Transplant rejection
| Transplant rejection | |
|---|---|
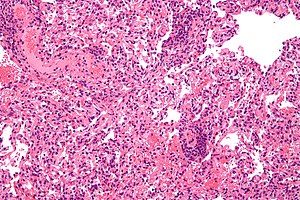 | |
| Micrograph showing lung transplant rejection. Lung biopsy. H&E stain. | |
| Specialty | Immunology |
| Treatment | Immunosuppressive drugs |
Transplant rejection occurs when transplanted tissue is rejected by the recipient's immune system, which destroys the transplanted tissue. Transplant rejection can be lessened by determining the molecular similitude between donor and recipient and by use of immunosuppressant drugs after transplant.[1]
Pretransplant rejection prevention
The first successful organ transplant, performed in 1954 by Joseph Murray, involved identical twins, and so no rejection was observed. Otherwise, the number of mismatched gene variants, namely alleles, encoding cell surface molecules called major histocompatibility complex (MHC), classes I and II, correlate with the rapidity and severity of transplant rejection. In humans MHC is also called human leukocyte antigen (HLA).[2]
Though cytotoxic-crossmatch assay can predict rejection mediated by cellular immunity, genetic-expression tests specific to the organ type to be transplanted, for instance AlloMap Molecular Expression Testing, have a high negative predictive value. Transplanting only ABO-compatible grafts (matching blood groups between donor and recipient) helps prevent rejection mediated by humoral immunity.[3]
ABO-incompatibility
Because very young children (generally under 12 months, but often as old as 24 months[4]) do not have a well-developed immune system,[5] it is possible for them to receive organs from otherwise incompatible donors. This is known as ABO-incompatible (ABOi) transplantation. Graft survival and patient mortality is approximately the same between ABOi and ABO-compatible (ABOc) recipients.[6] While focus has been on infant heart transplants, the principles generally apply to other forms of solid organ transplantation.[4]
The most important factors are that the recipient not have produced isohemagglutinins, and that they have low levels of T cell-independent antigens.[5][7] UNOS regulations allow for ABOi transplantation in children under two years of age if isohemagglutinin titers are 1:4 or below,[8][9] and if there is no matching ABOc recipient.[8][9][10] Studies have shown that the period under which a recipient may undergo ABOi transplantation may be prolonged by exposure to nonself A and B antigens.[11] Furthermore, should the recipient (for example, type B-positive with a type AB-positive graft) require eventual retransplantation, the recipient may receive a new organ of either blood type.[4][9]
Limited success has been achieved in ABO-incompatible heart transplants in adults,[12] though this requires that the adult recipients have low levels of anti-A or anti-B antibodies.[12] Kidney transplantation is more successful, with similar long-term graft survival rates to ABOc transplants.[9]
Immunologic mechanisms
Rejection is an adaptive immune response via cellular immunity (mediated by killer T cells inducing apoptosis of target cells) as well as humoral immunity (mediated by activated B cells secreting antibody molecules), though the action is joined by components of innate immune response (phagocytes and soluble immune proteins). Different types of transplanted tissues tend to favor different balances of rejection mechanisms.[13]
Immunization
An animal's exposure to the antigens of a different member of the same or similar species is allostimulation, and the tissue is allogenic. Transplanted organs are often acquired from a cadaver (usually a host who had succumbed to trauma), whose tissues had already sustained ischemia or inflammation.[citation needed]
Dendritic cells (DCs), which are the primary antigen-presenting cells (APCs), of the donor tissue migrate to the recipient's peripheral lymphoid tissue (lymphoid follicles and lymph nodes), and present the donor's self peptides to the recipient's lymphocytes (immune cells residing in lymphoid tissues). Lymphocytes include two classes that enact adaptive immunity, also called specific immunity. Lymphocytes of specific immunity T cells—including the subclasses helper T cells and killer T cells—and B cells.[citation needed]
The recipient's helper T cells coordinate specific immunity directed at the donor's self peptides or at the donor's Major histocompatibility complex molecules, or at both.
