Ischemia
| Ischemia | |
|---|---|
| Other names: Ischaemia, ischæmia | |
 | |
| Vascular ischemia of the toes with characteristic cyanosis | |
| Pronunciation | |
| Specialty | Vascular surgery |
| Usual onset | Gradual or sudden[3] |
| Types | Ischemic heart disease, stroke, peripheral vascular disease, mesenteric ischemia[4] |
| Causes | Gradual: Atherosclerosis, fibromuscular dysplasia, vasculitis, tumor[3] Sudden: Blot clot, vasospasm, aortic dissection[3][5] |
Ischemia is a restriction of the blood supply to tissue resulting in a shortage of oxygen.[3] Types include ischemic heart disease, stroke, peripheral vascular disease, and mesenteric ischemia.[4] Symptoms depend on the area of the body affected.[4] Onset may be gradual or sudden.[3]
Gradual cases may occur due to atherosclerosis, fibromuscular dysplasia, vasculitis, or external pressure from a tumor.[3] Sudden cases may occur due to a blot clot, vasospasm, or aortic dissection.[3][5]
Efforts to prevent the condition include a healthy lifestyle.[4] The first documented use of the term is from 1855.[2] The term is from the Greek ischein meaning "to restrain" plus haima meaning "blood".[2]
Signs and symptoms
Since oxygen is carried to tissues in the blood, insufficient blood supply causes tissue to become starved of oxygen. In the highly metabolically active tissues of the heart and brain, irreversible damage to tissues can occur in as little as 3–4 minutes at body temperature. The kidneys are also quickly damaged by loss of blood flow (renal ischemia). Tissues with slower metabolic rates may undergo irreversible damage after 20 minutes.
Without immediate intervention, ischemia may progress quickly to tissue necrosis and gangrene within a few hours. Paralysis is a very late sign of acute arterial ischemia and signals the death of nerves supplying the extremity. Foot drop may occur as a result of nerve damage. Because nerves are extremely sensitive to hypoxia, limb paralysis or ischemic neuropathy may persist after revascularization and may be permanent.[6]
Heart
Cardiac ischemia may be asymptomatic or may cause chest pain, known as angina pectoris. It occurs when the heart muscle, or myocardium, receives insufficient blood flow. This most frequently results from atherosclerosis, which is the long-term accumulation of cholesterol-rich plaques in the coronary arteries. Ischemic heart disease is the most common cause of death in most Western countries and a major cause of hospital admissions.[7]
Bowel
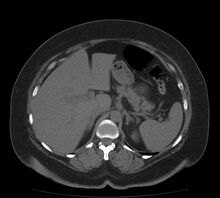
Both large and small bowel can be affected by ischemia. Ischemia of the large intestine may result in an inflammatory process known as ischemic colitis. Ischemia of the small bowel is called mesenteric ischemia.
Brain
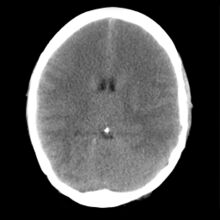
Brain ischemia is insufficient blood flow to the brain, and can be acute or chronic. Acute ischemic stroke is a neurologic emergency that may be reversible if treated rapidly. Chronic ischemia of the brain may result in a form of dementia called vascular dementia.[8] A brief episode of ischemia affecting the brain is called a transient ischemic attack (TIA), often called a mini-stroke. 10% of TIAs will develop into a stroke within 90 days, half of which will occur in the first two days following the TIA.[9]
Limb
Lack of blood flow to a limb results in acute limb ischemia. Symptoms of acute limb ischemia (which can be summarized as the "six P's") include pain, pallor, pulseless, paresthesia, paralysis, and poikilothermia.[10]
Cutaneous
Reduced blood flow to the skin layers may result in mottling or uneven, patchy discoloration of the skin
Causes
Ischemia is a vascular disease involving an interruption in the arterial blood supply to a tissue, organ, or extremity that, if untreated, can lead to tissue death. It can be caused by embolism, thrombosis of an atherosclerotic artery, or trauma. Venous problems like venous outflow obstruction and low-flow states can cause acute arterial ischemia. An aneurysm is one of the most frequent causes of acute arterial ischemia. Other causes are heart conditions including myocardial infarction, mitral valve disease, chronic atrial fibrillation, cardiomyopathies, and prosthesis, in all of which thrombi are prone to develop.[6]
Occlusion
The thrombi may dislodge and may travel anywhere in the circulatory system, where they may lead to pulmonary embolus, an acute arterial occlusion causing the oxygen and blood supply distal to the embolus to decrease suddenly. The degree and extent of symptoms depend on the size and location of the obstruction, the occurrence of clot fragmentation with embolism to smaller vessels, and the degree of peripheral arterial disease (PAD).[6]
