Ethinylestradiol/desogestrel
 Ethinylestradiol | |
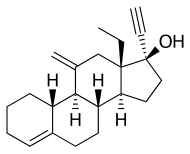 Desogestrel | |
| Combination of | |
|---|---|
| Ethinylestradiol | Estrogen |
| Desogestrel | Progestogen; Progestin |
| Names | |
| Trade names | Gracial, Marvelon, Mercilon, Novynette, Regulon, others |
| Other names | EE/DSG |
| Clinical data | |
| Drug class | Estrogen; Progestin; Progestogen |
| Routes of use | By mouth |
| Legal | |
| Legal status |
|
| Identifiers | |
| CAS Number |
|
| ATC code | |
Ethinylestradiol/desogestrel (EE/DSG), sold under the brand name Marvelon among others, is a combination of ethinylestradiol (EE), an estrogen, and desogestrel (DSG), a progestin, which is used as a birth control pill to prevent pregnancy in women.[1][2] It is taken by mouth and contains 30 μg EE and 0.15 mg DSG per tablet (brand names Marvelon, others) or 20 μg EE and 0.15 mg DSG per tablet (brand names Mercilon, others).[1][2] EE/DSG is marketed widely throughout the world.[3]
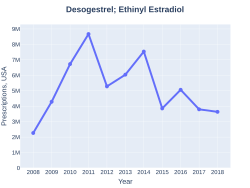
In 2017, it was the 164th most commonly prescribed medication in the United States, with more than three million prescriptions.[4][5]
Society and culture
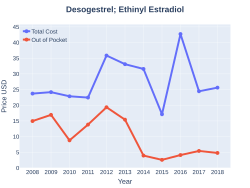
Cost
The cost for this medication in the U.S. is $126 (USD) for 84 tablets[6]In 2017, it was the 164th most commonly prescribed medication in the United States, with more than three million prescriptions.[4][5]
-
DesogestrelEthinylEstradiol costs (US)
-
DesogestrelEthinylEstradiol prescriptions (US)
See also
References
- ↑ 1.0 1.1 Pundir J, Coomarasamy A (21 April 2016). Gynaecology: Evidence-Based Algorithms. Cambridge University Press. pp. 260–. ISBN 978-1-316-30110-4. Archived from the original on 28 August 2021. Retrieved 8 March 2021.
- ↑ 2.0 2.1 IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; World Health Organization; International Agency for Research on Cancer (2007). Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy. World Health Organization. pp. 435, 436, 455, 459. ISBN 978-92-832-1291-1. Archived from the original on 2021-08-28. Retrieved 2021-03-08.
- ↑ "Desogestrel". Archived from the original on 2017-08-03. Retrieved 2021-03-08.
- ↑ 4.0 4.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 5.0 5.1 "Desogestrel; Ethinyl Estradiol - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
- ↑ "Desogestrel/ethinyl estradiol Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 16 February 2021. Retrieved 27 March 2021.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
This drug article relating to the genito-urinary system is a stub. You can help MDWiki by expanding it. |
- Pages using duplicate arguments in template calls
- Infobox drug articles with non-default infobox title
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Drugs that are a combination of chemicals
- Combined oral contraceptives
- All stub articles
- Genito-urinary system drug stubs