Immune memory
When memory helper T cells' CD4 receptors bind to the MHC class II molecules which are expressed on the surfaces of the target cells of the graft tissue, the memory helper T cells' T cell receptors (TCRs) can recognize their target antigen that is presented by the MHC class II molecules. The memory helper T cell subsequently produces clones that, as effector cells, secrete immune signalling molecules (cytokines) in approximately the cytokine balance that had prevailed at the memory helper T cell's priming to memorize the antigen. As the priming event in this instance occurred amid inflammation, the immune memory is pro-inflammatory.[citation needed]
Cellular immunity
Transplant rejection is a Type IV (“delayed”) hypersensitivity reaction mediated by T cells in which the transplant recipient’s T cells become alloreactive, recognizing major histocompatibility complex (MHC) antigens on the donated organ, and promote local immune and inflammatory responses to defend against the perceived threat.[14] The T cells can recognize the donated organ’s MHC antigens through one of two routes: a direct pathway, where the transplanted organ’s antigen-presenting cells (such as dendritic cells that contain MHC class I molecules) migrate to the recipient’s lymph nodes and activate T cells that reside there; or an indirect pathway, in which the recipient’s dendritic cells can endocytose donor proteins such as donor MHC proteins and activate T cells in secondary lymphoid tissues.[15] Alloreactive killer T cells, also called cytotoxic T lymphocytes (CTLs), have CD8 receptors that dock to the transplanted tissue's MHC class I molecules, which display the donor's self peptides. (In the living donor, such presentation of self antigens helped maintain self tolerance.) Thereupon, the T cell receptors (TCRs) of the killer T cells recognize their matching epitope, and trigger the target cell's programmed cell death by apoptosis.
Humoral immunity
Developed through an earlier primary exposure that primed specific immunity to the nonself antigen, a transplant recipient can have specific antibody crossreacting with the donor tissue upon the transplant event, a secondary exposure. This is typical of minor blood group exposure (e.g. Kell) following allogenic blood transfusion or trauma during pregnancy. At secondary exposure, these crossreactive antibody molecules interact with aspects of innate immunity—soluble immune proteins called complement and innate immune cells called phagocytes—which inflames and destroys the transplanted tissue.[citation needed]
Antibody
Secreted by an activated B cell, then called plasma cell, an antibody molecule is a soluble immunoglobulin (Ig) whose basic unit is shaped like the letter Y: the two arms are the Fab regions, while the single stalk is the Fc region. Each of the two tips of Fab region is the paratope, which binds a matching molecular sequence and its 3D shape (conformation), altogether called epitope, within the target antigen.
Opsonization
The IgG's Fc region also enables opsonization by a phagocyte, a process by which the Fc receptor on the phagocyte—such as neutrophils in blood and macrophages in tissues—binds the antibody molecule's FC stalk, and the phagocyte exhibits enhanced uptake of the antigen, attached to the antibody molecule's Fab region.[citation needed]
Complement cascade
When the paratope of Ig class gamma (IgG) binds its matching epitope, IgG's Fc region conformationally shifts and can host a complement protein, initiating the complement cascade that terminates by punching a hole in a cell membrane. With many holes so punched, fluid rushes into the cell and ruptures it.[citation needed]
Cell debris can be recognized as damage associated molecular patterns (DAMPs) by pattern recognition receptors (PRRs), such as Toll-like receptors (TLRs), on membranes of phagocytes, which thereupon secrete proinflammatory cytokines, recruiting more phagocytes to traffic to the area by sensing the concentration gradient of the secreted cytokines (chemotaxis).[citation needed]
| Tissue | Mechanism |
|---|---|
| Blood | Antibodies (isohaemagglutinins) |
| Kidney | Antibodies, cell-mediated immunity (CMI) |
| Heart | Antibodies, CMI |
| Skin | CMI |
| Bonemarrow | CMI |
| Cornea | Usually accepted unless vascularised: CMI |
Medical categories
Hyperacute rejection
Initiated by preexisting humoral immunity, hyperacute rejection manifests within minutes after transplant, and if tissue is left implanted brings systemic inflammatory response syndrome. Of high risk in kidney transplants is rapid clumping, namely agglutination, of red blood cells (RBCs or erythrocytes), as an antibody molecule binds multiple target cells at once.[citation needed]