- Thromboembolism (blood clots)
- Embolism (foreign bodies in the circulation, e.g. amniotic fluid embolism)
Trauma
Traumatic injury to an extremity may produce partial or total occlusion of a vessel from compression, shearing, or laceration. Acute arterial occlusion may develop as a result of arterial dissection in the carotid artery or aorta or as a result of iatrogenic arterial injury (e.g., after angiography).[6]
Other
An inadequate flow of blood to a part of the body may be caused by any of the following:
- Thoracic outlet syndrome (compression of the brachial plexus)
- Atherosclerosis (lipid-laden plaques obstructing the lumen of arteries)
- Hypoglycemia (lower than normal level of glucose)
- Tachycardia (abnormally rapid beating of the heart)
- Radiotherapy
- Hypotension (low blood pressure, e.g. in septic shock, heart failure)
- Outside compression of a blood vessel, e.g. by a tumor or in the case of superior mesenteric artery syndrome
- Sickle cell disease (abnormally shaped red blood cells)
- Induced g-forces which restrict the blood flow and force the blood to the extremities of the body, as in acrobatics and military flying
- Localized extreme cold, such as by frostbite or improper cold compression therapy
- Tourniquet application
- An increased level of glutamate receptor stimulation[citation needed]
- Arteriovenous malformations and peripheral artery occlusive disease
- rupture of significant blood vessels supplying a tissue or organ.
- Anemia vasoconstricts the periphery so that red blood cells can work internally on vital organs such as the heart, brain, etc., thus causing lack of oxygen to the periphery.
- Premature discontinuation of any oral anticoagulant.
- Unconsciousness, such as due to the ingestion of excessive doses of central depressants like alcohol or opioids, can result in ischemia of the extremities due to unusual body positions that prevent normal circulation
Pathophysiology

Ischemia results in tissue damage in a process known as ischemic cascade. The damage is the result of the build-up of metabolic waste products, inability to maintain cell membranes, mitochondrial damage, and eventual leakage of autolyzing proteolytic enzymes into the cell and surrounding tissues.
Restoration of blood supply to ischemic tissues can cause additional damage known as reperfusion injury that can be more damaging than the initial ischemia. Reintroduction of blood flow brings oxygen back to the tissues, causing a greater production of free radicals and reactive oxygen species that damage cells. It also brings more calcium ions to the tissues causing further calcium overloading and can result in potentially fatal cardiac arrhythmias and also accelerates cellular self-destruction. The restored blood flow also exaggerates the inflammation response of damaged tissues, causing white blood cells to destroy damaged cells that may otherwise still be viable.[11]
Treatment
Early treatment is essential to keep the affected limb viable. The treatment options include injection of an anticoagulant, thrombolysis, embolectomy, surgical revascularisation, or partial amputation. Anticoagulant therapy is initiated to prevent further enlargement of the thrombus. Continuous IV unfractionated heparin has been the traditional agent of choice.[6]
If the condition of the ischemic limb is stabilized with anticoagulation, recently formed emboli may be treated with catheter-directed thrombolysis using intra-arterial infusion of a thrombolytic agent (e.g., recombinant tissue plasminogen activator (tPA), streptokinase, or urokinase). A percutaneous catheter inserted into the femoral artery and threaded to the site of the clot is used to infuse the drug. Unlike anticoagulants, thrombolytic agents work directly to resolve the clot over a period of 24 to 48 hours.[6]
Direct arteriotomy may be necessary to remove the clot. Surgical revascularization may be used in the setting of trauma (e.g., laceration of the artery). Amputation is reserved for cases where limb salvage is not possible. If the patient continues to have a risk of further embolization from some persistent source, such as chronic atrial fibrillation, treatment includes long-term oral anticoagulation to prevent further acute arterial ischemic episodes.[6]
Decrease in body temperature reduces the aerobic metabolic rate of the affected cells, reducing the immediate effects of hypoxia. Reduction of body temperature also reduces the inflammation response and reperfusion injury. For frostbite injuries, limiting thawing and warming of tissues until warmer temperatures can be sustained may reduce reperfusion injury.
