blind, randomized, placebo‐controlled trial, stem cell mobilization with plerixafor did not improve healing of ischemic diabetic wounds.[21]
Neurologic
Blockade of CXCR4 signalling by plerixafor has also unexpectedly been found to be effective at counteracting opioid-induced hyperalgesia produced by chronic treatment with morphine, though only animal studies have been conducted as yet.[22]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 "Mozobil". Archived from the original on 23 July 2021. Retrieved 28 October 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Plerixafor Monograph for Professionals". Drugs.com. Archived from the original on 7 March 2016. Retrieved 28 October 2021.
- ↑ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 1079. ISBN 978-0857114105.
- ↑ "Mozobil Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 18 April 2021. Retrieved 28 October 2021.
- ↑ 5.0 5.1 "Plerixafor". Drugs in R&D. 8 (2): 113–119. 2007. doi:10.2165/00126839-200708020-00006. PMID 17324009.
- ↑ 6.0 6.1 6.2 6.3 6.4 Haberfeld, H, ed. (2009). Austria-Codex (in Deutsch) (2009/2010 ed.). Vienna: Österreichischer Apothekerverlag. ISBN 978-3-85200-196-8.
- ↑ Wagstaff, A. J. (2009). "Plerixafor: in patients with non-Hodgkin's lymphoma or multiple myeloma". Drugs. 69 (3): 319–26. doi:10.2165/00003495-200969030-00007. PMID 19275275.
- ↑ 8.0 8.1 "CHMP Assessment Report for Mozobil" (PDF). European Medicines Agency. Archived (PDF) from the original on 2018-06-18. Retrieved 2021-04-25.
- ↑ Kalatskaya, I.; Berchiche, Y. A.; Gravel, S.; Limberg, B. J.; Rosenbaum, J. S.; Heveker, N. (2009). "AMD3100 is a CXCR7 Ligand with Allosteric Agonist Properties". Molecular Pharmacology. 75 (5): 1240–7. doi:10.1124/mol.108.053389. PMID 19255243. S2CID 28540154.
- ↑ Cashen, A. F.; Nervi, B.; Dipersio, J. (2007). "AMD3100: CXCR4 antagonist and rapid stem cell-mobilizing agent". Future Oncology. 3 (1): 19–27. doi:10.2217/14796694.3.1.19. PMID 17280498.
- ↑ Esté, J. A.; Cabrera, C.; De Clercq, E.; Struyf, S.; Van Damme, J.; Bridger, G.; Skerlj, R. T.; Abrams, M. J.; Henson, G.; Gutierrez, A.; Clotet, B.; Schols, D. (1999). "Activity of different bicyclam derivatives against human immunodeficiency virus depends on their interaction with the CXCR4 chemokine receptor". Molecular Pharmacology. 55 (1): 67–73. doi:10.1124/mol.55.1.67. PMID 9882699.
- ↑ Bridger, G.; et al. (1993). "Linked cyclic polyamines with activity against HIV. WO/1993/012096".[permanent dead link]
- ↑ Liu, Tao; Li, Xiaobo; You, Shuo; Bhuyan, Soumitra S.; Dong, Lei (December 2015). "Effectiveness of AMD3100 in treatment of leukemia and solid tumors: from original discovery to use in current clinical practice". Experimental Hematology & Oncology. 5 (1). doi:10.1186/s40164-016-0050-5.
- ↑ Ciampolini, M.; Fabbrizzi, L.; Perotti, A.; Poggi, A.; Seghi, B.; Zanobini, F. (1987). "Dinickel and dicopper complexes with N,N-linked bis(cyclam) ligands. An ideal system for the investigation of electrostatic effects on the redox behavior of pairs of metal ions". Inorganic Chemistry. 26 (21): 3527–3533. doi:10.1021/ic00268a022.
- ↑ 15.0 15.1 Davies, S. L.; Serradell, N.; Bolós, J.; Bayés, M. (2007). "Plerixafor Hydrochloride". Drugs of the Future. 32 (2): 123. doi:10.1358/dof.2007.032.02.1071897.
- ↑ "Mozobil approved for non-Hodgkin's lymphoma and multiple myeloma" (Press release). Monthly Prescribing Reference. December 18, 2008. Archived from the original on January 6, 2009. Retrieved January 3, 2009.
- ↑ "Notice of Compliance information". Archived from the original on 2021-10-10. Retrieved 2021-04-25.
- ↑ Smith, M. C. P.; Luker, K. E.; Garbow, J. R.; Prior, J. L.; Jackson, E.; Piwnica-Worms, D.; Luker, G. D. (2004). "CXCR4 Regulates Growth of Both Primary and Metastatic Breast Cancer". Cancer Research. 64 (23): 8604–8612. doi:10.1158/0008-5472.CAN-04-1844. PMID 15574767.
- ↑ Kioi, M.; Vogel, H.; Schultz, G.; Hoffman, R. M.; Harsh, G. R.; Brown, J. M. (2010). "Inhibition of vasculogenesis, but not angiogenesis, prevents the recurrence of glioblastoma after irradiation in mice". Journal of Clinical Investigation. 120 (3): 694–705. doi:10.1172/JCI40283. PMC 2827954. PMID 20179352.
- ↑ Pitchford, S.; Furze, R.; Jones, C.; Wengner, A.; Rankin, S. (2009). "Differential Mobilization of Subsets of Progenitor Cells from the Bone Marrow". Cell Stem Cell. 4 (1): 62–72. doi:10.1016/j.stem.2008.10.017. hdl:10044/1/23497. PMID 19128793.
- ↑ Bonora, Benedetta Maria; Cappellari, Roberta; Mazzucato, Marta; Rigato, Mauro; Grasso, Marco; Menegolo, Mirko; Bruttocao, Andrea; Avogaro, Angelo; Fadini, Gian Paolo (2020). "Stem cell mobilization with plerixafor and healing of diabetic ischemic wounds: A phase IIA , randomized, double‐blind , placebo‐controlled trial". Stem Cells Translational Medicine. 9 (9): 965–973. doi:10.1002/sctm.20-0020. PMC 7445026. PMID 32485785. S2CID 219285881.
- ↑ Wilson NM, Jung H, Ripsch MS, Miller RJ, White FA (March 2011). "CXCR4 Signaling Mediates Morphine-induced Tactile Hyperalgesia". Brain, Behavior, and Immunity. 25 (3): 565–73. doi:10.1016/j.bbi.2010.12.014. PMC 3039030. PMID 21193025.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 Deutsch-language sources (de)
- All articles with dead external links
- Articles with dead external links from May 2020
- Articles with invalid date parameter in template
- Articles with permanently dead external links
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Chemicals using indexlabels
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from November 2018
- Orphan drugs
- Immunostimulants
- Nitrogen heterocycles
- Sanofi
- RTT