While kidneys can routinely be obtained from human donors, most organs are in short supply leading to consideration of xenotransplants from other species. Pigs are especially likely sources for xenotransplants, chosen for the anatomical and physiological characteristics they share with humans.[16] However, the sugar galactose-alpha-1,3-galactose (αGal) has been implicated as a major factor in hyperacute rejection in xenotransplantation. Unlike virtually all other mammals, humans and other primates do not make αGal, and in fact recognize it as an antigen.[17] During transplantation, xenoreactive natural antibodies recognize αGal on the graft endothelium as an antigen, and the resulting complement-mediated immune response leads to a rejection of the transplant.[18]
Acute rejection
Developing with formation of cellular immunity, acute rejection occurs to some degree in all transplants, except between identical twins, unless immunosuppression is achieved (usually through drugs). Acute rejection begins as early as one week after transplant, the risk being highest in the first three months, though it can occur months to years later. Highly vascular tissues such as kidney or liver often host the earliest signs—particularly at endothelial cells lining blood vessels—though it eventually occurs in roughly 10 to 30% of liver transplants, and 10 to 20% of kidney transplants. A single episode of acute rejection can be recognized and promptly treated, usually preventing organ failure, but recurrent episodes lead to chronic rejection. It is believed that the process of acute rejection is mediated by the cell mediated pathway, specifically by mononuclear macrophages and T-lymphocytes. Histology of acute rejection is defined by dense lymphocytic cellular infiltrate as well as vasculitis of organ donor vessels.[citation needed]
Chronic rejection
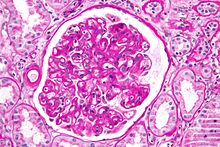
The term chronic rejection initially described long-term loss of function in transplanted organs via fibrosis of the transplanted tissue's blood vessels. This is now chronic allograft vasculopathy, however, leaving chronic rejection referring to rejection due to more patent aspects of immunity.[citation needed]
Chronic rejection explains long-term morbidity in most lung-transplant recipients,[19][20] the median survival roughly 4.7 years, about half the span versus other major organ transplants.[21] In histopathology the condition is bronchiolitis obliterans, which clinically presents as progressive airflow obstruction, often involving dyspnea and coughing, and the patient eventually succumbs to pulmonary insufficiency or secondary acute infection.[citation needed]
Airflow obstruction not ascribable to other cause is labeled bronchiolitis obliterans syndrome (BOS), confirmed by a persistent drop—three or more weeks—in forced expiratory volume (FEV1) by at least 20%.[22] BOS is seen in over 50% of lung-transplant recipients by 5 years, and in over 80% by ten years. First noted is infiltration by lymphocytes, followed by epithelial cell injury, then inflammatory lesions and recruitment of fibroblasts and myofibroblasts, which proliferate and secrete proteins forming scar tissue.[23] Generally thought unpredictable, BOS progression varies widely: lung function may suddenly fall but stabilize for years, or rapidly progress to death within a few months. Risk factors include prior acute rejection episodes, gastroesophageal reflux disease, acute infections, particular age groups, HLA mis-matching, lymphocytic bronchiolitis, and graft dysfunction (e.g., airway ischemia).[24]
Rejection due to non-adherence
One principal reason for transplant rejection is non-adherence to prescribed immunosuppressant regimens. This is particularly the case with adolescent recipients,[25] with non-adherence rates near 50% in some instances.[25]
Diagnosis
Diagnosis of acute rejection relies on clinical data—patient signs and symptoms but also calls on laboratory data such as blood or even tissue biopsy. The laboratory pathologist generally seeks three main histological signs: (1) infiltrating T cells, perhaps accompanied by infiltrating eosinophils, plasma cells, and neutrophils, particularly in telltale ratios, (2) structural compromise of tissue anatomy, varying by tissue type transplanted, and (3) injury to blood vessels. Tissue biopsy is restricted, however, by sampling limitations and risks/complications of the invasive procedure.[26][27][28] Cellular magnetic resonance imaging (MRI) of immune cells radiolabeled in vivo might—similarly to Gene Expression Profiling (GEP)—offer noninvasive testing.[29][30]
Treatment
Hyperacute rejection manifests severely and within minutes, and so treatment is immediate: removal of the tissue. Chronic rejection is generally considered irreversible and poorly amenable to treatment—only retransplant generally indicated if feasible—though inhaled ciclosporin is being investigated to delay or prevent chronic rejection of lung transplants. Acute rejection is treated with one or several of a few strategies. Despite treatment, rejection remains a major cause of transplant failure.[31]
Immunosuppressive therapy
A short course of high-dose corticosteroids can be applied, and repeated. Triple therapy adds a calcineurin inhibitor and an anti-proliferative agent. Where calcineurin inhibitors or steroids are contraindicated, mTOR inhibitors are used.