Ischemic stroke is at times treated with various levels of statin therapy at hospital discharge, followed by home time, in an attempt to lower the risk of adverse events.[12][13]
Word origin
The word ischemia (/ɪˈskiːmiə/) is from Greek ἴσχαιμος iskhaimos, "staunching blood" from ἴσχω iskhο, "keep back, restrain" and αἷμα haima, "blood".
See also
- Infarction
- Inhibitor protein
- Trauma triad of death
- Ischemia-reperfusion injury of the appendicular musculoskeletal system
References
- ↑ Martin, Elizabeth A. (1987). Concise Medical Dictionary. Oxford University Press. p. 107. ISBN 978-0-19-281991-8. Archived from the original on 2021-04-27. Retrieved 2016-05-15.
- ↑ 2.0 2.1 2.2 "Definition of ISCHEMIA". www.merriam-webster.com. Archived from the original on 26 January 2021. Retrieved 27 February 2021.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 "Occlusive Peripheral Arterial Disease - Heart and Blood Vessel Disorders". Merck Manuals Consumer Version. Archived from the original on 19 March 2021. Retrieved 27 February 2021.
- ↑ 4.0 4.1 4.2 4.3 "What Is Ischemia?". WebMD. Archived from the original on 1 March 2021. Retrieved 27 February 2021.
- ↑ 5.0 5.1 Hotchkiss, R; Marks, T (March 2014). "Management of acute and chronic vascular conditions of the hand". Current reviews in musculoskeletal medicine. 7 (1): 47–52. doi:10.1007/s12178-014-9202-6. PMID 24668045.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 Lewis. S.L (2008). Medical-Surgical Nursing (7th ed.). Vascular disorder. pp. 907–908.
- ↑ World Health Organization Department of Health Statistics and Informatics in the Information, Evidence and Research Cluster (2004). The global burden of disease 2004 update. Geneva: WHO. ISBN 92-4-156371-0.
- ↑ Kuźma, Elżbieta; Lourida, Ilianna; Moore, Sarah F.; Levine, Deborah A.; Ukoumunne, Obioha C.; Llewellyn, David J. (August 2018). "Stroke and dementia risk: A systematic review and meta-analysis". Alzheimer's & Dementia. 0 (11): 1416–1426. doi:10.1016/j.jalz.2018.06.3061. hdl:2027.42/152961. ISSN 1552-5260. PMC 6231970. PMID 30177276. Archived from the original on 2021-08-28. Retrieved 2018-09-07.
- ↑ "What is TIA?". Stroke.org. Archived from the original on 2014-10-25. Retrieved 2015-08-19.
- ↑ "Vascular and Interventional Radiology: The Requisites (2nd Edition), John A. Kaufman & Michael J. Lee, Publisher:Elsevier - Active as of 11/13/2014". Archived from the original on 2019-12-17. Retrieved 2014-11-14.
- ↑ Sims N.R.; Muyderman H. (2010). "Mitochondria, oxidative metabolism and cell death in stroke" (PDF). Biochimica et Biophysica Acta. 1802 (1): 80–91. doi:10.1016/j.bbadis.2009.09.003. PMID 19751827. Archived (PDF) from the original on 2020-08-01. Retrieved 2019-09-03.
- ↑ Li, Yi-Heng; Ueng, Kwo-Chang; Jeng, Jiann-Shing; Charng, Min-Ji; Lin, Tsung-Hsien; Chien, Kuo-Liong; Wang, Chih-Yuan; Chao, Ting-Hsing; Liu, Ping-Yen (2017-04-01). "2017 Taiwan lipid guidelines for high risk patients". Journal of the Formosan Medical Association. 116 (4): 217–248. doi:10.1016/j.jfma.2016.11.013. ISSN 0929-6646. PMID 28242176.
- ↑ O’Brien, Emily C.; Greiner, Melissa A.; Xian, Ying; Fonarow, Gregg C.; Olson, DaiWai M.; Schwamm, Lee H.; Bhatt, Deepak L.; Smith, Eric E.; Maisch, Lesley (2015-10-13). "Clinical Effectiveness of Statin Therapy After Ischemic Stroke: Primary Results From the Statin Therapeutic Area of the Patient-Centered Research Into Outcomes Stroke Patients Prefer and Effectiveness Research (PROSPER) Study". Circulation. 132 (15): 1404–1413. doi:10.1161/CIRCULATIONAHA.115.016183. ISSN 0009-7322. PMID 26246175.