Immunosuppressive drugs:
- Corticosteroids
- Calcineurin inhibitors
- Anti-proliferatives
- mTOR inhibitors
Antibody-based treatments
Antibody specific to select immune components can be added to immunosuppressive therapy. The monoclonal anti-T cell antibody OKT3, once used to prevent rejection, and still occasionally used to treat severe acute rejection, has fallen into disfavor, as it commonly brings severe cytokine release syndrome and late post-transplant lymphoproliferative disorder. (OKT3 is available in the United Kingdom for named-patient use only.)
Antibody drugs:
- Monoclonal anti-IL-2Rα receptor antibodies
- Monoclonal anti-IL-6R receptor antibodies
- Polyclonal anti-T-cell antibodies
- Anti-thymocyte globulin (ATG)
- Anti-lymphocyte globulin (ALG)
- Monoclonal anti-CD20 antibodies
Blood transfer
Cases refractory to immunosuppressive or antibody therapy are sometimes treated with photopheresis, or extracorporeal photoimmune therapy (ECP), to remove antibody molecules specific to the transplanted tissue.
Marrow transplant
Bone marrow transplant can replace the transplant recipient's immune system with the donor's, and the recipient accepts the new organ without rejection. The marrow's hematopoietic stem cells—the reservoir of stem cells replenishing exhausted blood cells including white blood cells forming the immune system—must be of the individual who donated the organ or of an identical twin or a clone. There is a risk of graft-versus-host disease (GVHD), however, whereby mature lymphocytes entering with marrow recognize the new host tissues as foreign and destroy them.
Gene therapy
Gene therapy is another method that can be used. In this method, the genes that cause the body to reject transplants would be deactivated. Research is still being conducted, and no gene therapies are being used to date to treat patients.[32][33][34][35] Current research tends to focus on Th1 and Th17 which mediate allograft rejection via the CD4 and CD8 T cells.[36]
See also
References
- ↑ Frohn C, Fricke L, Puchta JC, Kirchner H (February 2001). "The effect of HLA-C matching on acute renal transplant rejection". Nephrology, Dialysis, Transplantation. 16 (2): 355–60. doi:10.1093/ndt/16.2.355. PMID 11158412. Archived from the original on 2016-05-16. Retrieved 2021-08-01.
- ↑ Delves PJ. "Human Leukocyte Antigen (HLA) System - Immunology; Allergic Disorders". Merck Manuals Professional Edition. Merck & Co. Archived from the original on 24 October 2020. Retrieved 30 September 2020.
- ↑ Parham, Peter (2015). The Immune System (4th ed.). New York: Garland Science. p. 437. ISBN 9781317511564.
- ↑ 4.0 4.1 4.2 ABO Incompatible Heart Transplantation in Young Infants. (2009, July 30). ABO Incompatible Heart Transplantation in Young Infants. American Society of Transplantation. Retrieved from "Archived copy". Archived from the original on 2013-12-20. Retrieved 2013-12-25.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ 5.0 5.1 West LJ (October 2011). "ABO-incompatible hearts for infant transplantation". Current Opinion in Organ Transplantation. 16 (5): 548–54. doi:10.1097/MOT.0b013e32834a97a5. PMID 21836514. S2CID 26070409.
- ↑ Saczkowski R, Dacey C, Bernier PL (June 2010). "Does ABO-incompatible and ABO-compatible neonatal heart transplant have equivalent survival?". Interactive Cardiovascular and Thoracic Surgery. 10 (6): 1026–33. doi:10.1510/icvts.2009.229757. PMID 20308266.
- ↑ Burch M, Aurora P (April 2004). "Current status of paediatric heart, lung, and heart-lung transplantation". Archives of Disease in Childhood. 89 (4): 386–9. doi:10.1136/adc.2002.017186. PMC 1719883. PMID 15033856.
- ↑ 8.0 8.1 United Network for Organ Sharing. (2013, January 31). OPTN Policy 3.7 - Allocation of Thoracic Organs. Retrieved from "Archived copy". Archived from the original on 2013-12-07. Retrieved 2013-12-25.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ 9.0 9.1 9.2 9.3 Urschel S, Larsen IM, Kirk R, Flett J, Burch M, Shaw N, et al. (March 2013). "ABO-incompatible heart transplantation in early childhood: an international multicenter study of clinical experiences and limits". The Journal of Heart and Lung Transplantation. 32 (3): 285–92. doi:10.1016/j.healun.2012.11.022. PMID 23305695.
- ↑ Almond CS, Gauvreau K, Thiagarajan RR, Piercey GE, Blume ED, Smoot LB, et al. (May 2010). "Impact of ABO-incompatible listing on wait-list outcomes among infants listed for heart transplantation in the United States: a propensity analysis". Circulation. 121 (17): 1926–33. doi:10.1161/CIRCULATIONAHA.109.885756. PMC 4273502. PMID 20404257.
- ↑ Fan X, Ang A, Pollock-Barziv SM, Dipchand AI, Ruiz P, Wilson G, et al. (November 2004). "Donor-specific B-cell tolerance after ABO-incompatible infant heart transplantation". Nature Medicine. 10 (11): 1227–33. doi:10.1038/nm1126. PMID 15502841. S2CID 26566529.
- ↑ 12.0 12.1 Tydén G, Hagerman I, Grinnemo KH, Svenarud P, van der Linden J, Kumlien G, Wernerson A (December 2012). "Intentional ABO-incompatible heart transplantation: a case report of 2 adult patients". The Journal of Heart and Lung Transplantation. 31 (12): 1307–10. doi:10.1016/j.healun.2012.09.011. PMID 23107062.
- ↑ Moreau, Aurélie (November 2013). "Effector Mechanisms of Rejection". Cold Spring Harbor Perspectives in Medicine. 3 (11): a015461. doi:10.1101/cshperspect.a015461. PMC 3808773. PMID 24186491. Archived from the original on 2021-05-06. Retrieved 2021-08-01.
- ↑ Marwa K, Kondamudi NP (2020). "Type IV Hypersensitivity Reaction". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 32965899. Archived from the original on 2021-01-10. Retrieved 2020-11-27.
- ↑ Ingulli E (January 2010). "Mechanism of cellular rejection in transplantation". Pediatric Nephrology. 25 (1): 61–74. doi:10.1007/s00467-008-1020-x. PMC 2778785. PMID 21476231.
- ↑ Klymiuk N, Aigner B, Brem G, Wolf E (March 2010). "Genetic modification of pigs as organ donors for xenotransplantation". Molecular Reproduction and Development. 77 (3): 209–21. doi:10.1002/mrd.21127. PMID 19998476. S2CID 11280460.
- ↑ Galili U (December 2005). "The alpha-gal epitope and the anti-Gal antibody in xenotransplantation and in cancer immunotherapy". Immunology and Cell Biology. 83 (6): 674–86. doi:10.1111/j.1440-1711.2005.01366.x. PMID 16266320. S2CID 85941122.
- ↑ Candinas D, Adams DH (February 2000). "Xenotransplantation: postponed by a millennium?". QJM. 93 (2): 63–6. doi:10.1093/qjmed/93.2.63. PMID 10700475.
- ↑ Jaramillo A, Fernández FG, Kuo EY, Trulock EP, Patterson GA, Mohanakumar T (February 2005). "Immune mechanisms in the pathogenesis of bronchiolitis obliterans syndrome after lung transplantation". Pediatric Transplantation. 9 (1): 84–93. doi:10.1111/j.1399-3046.2004.00270.x. PMID 15667618. S2CID 25841425.
- ↑ Lau CL, Patterson GA (November 2003). "Current status of lung transplantation". The European Respiratory Journal. Supplement. 47: 57s–64s. doi:10.1183/09031936.03.00022103. PMID 14621118.
- ↑ http://www.OPTN.org Archived 2011-11-11 at the Wayback Machine
- ↑ Lama VN, Murray S, Lonigro RJ, Toews GB, Chang A, Lau C, et al. (June 2007). "Course of FEV(1) after onset of bronchiolitis obliterans syndrome in lung transplant recipients". American Journal of Respiratory and Critical Care Medicine. 175 (11): 1192–8. doi:10.1164/rccm.200609-1344OC. PMC 1899272. PMID 17347496.
- ↑ Nicod LP (July 2006). "Mechanisms of airway obliteration after lung transplantation". Proceedings of the American Thoracic Society. 3 (5): 444–9. doi:10.1513/pats.200601-007AW. PMID 16799090.
- ↑ Belperio JA, Weigt SS, Fishbein MC, Lynch JP (January 2009). "Chronic lung allograft rejection: mechanisms and therapy". Proceedings of the American Thoracic Society. 6 (1): 108–21. doi:10.1513/pats.200807-073GO. PMID 19131536. S2CID 23071315. Archived from the original on 2021-10-24. Retrieved 2021-08-01.
- ↑ 25.0 25.1 Dobbels F, Hames A, Aujoulat I, Heaton N, Samyn M (February 2012). "Should we retransplant a patient who is non-adherent? A literature review and critical reflection". Pediatric Transplantation. 16 (1): 4–11. doi:10.1111/j.1399-3046.2011.01633.x. PMID 22248250. S2CID 1895827.
{{cite journal}}: CS1 maint: url-status (link) - ↑ "Kidney biopsy:risks". Archived from the original on 2017-11-11. Retrieved 2021-08-01.
- ↑ "Heart biopsy:risks". Archived from the original on 2021-04-29. Retrieved 2021-08-01.
- ↑ "Lung biopsy:risks". Archived from the original on 2018-07-29. Retrieved 2021-08-01.
- ↑ Hitchens TK, Ye Q, Eytan DF, Janjic JM, Ahrens ET, Ho C (April 2011). "19F MRI detection of acute allograft rejection with in vivo perfluorocarbon labeling of immune cells". Magnetic Resonance in Medicine. 65 (4): 1144–53. doi:10.1002/mrm.22702. PMC 3135171. PMID 21305593.
- ↑ Gheith OA (2011). "Gene expression profiling in organ transplantation". International Journal of Nephrology. 2011: 180201. doi:10.4061/2011/180201. PMC 3154482. PMID 21845224.
- ↑ Naesens M, Kuypers DR, De Vusser K, Evenepoel P, Claes K, Bammens B, et al. (August 2014). "The histology of kidney transplant failure: a long-term follow-up study". Transplantation. 98 (4): 427–35. doi:10.1097/TP.0000000000000183. PMID 25243513. S2CID 20703626.
- ↑ "Gene group responsible for transplant rejection: the Major_histocompatibility_complex". Archived from the original on 2021-05-06. Retrieved 2021-08-01.
- ↑ Yang, JY; Sarwal, MM (May 2017). "Transplant genetics and genomics". Nat Rev Genet. 18 (5): 309–326. doi:10.1038/nrg.2017.12. PMID 28286337.
- ↑ "Gene Therapy Progress and Prospects: Gene therapy in organ transplantation". Archived from the original on 2017-04-28. Retrieved 2021-08-01.
- ↑ "Gene therapy in transplantation". Archived from the original on 2015-11-05. Retrieved 2021-08-01.
- ↑ "The John Iacomini Lab". Archived from the original on 2018-04-23. Retrieved 2021-08-01.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 maint: archived copy as title
- Webarchive template wayback links
- CS1 maint: url-status
- Missing redirects
- Articles with hatnote templates targeting a nonexistent page
- All articles with unsourced statements
- Articles with unsourced statements from August 2020
- Articles with invalid date parameter in template
- Immune system disorders
- Transplantation medicine